2023.08.01.28
Files > Volume 8 > Vol 8 No 1 2023
Production of environmentally friendly attractants for the trap flies Megaselia halterata and Lycoriella ingenue parasites on edible mushrooms in Iraq
1Department of plant protection – College of Agriculture – University of Tikrit-Iraq;
2Agriculture researches office – Ministry of Science and Technology-Iraq;
*Corresponding author. [email protected].
Available from: http://dx.doi.org/10.21931/RB/2023.08.01.28ABSTRACT
Among several tested mushroom-related materials, full-grown compost followed by fermented corn cobs and the compost were the best baits for attracting and catching of both insects M. halterata and L. ingenua. At the same time, there was no effect on attracting insects for the wheat straw, unfermented corn cobs, unfermented bran and water (control). The results proved that the highest attraction in the hunting of the two insects, M. halterata and L. ingenua, was in the treatment of cut fruit bodies for all studied A. bisporus strains, the highest number was 6.49 and 5.43 insects/bait; in the treatment of cut fruits of A.bisporus B62, respectively, At the level of mushroom species, the brown strain of A.bisporus showed the lowest attraction to the studied insects. Chopped fruit bodies and the spawn of some species/strains of oyster mushroom Pleurotus led to the highest interest in insects, followed by the treatment of mashed fruit bodies; the chopped fruits reached the highest attraction of insects for P. eryngii, the number of insects, M. halterata and L. ingenua, was 6.56 and 5.32 insects/bait, respectively. In the combination baits that were made from mixtures of the most efficient treatments resulting from the media and the fruit bodies of the A.bisporus and Pleurotus spp., the results showed that all treatments led to attracting the two insects at a rate of 4.55 - 8.7 insects/bait for M.halterata and 4.06 - 7.82 insects/bait for L. ingenue. The results also showed that there were significant differences in the reduction of both mushrooms A.bisporus and P. ostreatus infection rate by all types of tested baits; the lowest infection rates were in the combination bait treatment, resulting in 1.86 and 2.17%, respectively, compared to the control treatment (without bait) in which the infection rate was 87.3 and 91.25%, respectively.
Keywords: Natural baits, Traps, Mushroom flies, Biological control.
INTRODUCTION
The white button mushroom Agaricus bisporus and the oyster mushroom Pleurotus spp. are mainly edible mushrooms that can provide many essential nutrients, including protein, vitamins and minerals, and medicinal properties that have various health benefits for humans1. Mushroom yield is exposed to many pests and diseases that cause losses in the quantity and quality of the product. The mushroom flies belong to some genera, such as Lycoriella sp., Bradysia sp., Megaselia sp. and others. They are essential flies that attack most stages of mushroom production, starting from the nutrient medium and ending with the location of producing fruit bodies. These insects also infect most commercially produced edible mushrooms such as A. bisporus, Pleurotus spp., Lentinus edodes and others2,3. Mushroom flies cause damage to the mushroom through various methods, such as direct feeding of the larvae on the developing fruiting bodies and the mycelium growing in compost or substrate (nutrient medium), in addition to competition between the larvae and the grown mycelium to obtain nutrients in the mushroom medium. The indirect harmful effects of the mushroom fly are likely to transmit many pathogens such as bacteria, fungi, viruses and nematodes2,4. It is known that a significant decrease in the mushroom yield occurs at a density of only one larva per 125 g of medium5. The infection of mushrooms such as Agaricus spp. and Pleurotus spp. with the insect pests, especially Lycoriella sp. and Megaselia sp., leads to significant losses in the quantity and quality of the mushroom yield. Therefore, many studies have used the applications of control of these insects by chemical, biological and physical methods4. Several strategies have been used to control mushroom flies, including the use of chemical pesticides such as diazinon and imadocloprid6, cyromazine, diflubenzuron, diazinon, endosulfan and chlorfenvinphos7, sex hormones8, chitin synthesis inhibitors9, in addition, biological control has been used against these insects such as entomopathogenic nematodes10 and larval predators11. The biological factors were not widely applied in mushroom farms12, 3. Therefore, chemical pesticides remain the fastest effective in eradicating mushroom flies, but there are several problems associated with the use of chemicals, such as the impact on human health and the environment as well as the emergence of Pesticide-resistant strains and their effect on natural organisms that beneficial for mushroom growth. Furthermore, spraying pesticides is challenging to apply when insects attack the fruiting bodies because the larvae move after hatching into the fruits and protect themselves from the effects of pesticides8,13. Therefore, attention is focused on physical methods, including color and sticky traps14, as these traps are considered an inefficient means in the case of mushroom farms because the smell of fruiting bodies attracts insects to them more than the colored traps, in addition to the fact that these traps are not specialized to attract edible mushrooms flies. The use of plants in the form of plant extracts can benefit from some of their insect-repellent, anti-nutritive and growth-regulatory properties15. Also, extracts of some plants such as garlic, onion, neem and eucalyptus were used as alternatives to chemical pesticides in the control of Lycoriella ingenua that parasitizes Pleurotus mushroom. During four weeks, the insect mortality rate was 40-60%, depending on the type of plant16. We did not find in the literature the use of mushrooms related materials such as compost or the mycelium and fruit bodies of mushrooms themselves as baits to attract and get rid of the insect. In Iraq, many of these pests and pathogens have been studied previously17, while mushroom flies, Megaselia halterata and Lycoriella ingenua were recorded in 2021 for the first time in Iraq18.
