2022.07.03.51
Files > Volume 7 > Vol 7 No 3 2022
Investigation of the Omentin-1 V109D gene polymorphism as a risk factor for the incidence of Polycystic Ovary Syndrome

1,2 Collage of Science, Al-Qasim Green University, Iraq. Email: [email protected]. , Email: [email protected].
3.4 Collage of Veterinary Medicine, Al-Qasim Green University, Iraq. Email: [email protected]., Email: [email protected].
5 Collage of Medicine, University of Al-Ameed, Karbala, Iraq. Email: [email protected].
* Correspondence Author: [email protected]
Available from: http://dx.doi.org/10.21931/RB/2022.07.03.51
ABSTRACT
This study was conducted to assess the omentin-1 (OME-1) V109D gene SNP in women with PCOS and healthy control and to investigate the possible relationship between this adipokine and metabolic profile. This study includes 50 women in the range of age (19-43 Y) who have been diagnosed with PCOC as a patient group and 40 healthy women in age (17-45 Y) as a control group. Five ml of peripheral blood were drawn from both groups to perform the biochemical and genetic study. Metabolic profiles were assessed by spectrophotometric methods, and PCR-RFLP investigated V109D of OME-1 SNP. The results suggest a statistical decrease in FBG, BMI, TG, TC, and LDL(p-value<0.05) in the control group compared to the PCOS group and a statistical increase in HDL levels in control compared to the PCOS group. Genotyping analysis shows statistically significant differences between DD and VV genotypes in PCOS and control groups (OR=4.66 (CI 95%,1.6-8.3), (OR=2.4 (CI95%,0.7-6.9) respectively. These results suggest statistically significant differences in three genotypes (DD, DV, and VV) of the PCOS group depending on age and BMI (p-value <0.05). DD and VV genotypes of OME-1 V109D SNP have more risk factors for PCOS incidence and changes in metabolic profile in Iraqi women.
Keywords: Polycystic ovary syndrome, Omentin-1, risk factor, polymorphism genetic
INTRODUCTION
It has been found that the most frequent endocrine disorder in women of reproductive age and, in some patients, presents with abdominal adiposity and dyslipidemia is called polycystic ovary syndrome (PCOS) 1. The set of symptoms is here due to elevated androgens in females and is the main ovarian dysfunction in PCOS 2. One of the well-known signs and symptoms of PCOS include irregular or no menstrual periods, heavy periods, excess body and facial hair, acne, pelvic pain, difficulty getting pregnant, and patches of thick, darker, velvety skin 3. Associated conditions include T2DM, obesity, obstructive sleep apnea, heart disease, mood disorders, and endometrial cancer4. PCOS is the most common endocrine disorder among women between 18 and 44, affecting approximately 2% to 20% of this age group, 5-6. Poor dietary choices and physical inactivity can exacerbate environmental factors implicated in PCOS, such as obesity; infectious agents and toxins may also play a role 7. The reproductive and metabolic features of PCOS are sometimes reversible with lifestyle modifications such as weight loss and exercise 8. Omentin-1 (OME-1) has been identified as a significant visceral (omental) fat secretory adipokine. This adipokine may act as an endocrine factor affecting muscles, liver, and omental adipose depot; it enhances insulin sensitivity and glucose metabolism 9. It has been suggested that serum omentin-1 is elevated in patients with fatty liver diseases and represents an independent predictor for hepatocyte ballooning in these patients 10. Omentin-1, also known as intelectin-1, is a recently identified novel adipocytokine of 313 amino acids, expressed in visceral (omental and epicardial) fat and mesothelial cells, vascular cells, airway goblet cells, small intestine, colon, ovary, and plasma 11. This study aimed to determine OME-1 rs2274907 gene polymorphism in women with PCOS and healthy control and investigate the possible relationship between this adipokine and metabolic profile.
