2018.03.02.13
Files > Volume 3 > Vol 3 No 2 2018
NOTICIAS Y OPINIONES / NEWS AND VIEWS
Schizophrenia related genes and its molecular treatments: systematic review and analysis
Los genes relacionados con la esquizofrenia y sus tratamientos moleculares: revisión sistemática y análisis
Isaac Bravo, Freddy Figueroa, Cristhian Preciado
Available from: http://dx.doi.org/10.21931/RB/2018.03.02.13
ABSTRACT
Schizophrenia is a mental disorder characterized by strong psychotic episodes of hallucinations and delusions, which affects, according to statistics, 1% of the world population. Although its pathogenesis remains unknown, multiple investigations suggest that several associations of chromosomal locus are responsible in the neurodevelopment of the disorder. This polygenic theory, shared by diseases such as cancer and diabetes, can also be influenced by environmental and epigenetic factors. Despite the existence of antipsychotic drugs that partially control the disease, these in many cases suppose counterproductive and ineffective effects. That is why a new research line on epigenetic factors and chromosomal associations, linked to the disorder origin, could lead to new pharmacological treatments that represent a real breakthrough.
Keywords: schizophrenia, candidate’s genes, locus, dopamine, drugs, neurodevelopment, models.
RESUMEN
La esquizofrenia es un trastorno mental caracterizado por sus fuertes episodios psicóticos de alucinaciones y delirios, el cual afecta, según las estadísticas, al 1% de la población mundial. Aunque su patogénesis sigue siendo desconocida, múltiples investigaciones apuntan que varias asociaciones de locis cromosomales tienen relevancia en el neurodesarrollo del desorden. Esta teoría poligénica, la cual comparten enfermedades como el cáncer y la diabetes, también puede estar influenciada por factores ambientales y epigenéticos. A pesar de la existencia de fármacos antipsicóticos que controlan la enfermedad parcialmente, estos suponen en numerosos casos efectos contraproducentes y poco eficaces. Es por ello que una nueva línea investigativa sobre factores epigenéticos y asociaciones cromosomales, ligados a su origen, podrían conducir a nuevos tratamientos farmacológicos que supongan un verdadero avance.
Palabras Clave: esquizofrenia, genes candidatos, locus, dopamina, fármacos, neurodesarrollo, modelos.
INTRODUCTION
Schizophrenia is considered a mental disorder that severely disrupts behavior, creating classic psychotic episodes of strong hallucinations and delusions.1,2 This disorder involves several neuromorphological and neurochemical abnormalities.3 Among its symptoms, we can find the reduction of emotions, the disorganization of thought, the generation of language deficit, a several motivation decrease and the decrease on cognitive function.1,4Schizophrenia is classified as the disease with the highest degree of chronic disability. Statistics show that at least 1% of the world population suffers schizophrenia,3,5 and that the hereditary factor of the disorder is between 80-85%6. Due to its high heritability index, its strong social impact and its devastating consequences, it has been named as one of the worst diseases affecting humanity.1,7 It is also proven that it is one of the diseases whose treatment is one of the most expensive in the world.2,7
Although there is not enough evidence about the causes that originate schizophrenia, its pathogenesis is considered as a set of neurochemical factors associated with a cerebral malformation, possibly due to viral infections during pregnancy or obstetric complications.1,8 In recent decades much emphasis has been placed on this idea, and it is believed that the origin of schizophrenia is a consequence of possible neuronal alterations during the development of the central nervous system (CNS)7,9. These alterations, even if they can be inherited or associated with the genetic load, also can be induced by environmental or epigenetic factors.7
Thanks to advances in the study of genomics and the latest therapeutic tools developed in molecular biology, it is possible to perform analyzes in the genome of a population affected with this disease, and thus determine the genes involved in the formation of the disease.10 Through investigations into the genetic material, it has been possible to deepen into the study of candidate genes or genes involved in the genomic alterations of people suffering from schizophrenic disorder.11 In this way, several chromosomal sectors have been selected, as well as small loci in certain chromosomes that may be related to the pathology.10
The purpose of the present article is to analyze multiple works and investigations about the different genes involved in the malformation of the CNS. Also, to explain the interaction of these genes with the disease and discuss the repercussion of alterations in the genome by epigenetic and environmental factors. Another objective of the article is to present the current molecular mechanisms that rule schizophrenia. We’ll discuss the main actual molecular treatments applied to schizophrenia, make a comparison between the different techniques and present them according to their performance and efficiency. And finally, the effects of cannabis use on schizophrenia will be introduced and discussed.