For the importance of mushroom yield, reducing the infection of the mushroom fly, and reducing the use of chemical pesticides in controlling them, this study aimed to evaluate several mixtures of the culture media of these mushrooms and their fruit bodies for studying their attractive role in insect traps.
MATERIALS AND METHODS
Cultivation of the white button mushroom A. bisporus and the oyster mushroom Pleurotus spp.
A. bisporus was grown on compost in nylon bags 40 cm wide x 80 cm long x 20 cm deep (Fig. 1), while oyster mushroom was grown in bags of 30 cm length and 20 cm width using wheat straw medium supplemented with wheat bran (10%) and calcium carbonate (2%) (Fig. 1). The steps for cultivating mushrooms were carried out in a typical mushroom farm at the College of Agriculture, Tikrit University, including the preparation of the mushroom spawn, compost and all other processes such as spawning, incubation, and the production of fruit bodies according to the previous studies19,20,21,22.

Figure 1. Cultivation of A. bisporus and P. ostreatus in the growing room with the distribution of treatments for baits experiments.
Mushroom flies
The initial identification of the insects was carried out by Prof. Dr. Abdullah A. Hassan based on the insect's morphology and confirmed molecularly based on the nucleotide sequence of the cytochrome oxidase -subunit (1) gene in mitochondria. They were registered in the NCBI Database under accession numbers MZ021516.1 and MZ021517.1 for Megaselia halterata and Lycoriella ingenua, respectively, in our previous study18.
Study of insect damage on the mushroom fruit bodies
Fruit bodies of Agaricus spp. and Pleurotus spp. showed symptoms of infection with the studied insects harvested from the mushroom farm during 2018/2019. The pathological symptoms of fruit bodies were examined morphologically with the longitudinal and transverse sections by a magnifying lens.
Preparation of insects traps and baits
Organic baits
The traps were prepared locally using plastic bottles with a capacity of 2 liters, perforated from the top with holes of a diameter of 0.5 cm and several 20 holes. These bottles (traps) were filled with 100 g of the following media (act as baits) + 100 ml of distilled water then the bottles' nozzles were closed with cotton stoppers. The organic baits include the following treatments: Unfermented wheat straw: It was prepared by soaking the shredded wheat straw (1-2 cm) using a field grinder with tap water. The soaking time was 24 hours, then the straw was filtered from the excess water and pasteurized at 70 °C for one hour22, then used directly in filling the traps after cooling it to the next day. II; Fermented wheat straw: The same previous steps were followed, but the medium, after cooling, was packed in nylon bags, tightly closed, and incubated for 7 days at a temperature of 30°C to ferment it with natural flora, after which it was filled in traps. III; Unfermented and fermented corn cobs: The same previous steps were followed on the chopped corn cobs (1 cm), replacing wheat straw with corn cobs. IV; Agaricus spp. compost: Pasteurized compost prepared according to Hassan and Mahmoud21 after cooling it to 25° C, it packaged in insect traps. V; Full growth compost: The compost medium was inoculated with the A.bisporus spawn of commercial strain A15. The bags were incubated at 25°C for two weeks after the mycelium spread into the compost, it packaged in insect traps. VI; Wheat bran: It was prepared by the steps of preparing the unfermented wheat straw. VII; Wheat bran + yeast and sugar: 50 ml of yeast and sugar solution (1% commercial Saccharomyces cerevisiae yeast + 2 g table sugar) were added to 1 kg of pasteurized wheat bran after mixing well, incubated at 25 °C for 7 days and then packed in traps. VIII; Wheat bran + local date vinegar: It was prepared by the same previous steps except adding 100 ml of local date vinegar. IX; Dap fertilizer (Jordanian Fabco Company) The bait consists of 100 g of dab fertilizer and 5 g of S. cerevisiae yeast with a liter of water. Use this bait as a positive control for attracting some insects23. X; Control treatment: Use only water and XI; The yellow sticky trap for comparison
.