MATERIALS AND METHODS
Study group
This study includes 50 women in the range of age (19-43 Y) who have been diagnosed with PCOC as a patient group and 40 healthy women in age (17-45 Y) as a control group. Five ml of peripheral blood were drawn from both groups to perform of biochemical and genetic studies.
Measurement of metabolic profile
FBG, TG, TC, LDL, and HDL were performed by spectrophotometric methods depending on protocols provided by the manual kits.
Genetic analysis of OME-1 V109D
The genomic DNA Kit (Bioneer, Korea) provides AccuPrep®, an optimal protocol for extracting and purifying the genomic DNA from whole blood. Proteinase K and the specific buffers were used to lysate cells and destroy proteins. Amplification conditions of amplicon (471bp) up to 30 cycles of OME-1 gene were performed by PCR-RFLP depending on the method described by Khoshi et al. 12.
Statistical Analysis
Statistical analysis was performed using SPSS 22 software. Observed and expected genotype frequencies using the Hardy-Weinberg equation by odds ratio (OR) and 95% confidence intervals (CI 95%). The association of OME-1 rs2274907 and metabolic profile was estimated by t-test.
RESULTS
Genetic analysis of OME-1 V109D:
The following pairs of primes (forward and reverse) were used in PCR analysis (Table1):
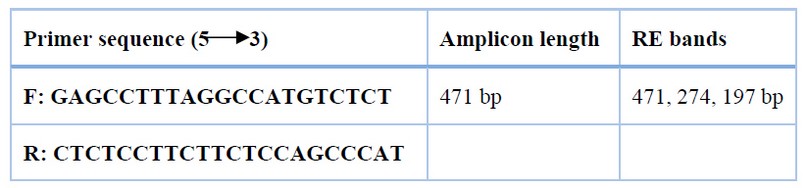
Table 1. Primers of OME-1 V109D method of PCR-RFLP that used in genotyping analysis
The PCR product (471 bp) is shown in figure (1). PCR-RFLP investigates the V109D of the OME-1 gene in patients with PCOS and control groups involved in this study by using XmiI as a restriction enzyme (RE). The PCR was performed by an Exicycler 96R (Bioneer, Korea), in which the DNA templates were denatured at 95 °C for 4 min. The amplification step consisted of 30 cycles of 30 sec at 95 °C, 45 sec at 58.5 °C, 72 °C for 1 min s, with a final extension of 5 min at 72 °C. Gel electrophoresis was running the final products on a 2%, non-digested by RE represented 471 bp of D allele (DD genotype), (274 and 197 bp) were identified as homozygotes for V allele (genotype VV). Moreover, both digested and undigested fragments (471, 274, and 197 bp) were detected as heterozygotes (genotype DV), as shown in figure (2).
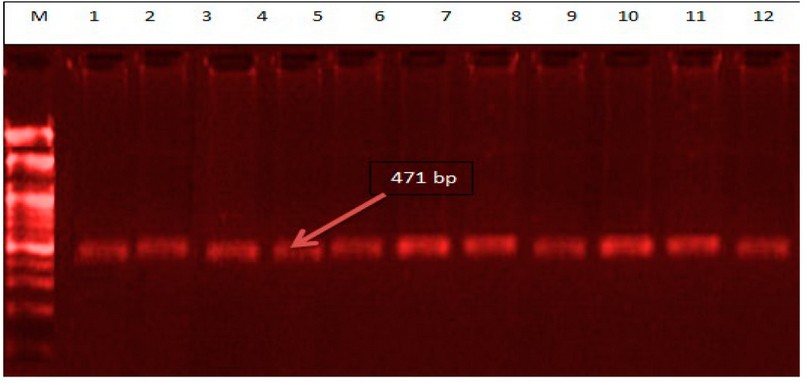
Figure 1. PCR product (471 bp) of OME-1 V109D gene. Lane M: DNA marker, lanes 1-12 represented samples of PCOS and control
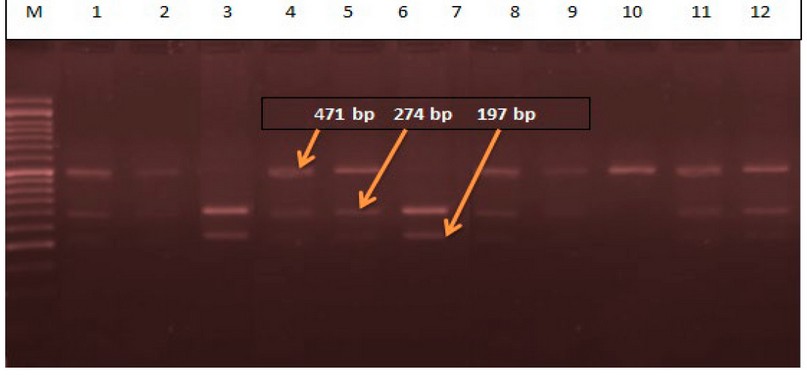
Figure 2. XmiI restriction enzyme of the PCR product of OME-1 V109D gel electrophoresis. Lane M: DNA marker, lanes 1,4,5,8,9, 11 and 12) represented DV genotype, lanes 2 and 10 represented DD genotype, and lanes 3 and 7 represented VV genotype
Table 2 shows the metabolic profile in both PCOS and control groups. The results suggest a statistical decrease in FBG, BMI, TG, TC, and LDL(p-value<0.05) in the control group compared to the PCOS group and a statistical increase in HDL levels in control compared to the PCOS group.
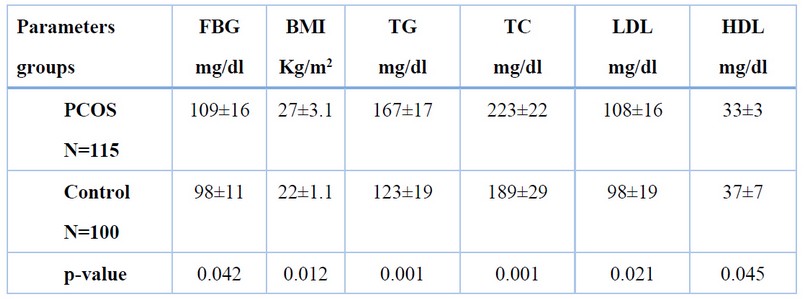
Table 2. Metabolic profile in study groups
Table 3. Shows the distribution of genotyping and allele frequencies of OME-1 V109D SNP in PCOS and control groups. Genotyping analysis shows statistically significant differences between DD and VV genotypes in PCOS and control groups (OR=4.66(CI 95%,1.6-8.3), (OR=2.4(CI95%,0.7-6.9) respectively. The results also show the significant differences between D and V alleles in PCOS and control groups (OR=3.2(CI 95%,0.8-6.2).