Genetics of Schizophrenia
Recent genetic studies on the etiology of schizophrenia have been based on subjects such as the inheritance shared by consanguineous, the localization of possible chromosome groups responsible for the pathology and hypothetical genetic mechanisms which participate in the formation of the disorder, among others. Consequently, several models of transmission and genetic origin have been proposed for the disease.12 Among these we have:
Monogenic model: Which shows that all types of schizophrenia share the same gene but with a different level of penetration. The fact that only between 30% and 35% of the offspring of schizophrenic patients develop the pathology discredits this model.
Heterogenetic Model: It defines schizophrenia as a set of pathologies, associated with a greater locus, which are transmitted by inheritance, either dominant, recessive or linked to the X chromosome. However, there is insufficient data or analysis in favour of this hypothesis.
Polygenic Model: It maintains that schizophrenia comes from the combination of multiple defective genes, with several interactions with the environment. This model proposes that the risk of developing schizophrenia is linked to a continuous distribution trait. This would explain why there are subclinical expressions in twins and other relatives of schizophrenic patients.9
In other studies, have been developed meta-analysis of the significance that has the inheritance for the development of this disorder, showing a rate of 80 to 85%.13 In the same way they have calculated that the weight of the environmental factors is almost 11%. In relation to these studies, it has noted the existence of multiple chromosomal abnormalities; although it has not been possible to find a candidate gene of high reliability, several chromosome loci (as shown in Fig. 1) have been proposed that present susceptibility to the disease that include 1q21-22, 1q32-43, 6p24, 8p21, 10p14, 13q32, 18p11 and 22q11-13.10,14
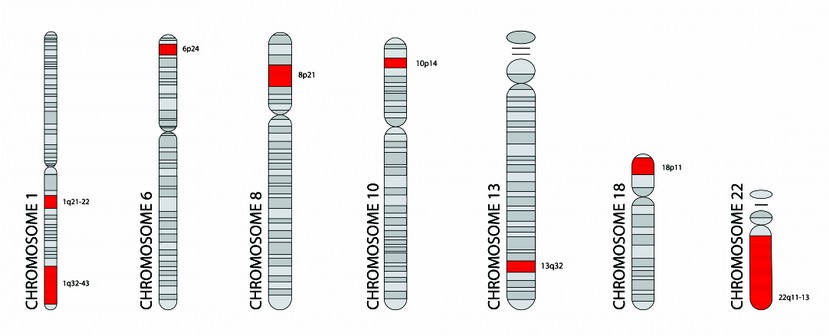
Figure 1.- Chromosomal map with the locations of genes, marked with a red strip, related to some susceptibility to schizophrenia disorder, as Harrison P and Owen M. mentioned on their work.