Spawn and fruit bodies baits of Agaricus spp. and Pleurotus spp.
Spawn and fruit bodies of five strains of A. bisporus cultured in the mushroom production farm, College of Agriculture / Tikrit University, were used. The strains included brown strain, A15, B62, S11, and the local isolate A. campestris. Spawn and fruit bodies of six strains of oyster mushrooms cultured in the same mushroom production farm, including P. ostreatus, P.florida, P.sajor-caju, P.eryngii, P. pulmonarius and P. ostreatus A19 (local isolate) were used. Traps were filled with 100 g of the following media (act as baits for both mushrooms) + 100 ml of distilled water, then the bottles' nozzles were closed with cotton stoppers; I; The pure spawn has grown on wheat grains prepared according to Hassan24, II; The dried and ground fruit bodies in a powder form. III; Fresh cut fruit bodies 1-2 cm2. IV; Fresh fruit bodies mashed in a blender in the form of an emulsion and V; Water: control.
Combination of baits
It included various combinations of previous treatments. The best treatments were used in proportions of 50 g for each bait material + 100 ml of distilled water for the binary mixtures.
Insect breeding in the laboratory
100 g of the infected Pleurotus ostreatus fruit bodies with both insects, which show necrosis, tunnels, and larvae, were placed in plastic cups, their mouths closed with two layers of gauze, and incubated at 22 °C, 70% humidity and 12:12 light: dark cycle, after 10-14 days the pupation of the larvae is observed (morphologically similar to both insects) , and after 3-6 days the adult insects emerge. Adults were isolated from the pupae separately, placed in plastic cups containing intact (uninfected) pieces of the P. ostreatus fruit bodies, and incubated under the above conditions to obtain the insects of both species separately. For the sustainability of the insect culture, fresh fruit bodies are constantly placed in the culture medium. This is the easiest method of breeding the insect recorded for the first time in Iraq, and we did not find it in the international literature.
Execution of the experiment
After cultivation of both Agaricus spp. and Pleurotus spp. Mushrooms and the baits were immediately distributed separately in the growing room (Figure 1). 100 adult insect were released in each experiment. After the end of the mushrooms production cycle, the number of insects was recorded in each trap, then the Infestation rate of mushrooms fruit bodies was calculated as the following equation:
Infestation rate = number of fruits infected with the insect / total number of fruits x 100
The fruit was considered infected if there were larvae, necrosis or tunnels caused by the insects in the mushroom fruit bodies.
Statistical Analysis
Statistical analysis of factorial laboratory experiments was carried out using the Mini Tap 18 program, and the means were compared according to Duncan's multiple range test25 at a probability level of 0.05.
RESULTS
Effect of the type of baits on attracting M. halterata and L. ingenua in traps
Effect of media
The results of table (1) showed the significant superiority of full-grown compost in attracting and catching both insects M. halterata and L. ingenua, the number was 8.04 and 7.45 insects/bait, respectively, followed by the fermented corn cobs and the compost, where the number reached (7.7, 6.83) and (6.11 and 5.3) insect/bait for these two insects, respectively, compared with the dab fertilizer (2.78 and 2) and the yellow sticky trap (1.03 and 0.66) insect/bait, while there was no effect on attracting insects for the wheat straw, unfermented corn cobs, unfermented bran and water (control).

Table 1. Number of M. halterata and L. ingenua caught with different baits
Effect of Agaricus spp. baits on attracting insects
The results of table (2) proved that the highest attraction in the hunting of the two insects, M. halterata and L. ingenua, was in the treatment of cut fruit bodies for all studied A. bisporus strains, the highest number was 6.49 and 5.43 insects/bait, in the treatment of cut fruits of A.bisporus B62, respectively, followed by the attraction of insects in the treatment of the mushroom spawn. The results showed that the lowest number was in the treatment of dried fruit bodies (Table 2), while no insect attraction was recorded in the control treatment (water only). At the level of mushroom species, the brown strain of A.bisporus showed the lowest interest to the studied insects.