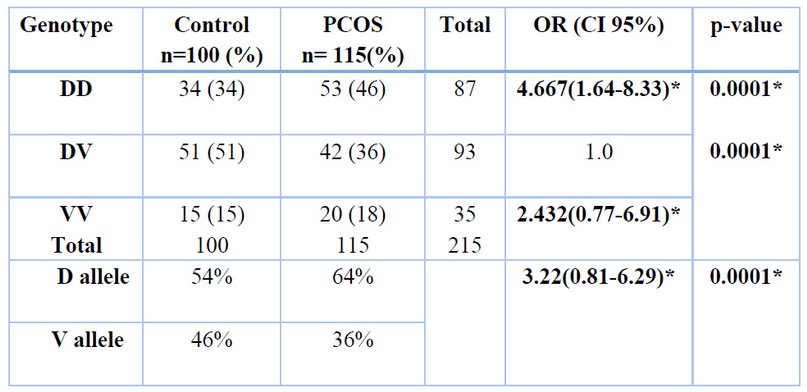
Data is represented as a number (%) *p< 0.05 is considered statistically significant. OR: odds ratio, CI: confidence interval
Table 3. Distribution of genotyping and alleles frequencies of OME-1 V109D SNP in PCOS and control groups
Table 4 shows the effects of age and BMI on OME-1 V109D SNP in the PCOS group. These results suggest statistically significant differences in three genotypes (DD, DV, and VV) of the PCOS group depending on age and BMI (p-value <0.05):
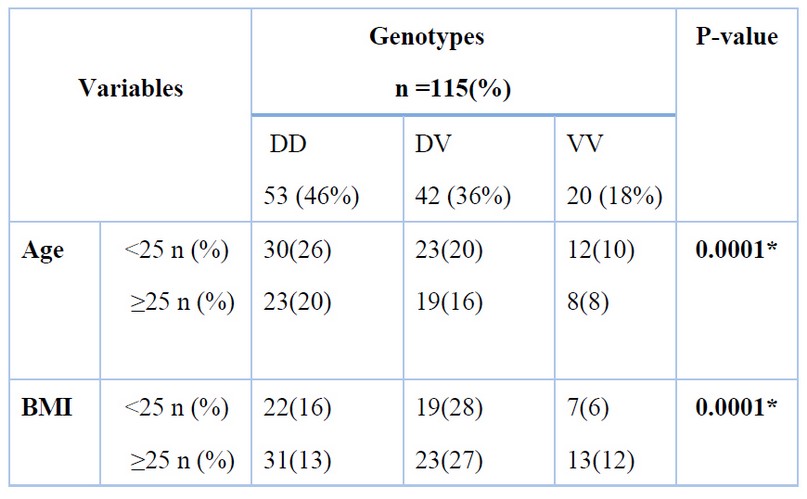
Table 4. Effects of age and BMI on OME-1 V109D SNP in PCOS group
Table 5, listed the distribution of metabolic profile of the PCOS group depending on different genotypes DD, DV, and VV. The results showed statistically significant differences in FBG and BMI in different genotypes of the PCOS group (p-value <0.05) but no statistical difference in TG, TC, LDL, and HDL levels (p-value> 0.05).
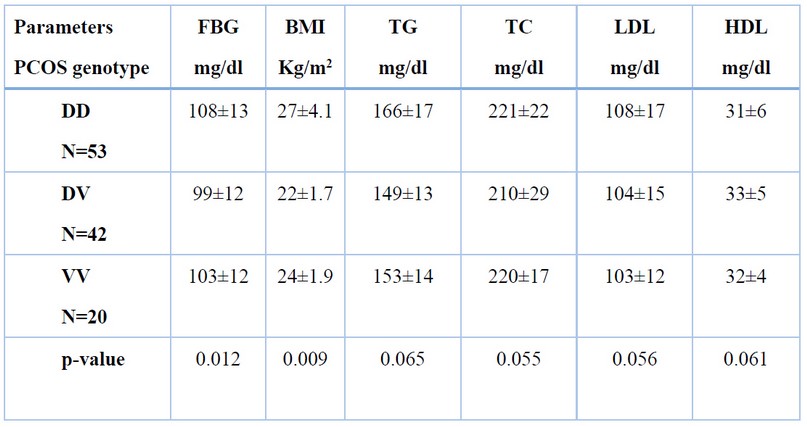
Table 5. Metabolic profile in DD, DV, and VV genotypes in PCOS group
DISCUSSION
One of the well-known, most frequent endocrine disorders in women of reproductive age and, in some patients, presents with abdominal adiposity and dyslipidemia is called polycystic ovary syndrome (PCOS). Although genetic, environmental, and behavioral factors are related to PCOS development, its etiology remains to be elucidated 13. OME is a newly identified adipokine that is highly and selectively expressed in visceral adipose tissue relative to subcutaneous adipose tissue, and there are two highly homologous isoforms of OME, OME-1 and OME-2; however, omentin-1 is the major circulating form in human plasma. OME-1 has been identified in other tissues at lower expression levels and is also named OME, intelectin, intestinal lactoferrin receptor and endothelial lectin 14. This study aimed to assess OME-1 V109D gene SNP in women with PCOS and healthy control and investigate the possible relationship between this genetic variation of adipokine and metabolic profile. The results suggest a statistical decrease in FBG, BMI, TG, TC, and LDL(p-value<0.05) in the control group compared to the PCOS group and a statistical increase in HDL levels in control compared to the PCOS group. Many studies have reported that in PCOS patients, there is a positive correlation between other adipokines called chemerin and their correlation with BMI, TG, HOMA-IR, LDL, and an inverse correlation between chemerin and HDL-C 15-18. Genotyping analysis shows statistically significant differences between DD and VV genotypes in PCOS and control groups (OR=4.66(CI 95%,1.6-8.3), (OR=2.4(CI95%,0.7-6.9) respectively. Kohan et al. (2016) suggested that OME-1 rs2274907 polymorphisms might be a candidate genetic