A study on candidate genes, made in the Research Center of Cellular and Molecular Biology in Costa Rica, established that the chromosomes that would be linked with the development of schizophrenia within the population would be 1, 8, 13, 18 and 22.15 But in view of the fact that none of these can cause the disease by itself, it has come to be the assumption that several genes could be combined in different ways in a number of individuals to generate the disease, and has created a possible model that integrates environmental factors in relation to the responsible genes. On the other hand, one of the major investigations carried out on schizophrenia, where 36989 cases and 113075 controls were studied and controlled, respectively, concluded with the finding of at least 128 genomic interactions between 108 chromosomal loci.16 It was also determined that these associations were more numerous and frequent on chromosome 6.17,18
Genetic and epidemiologic studies suggest to the polygenic model like a possible answer to the etiology of the schizophrenia, giving as an hypothesis the idea of multiple loci in the human genetic code, which provide minuscule individual effects increasing in the aggregation the risk of developing the disorder of substantial form.13,19 While the study and follow-up of individual cases provide strong inclinations towards the acceptance of a multigenic model, the study in families with schizophrenic history shows that like cancer, diabetes or diseases Cardiovascular, schizophrenia is a multiple genomic distortion characterized by a group of numerous "small" genetic inputs for the development of the disease.20 Other candidate genes that are mentioned by the Research Center of Cellular and Molecular Biology in Costa Rica are21:
NOTCH4: It’s a gene located on the chromosomal locus 6p21.3 (Fig. 2), that whose encoded protein participates in the regulation of all types of tissues and organisms, giving a fate to cell organizations that receive their signal and those that do not. Animal studies show that the NOTCH family of proteins determines cell fate. Therefore, some authors find an association between this gene and the risk of schizophrenia.21
Dysbindin or Dystrobrevin-binding Protein-1: It’s a neural tissue protein in which gene is located on chromosome 6p22.3 (Fig. 2). It forms part of the dystrophin complex. Its expression is given in the presynaptic terminals and participates in signal transduction. This protein can also decrease glutamate signaling, which links it as a precursor to schizophrenia.21
Neuregulin 1: This cell adhesion molecule (CAM), whose gene is located on chromosome 8 (Fig. 2), is consider to be a promoter of neuronal migration and cell differentiation. It is expressed in glutaminergic synaptic vesicles acting by means of NMDA receptors on the expression of glutamate and other neurotransmitter receptors.21,22
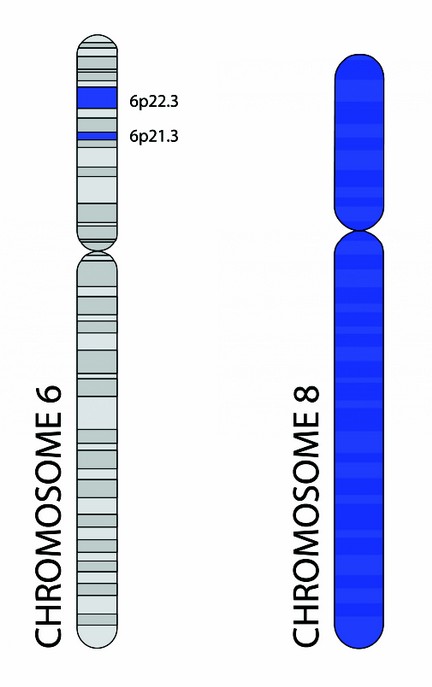
Figure 2.- Chromosome 6 showing the locations of NOTCH4 gene and Dysbindin Protein 1 gene, marked with a blue strip, and chromosome 8.