Table 2. Effect of the fruit bodies of Agaricus spp. baits on attracting the two insects M. halterata and L. ingenua
Effect of oyster mushroom baits on attracting insects
Chopped fruit bodies and the inoculum of some species/strains of oyster mushroom Pleurotus led to the highest attraction to insects, followed by the treatment of mashed fruit bodies, while the treatment of dried fruit bodies was less attractive to insects, while the control treatment (water only) recorded no attraction to insects, the highest attraction of insects was reached by the chopped fruits for P. eryngii, the number of insects, M. halterata and L. ingenua, was 6.56 and 5.32 insects/bait, respectively (Table 3).

Table 3. Effect of the fruit bodies of Pleurotus spp. Baits on attracting the two insects M. halterata and L. ingenue.
Effect of the combination baits
The results of the combination baits were made from mixtures of the most efficient treatments resulting from the media and the fruit bodies of the A.bisporus and Pleurotus spp. Table (4) showed that all treatments attracted the two insects at a rate of 4.55 - 8.7 insects/bait for M.halterata and 4.06 - 7.82 insects/bait for L. ingénue. The combinations of (A.bisporus B62 + P.eryngii), (fermented corn cobs + full-grown compost) and (fermented corn cobs + P.eryngii) achieved the rates of attraction for these two insects (Table 4).

Table 4. Effect of bait combinations on attracting M. halterata and L. ingenua
Infection of A.bisporus and P. ostreatus by M. halterata and L. ingenue using studied biats
Figure (2) shows the infection rate of the fruit bodies of A.bisporus and P. ostreatus in the presence of media baits and the fruit bodies of the A. bisporus and Pleurotus spp. The results showed that there were significant differences in the reduction of both mushroom A.bisporus and P. ostreatus infection rate by all types of tested baits; the lowest infection rates were in the combination bait treatment, resulting in 1.86 and 2.17%, respectively, compared to the control treatment (without appeal) in which the infection rate was 87.3 and 91.25%, respectively.
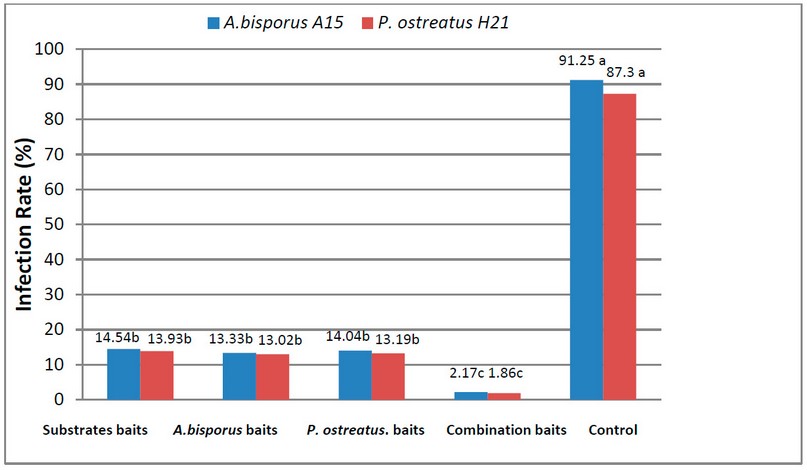
Figure 2. Infection rates of A. bisporus and P. ostreatus by the insects M. halterata and L. ingenue in the presence of different baits.
DISCUSSION
M. halterata and L. ingenua, cause severe damage to the yield of the edible mushrooms A. bisporus and P. ostreatus, tunnel digging, necrosis, perforation of fruit bodies with tissue laceration and the presence of larvae wastes are the most critical symptoms of these insects, which causes a loss of the entire mushroom yield, the infection of the mushroom fruit bodies may be due to the nutritional specialization of these two insects towards feeding on mushroom fruit bodies, this agreed with some relevant studies26,27. To avoid chemical pesticides in mushroom production farms, which are among the dangerous factors due to the rapid growth of edible mushrooms and their high efficiency in absorbing materials from the media, the research suggested physical, environmental and biological applications to reduce the impact of these two insects. The synthetic pheromones did not have completely positive effects in attracting these insects28,29. In addition, the biological resistance factors are still under ongoing studies. Cloonan et al. 3 mentioned the lack of a wide commercial application of biological agents to control these insects. By tracking the infection of the A. bisporus and Pleurotus spp. mushrooms by these two insects in several production cycles, we found that they are highly attracted to mushroom growth medium, spawn and fruit bodies, as they are the preferred nutrient sources for these insects30,31.