factor for nonalcoholic fatty liver disease 19. The results also show the significant differences between D and V alleles in PCOS and control groups (OR=3.2(CI 95%,0.8-6.2). These results suggest statistically significant differences in three genotypes (DD, DV, and VV) of the PCOS group depending on age and BMI (p-value <0.05). In line with these results, our data indicated that DD homozygous genotype was more frequent, 53% in PCOS compared to healthy women 34%. Bahadori et al. (2015) concluded that Iranian women with VV genotype had a greater risk of obesity complications than those with DD genotypes 20. Many studies demonstrated that A326T rs2274907 SNP (miss-sense in the exon-4 of the OME-1 gene, which substitutes valine instead of aspartate at position 109 (Val109Asp or V109D), could be accompanied by T2DM and obesity 21,22.
CONCLUSION
This study confirms the presence of an interesting correlation between the polymorphism of the OME-1 V109D SNP and increasing risk factors for incidence of PCOS and changes in metabolic profile in Iraqi women.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no conflicts of interest.
Funding
This study did not receive any funding.
Author contributions
Conceptualization, HHK and MEA; methodology, RAA and HHK.; software, HHK; validation, AMM, HKK and RAA.; formal analysis, HHK; investigation, HHK; resources, HHK; data curation, AMM; writing—original draft preparation, HHK; writing—review and editing, HHK; visualization, MEA; supervision, MEA; project administration, MEA; funding acquisition, MEA. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
We thanks to all women whom participated in this work.
REFERENCES
1- De Leo V, Musacchio MC, Cappelli V, Massaro MG, Morgante G, Petraglia F. Genetic, hormonal and metabolic aspects of PCOS: an update. Reproductive Biology and Endocrinology (Review). 2016; 14 (1): 38.
2- Diamanti-Kandarakis E, Kandarakis H. The role of genes and environment in the etiology of PCOS. Endocrine. 2006; 30 (1): 19–26.
3- Polycystic Ovary Syndrome (PCOS): Condition Information". National Institute of Child Health and Human Development. 31 January, 2017. Retrieved 19 November 2018.
4- Mortada R, Williams T. Metabolic Syndrome: Polycystic Ovary Syndrome. FP Essentials (Review). 2015; 435: 30–42.
5- Giallauria F, Palomba S, Vigorito C, Tafuri MG, Colao A, Lombardi G, Orio F. Androgens in polycystic ovary syndrome: the role of exercise and a diet. Seminars in Reproductive Medicine (Review). 2009;27 (4): 306–15.
6- Diamanti-Kandarakis E, Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocrine reviews. 2012 Dec 1;33(6):981-1030.
7- Shannon M, Wang Y. Polycystic ovary syndrome: A common but often unrecognized condition. J Midwifery Womens Health. 2012;57:221–230.