Epigenetic studies have shown that environmental changes and epigenetic modifications in DNA have a fundamental role in prenatal and postnatal neurodevelopment.23 So much so that the epigenetic regulation during the formation of the CNS is a crucial point for the possible origin of the disease.24 A recent publication in Nature Neuroscience, after reviewing around 526 samples of post-mortem brains, asserts that any significant changes in epigenetic mechanisms during genome regulation in the stages of brain formation may be crucial to the onset of schizophrenic disorder. This is why there is a close relationship between epigenetic intermediaries and factors that give rise to the disease.21
Molecular mechanisms linked to Schizophrenia
Once the different hypotheses about the etiological part of schizophrenia are approached and explained from a totally genetic focus, it’s time to introduce the molecular mechanisms associated with the disorder. We must know that there are several hypotheses linked to the molecular mechanisms that trigger the symptoms of schizophrenia. The dopaminergic hypothesis is the one that has prevailed during the last decades, being even the most accepted model by the scientific community. This hypothesis holds that the symptoms of this disorder are due to an excess of dopamine or a high sensitivity of this neurotransmitter. Evidence shows that dopamine is one of the neurotransmitters responsible for the genesis of schizophrenia and also affects other neurotransmission systems. It is also suggested that the disorder may be closely linked to a hyperactivation of cerebral dopaminergic pathways. Therefore, it is suggested that schizophrenia is linked to dysregulation of the dopaminergic factor.11,23
On the other hand, another hypothesis currently accepted for the community involves the neurotransmitter serotonin as the main responsible for the disease. Wooley and Shaw (1994) pioneers of the hypothesis, which states that schizophrenia is due to a deficiency in serotonergic function in the CNS. In addition, it describes the trophic role of serotonin in neurodevelopment, its activity in the dopaminergic system and its effects in the prefrontal cortex through 5HT2A receptors. In fact, it has been proving that serotonin has an inhibitory effect in the dopaminergic complex system. So the antipsychotic arresters of serotonergic factors disinhibit the dopaminergic factors improving the patient's behavior.25,26
Most common treatments against Schizophrenia
Schizophrenia for some time that is one of the most studied diseases by the human being, therefore many treatments have been elaborated by doctors and investigators to prevent, to cure or to attenuate the complications that this disorder entails. These treatments vary according to the phase or severity of the disease. One of the suggested treatments, based on all of these studies, is primarily on the use of antipsychotic drugs (AP). Although this treatment has its limitations and adverse effects, it is a proven fact that its application can significantly improve the symptoms of schizophrenia. The selection of these AP's, in turn, is delimited by certain parameters such as the antecedents of response to previous treatments, the profile of adverse effects of the drug, the cost of production and its possible side effects.27,28
There are two types of antipsychotics used as medication in schizophrenia: Typical or classic antipsychotics, and atypical or recent. The first ones are characterized by being competitive antagonists of the dopaminergic receptors. These are effective but do not provide optimal treatment, because they produce many undesirable effects. This is why they are considered to be of low acceptance by the patients. On the other hand, the atypical AP focus on the increase of the selectivity antidopaminergic or that are antiserotoninergics and at the same time antidopaminergic. The latter are more effective because they have less incidence of side effects and little ability to produce catalepsy. Recent AP, unlike typical AP's that are equipotential, block strongly induced hyperlocomotion. Of all this, we can conclude that it is necessary to improve the efficacy of these drugs or to produce new antipsychotics with a lower probability of inducing collateral or counterproductive effects.27,28
The principal antipsychotic that marked a treatment against schizophrenia was chlorpromazine.5 This phenothiazine antipsychotic is an antagonist of dopamine D2 receptors and similars, such as D3 and D5. It blocks the serotonergic receptors 5-5HT1 and 5-HTP2 in such a way that increases the neural dopaminergic activity and induces anxiolytic and anti-aggressive effects. Also, it has been proven to attenuate the extrapyramidal effects. However, as any drug, this AP has adverse effects, which is why many of the patients who are treated with this AP usually leave treatment because it worsens their quality of life by producing counterproductive effects like anemic degeneration, hypotension, sedation, gain of weight and difficulty in ejaculation. In addition, chlorpromazine blocks M1 and M2 muscarinic receptors. This arrest of M receptors leads to adverse side outcome by generation of anticholinergic effects such as constipation, dry lips, blurred vision and sinus tachycardia.27