The baits used in this study were characterized by their high efficiency in attracting the studied insects. Among the organic materials, fermented corn cobs and compost were distinguished by their highest attraction, which may be attributed to the presence of volatile organic compounds (VOCs) in the compost that attracts insects32. In terms of attracting insects to some mushroom species, the bait made from the cutting fruit bodies was more attractive than the dried and mashed fruits; this may be due to the loss of compounds responsible for the distinct aroma and flavor of the mushrooms (Agaricus, Pleurotus) by drying and mashing process such as benzyl alcohol, benzaldehyde, benzonitrile, methyl benzoate, and phenylacetic acid-like compound, as well as the distinctive smell of the mushroom itself 33,34. The same effect was recorded in the bait of the mushroom spawn; this is explained by the release of the fungal mycelium that surrounds the spawn grains, the same volatile substances for the smell and flavor compounds in A. bisporus and Pleurotus spp. mushrooms, conducted by Yin et al. 35 studies, in which he isolated insect-promoting compounds from 6 species of the Pleurotus spp. such as free amino acids and nucleotides, as well as many volatile compounds, which included 17 aldehyde compounds, 10 ketone compounds, 14 alcohol compounds, 5 hydrocarbons, 10 cyclic aromatic compounds, as well as some organic acids. The same study stated that the most important compounds that distinguish the smell of edible mushrooms are 1‐Octen‐3‐one, 1‐octen‐3‐ol, and 2‐octenal.
A study by Nyegue et al36 on Pleurotus spp. confirms the isolation of octen-3-ol, often-3-one, octan-3-one, 3-octanol, n-octanal, (E)-2-octenal and n -Octanol, Benzaldehyde (almond odor), benzyl alcohol (sweet-spicy odor), phenylethanol (rose odor), derivatives of monoterpenes such as linalool and linalool oxide, and many aliphatic compounds among the volatile compounds, which explains the attraction of these insects to the mycelium of mushrooms and their fruit bodies.Some mushroom species studied, especially A.bisporus (brown strain) and P.eryngii, were the most attractive, perhaps due to the difference in the genetic composition of these two mushrooms, which is reflected in their physiological activity in producing more concentrated insect-attracting compounds or perhaps other substances that have not been discovered until now. The organic materials, mushroom fruit bodies and the combinations of baits proved their efficiency in reducing the number of insects through a decrease in the infection rate of A. bisporus and Pleurotus spp., which reached a minimum percentage of 1.86-2.17% in the treatments of combinations of baits as a result of the success of attracting insects.
CONCLUSIONS
Through the parasitism of the insects M. halterata and L. ingenua on the mushrooms A. bisporus and Pleurotus spp., the most relevant materials for mushrooms were developed as baits to catch these two insects. Each of full-grown compost , fermented corn cobs , compost ,cut fruit bodies and spawn of A. bisporus and Pleurotus spp. strains in addition to the combinations included (A.bisporus B62 + P.eryngii), (fermented corn cobs + full-grown compost) and (fermented corn cobs + P.eryngii) , were the best baits for catching these insects and reducing their harmful effect on mushroom production farms. Using baits to attract and catch mushrooms is one of the safe ways to control these insects instead of insecticides and to avoid losses in the quality and yield of mushrooms.
Acknowledgements
We would like to acknowledge Mushroom farm staff in Tikrit University-Iraq for provided necessary facilities for the study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Availability of data and materials
The data set (table and graphs) supporting this article's conclusion is available.
Ethics approval and consent to participate
All the authors have read and agreed on the ethics for publishing the manuscript.
Consent for publication
The authors approved the consent for publishing the manuscript.
Competing interests
The authors declare that they have no competing interests
REFERENCES
1. Cheung P.C.K. (2010) The nutritional and health benefits of mushrooms. Nutr Bull 35:292–299.
2. O'Connor, L. and Keil, C. B. 2005. Mushroom Host Influence on Lycoriella mali (Diptera: Sciaridae) Life Cycle Journal of Economic Entomology, 98(2):342-349.
3. Cloonan, K. R. Stefanos S. Andreadis & Thomas C. Baker. 2016. Attraction of female fungus gnats, Lycoriella ingenua, to mushroom-growing substrates and the green mold Trichoderma aggressivum. Entomologia Experimentalis et Applicata 159: 298–304.