Several studies carried out in patients with schizophrenia in China lead us to analyze another drug antipsychotic, risperidone. This antipsychotic has been used enormously in the treatment and therapy of schizophrenia and other psychotic illnesses. This drug is metabolized to 9-Hidroxirisperidona and, similar to chlorpromazine,29 is an antagonist of the dopaminergic receptor D2.5 The response of Risperidone has a great variability that depends on the environmental, physiological, genetic and epigenetic factors. The latter can modulate the expression of the main genes responsible for the metabolism and distribution of drugs. There are studies focused on a few candidate genes, recently 14 candidate genes have been related to the treatment of schizophrenia using risperidone. Analyzing this, risperidone has shown efficacy, but with susceptibility to epigenetic factors mainly.30
Various experiments with good results in Brazil lead to clozapine, an antipsychotic that has proven to be quite effective. Its efficacy is due to its action against serotonin receptors (5-HT2A), dopaminergic (D1, D2, D3, D4), histaminergic, adrenergic and cholinergic. In clinical practices, the use of clozapine in patients with schizophrenia has had a great impact, we can assert that approximately 50% of individuals who did not respond satisfactorily to treatment with typical antipsychotics, did with this drug. But, similar to the antipsychotics mentioned above, it has adverse effects such as hypersalivation, sedation, dizziness, headaches, drowsiness, hypotension, among others.31
Cannabis use and Schizophrenia
Cannabis is one of the illicit drugs most known worldwide. It has been object of study because of its effects on the nervous system. In fact, evidence suggests that cannabis use has a linkage with schizophrenia symptoms; however, it is still unclear whether this is because of the effect of cannabis on psychosis progress or whether genes that increase cannabis use are the same that increase psychosis risk. Furthermore, it is well established that cannabis is used much higher among people who suffers schizophrenia than in the general population.6
During the last decades, cannabis use has increased until reach the title of being the most used drug around the world according to the information given by the United Nations Office on Drugs and Crime, and as its use increases, psychotic disorders and schizophrenia appearance also increase. Knowing the healthy aspects from schizophrenia and other psychotic disorders, the approach of use of cannabis as a cause of schizophrenia has impinged on discussion over the legislation surrounding its use.6 Nonetheless, this linkage between the disease and the drug seems to be more complicated to analyze. Several studies have stated that using cannabis is a risk factor for psychosis. Ferdinand and collaborators found that the relation is bidirectional.32 Notwithstanding, other experiment failed when it tried to obtain the same finding.6,33
Results from researchers working on the study of the association between cannabis and schizophrenia show that it is due to a shared genetic etiology across common variants. They propose the idea that people who are more likely to ingest cannabis are those who are genetically predisposed to schizophrenia. This idea states that this association, at least a part of it, presents a causal relationship in the opposite direction. In addition, if this association is completely right, it would not rule out the possibility of cannabis being a risk factor for such disorder. These findings are important to understand the effects of cannabis in schizophrenia development and to conclude saying that there is a relation, independently of what kind of association is.6
DISCUSSION
We can say that, despite the great amount of studies and analysis about the complexity of genetics that presents the development of schizophrenia, is still unknown how the implicated molecular mechanisms are. It is considered that the most acceptable form to analyze the pathogenesis is with a serie of major and minor contributions from a set of locus associated with multiple chromosomes. This idea of multiple genetic contributions conforms the polygenic model, which seems to be a model that explain the etiology of the schizophrenic spectrum. On the other hand, there is also enough evidence to propose the existence of a linkage between the development of the disease and the ambiental and epigenetic factors. Other alleged responsibles of the development of the disease, is the consume of cannabis, which effect in nervous system play a big role in the etiology of the disorder. Even though it’s not discovered yet the precise relationship between cannabis and schizophrenia, it is proved that its consumption means a risk factor for the onset of schizophrenia. Another important approach that have to be taken into account in the study of schizophrenia is the behaviour of patient in the society. This allow to analyze thoroughly the epigenetic factors that are involved in the development of the disorder and which of them participates on the genetic heritability.
Finally, there are still a few lines of researching, where molecular techniques supposed to be a progress against the illness. In other words, techniques that support directly the profound study of the chromosomal linkages and obtain new successful pharmacological treatments from that analysis. Furthermore, emerging investigations about genetic mechanisms could reveal new manners of pharmacologic development, and could even reach the generation of a model for polygenic diseases treatments. This could suppose a strong social advancement if we consider that, nowadays, drugs used such as chlorpromazine, risperidone and clozapine, which are antagonists of antidopaminergic and antiserotonergic receptors, have secondary perjudicial and counterproductive effects for the patients. Principally, the chlorpromazine, which often presents a setback for the schizophrenia treatment. Antipsychotics have been of great utility, yet their counter effects lead us to continue studying the nature of schizophrenia, its causes and its repercussions.
CONCLUSION
Due to the large number of chromosomal associations that this disease presents, it is difficult to find candidate genes from which to hypothesize that its regulation prevents the development of the disease or even manages to cure it. Although it is practically a fact that certain environmental changes and epigenetics play a very important role in the birth of the disease, there are still few studies about the influence and what types of epigenetic mechanisms are responsible for schizophrenia. It should be noted that even knowing that genomics has a lot to do with the malformation of the CNS, which leads to the development of disorder, there are few investigations applying molecular techniques different from drugs for its treatment.
There are few AP that perform in instances a serious regulation on schizophrenic patients, they are still an inefficient and expensive way of treating the disease. Association between cannabis use and schizophrenia is not completely determined yet because there are different results when trying to identify the type of relation, nevertheless it is a fact that cannabis has several implications on the development of such disorder.
REFERENCES
1. Aguilar Á. Identificación de factores genéticos en la etiología de la esquizofrenia. Sistema de Información Científica Redalyc. 2011 July; 16(3).
2. Gejman P, Sanders A. La etiología de la esquizofrenia. 2012; 72(3).
3. Giménez C. Bases moleculares de la esquizofrenia. 2012 November .
4. Mediavilla J. Esquizofrenia. Londres: Royal College of Psychiatrists, Consejo Editorial de Educación Pública; 2009.
5. Tamminga CA. The promise of new drugs for schizophrenia treatment. 1997 April; 42(3).
6. Power R, Verweij K, Zuhair M, Montgomery G, Henders A, Heath A, et al. Genetic predisposition to schizophrenia associated with increased use of cannabis. Molecular Psychiatry. 2014 June; 19.
7. Salamanca D, Vergara J, Escobar FRÁ, Caminos J. Avances genéticos y moleculares en el estudio de trastornos mentales. Revista Facultad Medica. 2014; 62(2).
8. Sacchi S, Rosini E, Pollegioni L, Molla G. D-Amino Acid Oxidase Inhibitors as a Novel Class of Drugs for Schizophrenia Therapy. Current Pharmaceutical Design. 2013; 19.
9. Obiols J, Vicens-Vilanova J. Etiología y Signos de Riesgo en la Esquizofrenia. International Journal of Psychology and Psychological Therapy. 2003; 3(2).
10. Consenso Nacional y Guía de Práctica Clínica de la Asociación Psiquiátrica Mexicana y de la Asociación Psiquiátrica de América Latina para el tratamiento de las personas con esquizofrenia. Salud Mental. 2014;37(1):VII-118.
11. Obiols J, Carulla M. Bases biológicas de la esquizofrenia: Aspectos Neuroquímicos y Neuroanatómicos. 1998; 6(1).
12. Yrjö A. LA ESQUIZOFRENIA. In Sanz ME, editor. LA ESQUIZOFRENIA: Sus Orígenes y su Tratamiento Adaptado a las Necesidades del Paciente. Madrid: Editorial H. Karnak Ltd.; 2003. p. 23-37.
13. Wang Q, Ji W, He K, Li Z, Chen J, Li W, et al. Genetic analysis of common variants in the ZNF804A gene with schizophrenia and major depressive disorder. 2017 September; 27.
14. Harrison P, Owen M. Genes for schizophrenia? Recent findings and their pathophysiological implications. Lancet. 2003 February ; 361.