4. Singh. A.U. and Sharma, K. 2016. Pests of Mushroom. Adv. Crop Sci Tech, 4:2
5. White, P.F. (1986) The effect of sciarid larvae (Lycoriella auripila) on cropping of the cultivated mushroom (Agaricus bisporus). Annals of Applied Biology 109: 11–17.
6. Shirvani-Farsani, N., Zamani, A.A., Abbasi, S. and Katayoon Kheradmand. 2013.Toxicity of three insecticides and tobacco extract against the fungus gnat, Lycoriella auripila and the economic injury level of the gnat on button mushroom. J Pest Sci 86, 591–597.
7. Dmoch, J. 1988. Effect of some insecticides on mushroom mycelial growth. Acta Hort. 219: 15-20.
8. Andreadis, S. S., Cloonan, K. R., Myrick, A.J., Chen, H., Baker, T. C. (2015). Isolation of a Female-Emitted Sex Pheromone Component of the Fungus Gnat, Lycoriella ingenua, Attractive to Males. J Chem Ecol. 41:1127–1136
9. Cantelo, W.W. (1983). Control of a mushroom infesting fly (Diptera: Sciaridae) with insecticides applied to the casing layer. Journal of Economic Entomology, 76(6), 1433–1436.
10. Jess S., H. and Kilpatrick M, Mushroom applications, in Nematodes as Biocontrol Agents, ed. by Grewal PS, Ehlers R-U and Shapiro DI. CAB International, Wallingford, Oxon, UK, pp. 191–213 (2005).
11. Lind R. (1993) Control of mushroom flies with the predatory mite, Hypoaspis miles. Contract Report Horticultural Development Council Project M9, Horticultural Development Council, West Malling, Kent, UK, 28 pp.
12. Jessa, S. and Schweizerb, H.. 2009. Biological control of Lycoriella ingénue (Diptera: Sciaridae) in commercial mushroom (Agaricus bisporus) cultivation: a comparison between Hypoaspismiles and Steinernema feltiae. Pest Manag Sci .65: 1195–1200.
13. Bartlett, G.R. and Keil, C.B.O. (1997) Identification and characterization of a permethrin resistance mechanism in populations of the fungus gnat Lycoriella mali (Fitch) (Diptera: Sciaridae). Pesticide Biochemistry and Physiology 58: 173–181.
14. Bellettini, M. B., Bellettini, S., Fiorda, F. A., Pedro, A. C., Bach F., Miriam F., Moron, F. and Hoffmann-Ribani, R. 2018. Diseases and pests noxious to Pleurotus spp. mushroom crops. Rev Argent Microbiol.;50(2):216-226.
15. Smith J, Challen M, White P, Edmondson R, Chandler D (2006) Differential effect of Agaricus host species on the population development of Megaselia halterata (Diptera: Phoridae). Bull Entomol Res 96(6):565
16. Badshah, K., Ullah, F., Ahmad, B. Ahmad, S., Alam, S., Ullah, M., Momana Jamil, M. and Sardar, S. (2021). Management of Lycoriella ingenua (Diptera: Sciaridae) on oyster mushroom (Pleurotus ostreatus) through different botanicals. Int J Trop Insect Sci 41, 1435–1440.
17. Hassan, A. Abdulkareem (2013) . First Report of Diseases and Pests of an Edible Mushroom Agaricus bisporus in Iraq. 1.The Fungal Infections. Journal of Tikrit University For Agriculture Sciences.13(4):60-71.
18. Hassan , A. A., and Al-Qaissi, A. R. (2021). First record of the two insects Megaselia halterata and Lycoriella ingenua parasite on some edible mushrooms in Iraq. Submitted to Arab Journal of Plant Protection.
19. Hassan , A. A.,Natheer , A.M. & Mahmoud, A. R.(2000). effect of application of some organic sources on the oyster mush room Pleurotus ostreatus yield.Iraqi J. Agric.5(4):185-190.
20. Hassan, A. A. Natheer, A. M. and Mahmoud, A. R. (2002). Improvement of agronomic characters and productivity of Agaricus bisporus Lange(Imbach) , using some organic sources . Iraqi J. Agric.7(3): 104-112.