15. Bolaños S. Análisis epistémico del modelo biológico para explicar el origen de la esquizofrenia. Sistema de Información Científica Redylac. 2003; 19(106).
16. O'Tuathaigh C, Moran P, Zhen X, Waddington J. Translating advances in the molecular basis of schizophrenia into novel cognitive treatment strategies. 2017 October; 174(19).
17. Morris B, Pratt J. Novel treatment strategies for schizophrenia from improved understanding of genetic risk. 2014 November; 86(5).
18. Howes O, et al. Treatment-Resistant Schizophrenia: Treatment Response and Resistance in Psychosis (TRRIP) Working Group Consensus Guidelines on Diagnosis and Terminology. The American Journal of Psychiatry. 2016 December; 174(3).
19. Valiente A, Lafuente A, Bernardo M. Revisión sistemática de los Genomewide Association Studies (GWAS) en esquizofrenia. Revista de Psiquiatría y Salud Mental. 2011 September; 4(4).
20. Harrison P, Weinberger D. Schizophrenia genes, gene expression, and neuropathology: on the matter of their convergence. Nature. 2005; 10.
21. Pacheco A, Raventós H. Genética de la esquizofrenia: avances en el estudio de genes candidatos. Revista de Biología Tropical. 2004 September; 52(3).
22. Xavier RM, Vorderstrasse A, Keefe R, Dungan J. Genetic correlates of insight in schizophrenia. Elsevier. 2017 October.
23. Saiz J, de la Vega D, Sánchez P. Bases Neurobiológicas de la Esquizofrenia. Clínica y Salud. 2010; 21(3).
24. Ovendena E, McGregora N, Emsleyb R, Warnicha L. DNA methylation and antipsychotic treatment mechanisms in schizophrenia: progress and future directions. 2017 October;(17).
25. Graff A, Apiquian R, Fresán A, García M. Perspectiva neurobiológica de la esquizofrenia. Sistema de Información Científica Redalyc. 2001 December; 24(6).
26. Wooley DW, Shaw E. A biochemical and pharmacological suggestion about certain. Proceedings of the National Academy of Sciences. 1954 April; 40(4).
27. Cabildo A, González M, Hernández M, Quintanar V, Sayago A. Diseño basado en quimioinformática y estudios de acoplamiento molecular de análogos de Clorpromazina para disminuir los efectos secundarios en el tratamiento de la esquizofrenia. 2016; 3(1).
28. Tajima K, Fernández H, López-Ibor JJ, Carrasco J, Díaz-Marsá M. Tratamientos para la esquizofrenia. Revisión crítica sobre la farmacología y mecanismos de acción de los antipsicóticos. Actas Esp Psiquiatría. 2009; 37(6).
29. Ghafelehbashi H, Kakhki M, Kular L, Moghbelinejad S, Ghafelehbashi S. Decreased expression of IFNG-AS1, IFNG and IL-1B inflammatory genes in medicated Schizophrenia and Bipolar patients. 2017 October.
30. Shi Y, Li M, Song C, Xu Q, Huo R, Shen L, et al. Combined study of genetic and epigenetic biomarker risperidone treatment efficacy in Chinese Han schizophrenia patients. Nature. 2017 May; 7.
31. Barzotti F. Pharmacogenetics in schizophrenia: a review of clozapine studies. Revista Brasileira de Psiquiatría. 2013 September; 35(3).
32. Ferdinand R, Sondeijker F, van der Ende J, Selten J, Huizink A, Verhulst F. Cannabis use predicts future psychotic symptoms, and vice versa. Addiction. 2005 May; 100(5).
33. Fergusson D, Horwood L, Ridder E. Tests of causal linkages between cannabis use and psychotic symptoms. Addiction. 2005 March; 100(3).
Received: 20 noviembre 2017
Approved: 21 mayo 2018
Isaac Bravo, Freddy Figueroa, Cristhian Preciado
Escuela de Ciencias Biológicas en Ingeniería, Universidad Yachay Tech. Ecuador