21. Hassan , A. A., and Mahmoud, A. R. (2003). Outdoor cultivation of two white edible mushrooms Agaricus bisporus and Agaricus bitorquis . Iraqi J. Agric. 8(2):59-66.
22. Hassan, A. A. and Mahmoud, A. R. (2008) Outdoor Cultivation Of Oyster Mushroom Pleurotus sp. Using Shaded Plastic Houses. Iraqi J. Agric. 13(1): 48-55.
23. Al-Jubouri, Raghad Khalaf Ibrahim and Al-Jassani, Radi Fadel (2010). Evaluation of the efficiency of some food attractant traps and colored traps in attracting adults of the Mediterranean fruit fly. Anbar Journal of Agricultural Sciences. 8(1): 263-270.
24. Hassan , A. A. (2009). Selection of new isolates from single spore and multispore cultures from two strains of Agaricus bisporus. Tikrit J. of Pure Science. 14(3):1-10.
25. Al-Rawi, K. Mahmoud and Khalaf Allah, A. Muhammad. 1980 . Design and analysis of agricultural experiments. Dar Al-Kutub for Printing and Publishing, University of Mosul.
26. Erler, F., Polat, E., Demir, H., Catal, M. and Tuna G (2011) Control of mushroom Sciarid fly Lycoriella ingenua populations with insect growth regulators applied by soil drench. J Econ Entomol 104:839–844.
27. Park, I. K., Choi, K. S., Kim, D.H., Choi, I.H. Kim, L.S. Bak, W.C. Choi ,J.W. and Shin, S.C. (2006) Fumigant activity of plant essential oils and components from horseradish (Armoracia rusticana), anise (Pimpinella anisum) and garlic (Allium sativum) oils against Lycoriella ingenue (Diptera: Sciaridae). Pest Manag Sci 62:723–728.
28. Gotoh, T., Nakamuta, K., Tokoro, M. and Nakashima, T. (1990) Copulatory behavior and sex pheromones in sciarid fly, Lycoriella mali (Fitch) (Sciaridae: Diptera). Jpn J Appl Entomol Zool 43:181–184
29. Stefanos, S. A. , Kevin R. C., Andrew J. M. Haibin, C. and Thomas C. B.2015. Isolation of a Female-Emitted Sex Pheromone Component of the Fungus Gnat, Lycoriella ingenua, Attractive to Males. J Chem Ecol (2015) 41:1127–1136
30. Lewandowski, M., Sznyk, A. and Bednarek, A. (2004) Biology and morphometry of Lycoriella ingenua (Diptera: Sciaridae). Biol Lett 41:41–50
31. Shamshad, A. (2010) The development of integrated pest management for the control of mushroom sciarid flies, Lycoriella ingenua (Dufour) and Bradysia ocellaris (Comstock), in cultivated mushrooms. Pest Manag Sci 66:1063–107
32. Dhamodharana, K., Varmaa, V. S. , Arivalagan, C. V., Rajendrand, P. K. 2019. Emission of volatile organic compounds from composting: A review on assessment, treatment and perspectives. Science of The Total Environment. 695: 1-33.
33. Chen, C. and Wu, C. 2006. Volatile Components of Mushroom (Agaricus subrufecens) .Journal of Food Science 49(4):1208 – 1209
34. Abend A. M, Chung L , Bibart R T, Brooks M, McCollum DG 2004.Concerning the stability of benzyl alcohol: formation of benzaldehyde dibenzyl acetal under aerobic conditions. J Pharm Biomed Anal. 10;34(5):957-62
35. Yin, C., Fan, X. , Fan, Z ., Shi, D., Yaoa, F. and Gao, H. 2018. Comparison of non‐volatile and volatile flavor compounds in six Pleurotus mushrooms. Journal of the Science of Food and Agriculture 99(4) : 1691–1699.
36. Nyegue, M., Zollo, A., Bessière, J. M. and Sylvie Rapior, S. 2003 . Volatile components of fresh Pleurotus ostreatus and Termitomyces shimperi from Cameroon. Journal of essential oil-bearing plants JEOP 6:153-157.
Received: October 23, 2022 / Accepted: January 15, 2023 / Published:15 February 2023
Citation: Abdulkareem Hassan A;, Mahmoud Al-Qaissi A R. Production of environmentally friendly attractants for the trap flies Megaselia halterata and Lycoriella ingenue parasites on edible mushrooms in Iraq. Revis Bionatura 2023;8 (1)28. http://dx.doi.org/10.21931/RB/2023.08.01.28