8- Tefagh G, Payab M, Qorbani M, Sharifi F, Sharifi Y, Ebrahimnegad Shirvani MS, Pourghazi F, Atlasi R, Shadman Z, Rezaei N, Mohammadi-Vajari E. Effect of vitamin E supplementation on cardiometabolic risk factors, inflammatory and oxidative markers and hormonal functions in PCOS (polycystic ovary syndrome): a systematic review and meta‐analysis. Scientific reports. 2022 6 April;12(1):1-6.
9- Yang RZ, Lee MJ, Hu H, Pray J, Wu HB, Hansen BC, Shuldiner AR, Fried SK, McLenithan JC, Gong DW. Identification of omentin as a novel depot-specific adipokine in human adipose tissue: possible role in modulating insulin action. Am J Physiol Endocrinol Metab 2006;290:E1253-1261.
10- Tan BK, Chen J, Digby JE, Keay SD, Kennedy CR, Randeva HS: Increased visfatin messenger ribonucleic acid and protein levels in adipose tissue and adipocytes in women with polycystic ovary syndrome: parallel increase in plasma visfatin. J Clin Endocrinol Metab91 :5022 –5028,2006.
11- Guerre-Millo M. Adipose tissue and adipokines: for better or worse. Diabetes Metab 2004;30:13-19.
12- Khoshi A, Bajestani MK, Shakeri H, Goodarzi G, Azizi F. Association of Omentin rs2274907 and FTO rs9939609 gene polymorphisms with insulin resistance in Iranian individuals with newly diagnosed type 2 diabetes. Lipids Health Dis. 2019 14 June;18(1):142.
13- Azziz R, Woods KS, Reyna R, et al. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab 2004; 89: 2745–2749.
14- Yang RZ, Lee MJ, Hu H, Pray J, Wu HB, Hansen BC, Shuldiner AR, Fried SK, McLenithan JC, Gong DW. Identification of omentin as a novel depot-specific adipokine in human adipose tissue: possible role in modulating insulin action. American journal of physiology. Endocrinology and metabolism 2006; 290, 1253–1261.
15- Wang L, Zhong Y, Ding Y, et al. Elevated serum chemerin in Chinese women with hyperandrogenic PCOS. Gynecol Endocrinol 2014; 30: 746–750.
16- Kzar,H,H.,Wtwt.M.A.,Al-Gazally ,M.E. Study the glucose transport, angiogenesis and apoptosis behavioral through chemotherapy treatment according to receptors status in women with breast cancer. Indian Journal of Forensic Medicine and Toxicology,2020,14,(3),pp.2555-2559.
17- Al-Gazally,M.E., Alaa S. Al-Awad, Hamzah H. Kzar. Study Some of the Risk Factors on Total Antioxidant Capacity in Iraqi Patients with Sporadic Colorectal Cancer. Journal of Global Pharma Technology,2018, 10(3), pp:48-55.
18- Al-Hussainy,A.D.O.,Al-Gazally,M.E.,Ewadh,W. Oxidative stress, Anti-melanocyte and anti-tyrosine antibody in vitiligo and response to treatment. Annals of Tropical Medicine and Public Health ,2020 ,23(5) ,pp,500-507.
19- Kort DH, Kostolias A, Sullivan C, et al. Chemerin as a marker of body fat and insulin resistance in woman with polycystic ovary syndrome. Gynecol Endocrinol 2015; 31:152–155.
20- Bahadori M, Kohan L, Jafari N. Association of assessment between Val109Asp omentin gene and obesity in Iranian women. Iran J Diabetes Metab. 2015;14(2):127–32.
21- Schäffler A, Zeitoun M, Wobser H, Buechler C, Aslanidis C, et al. Frequency and significance of the novel single nucleotide missense polymorphism Val109Asp in the human gene encoding omentin in Caucasian patients with type 2 diabetes mellitus or chronic inflammatory bowel diseases. Cardiovasc Diabetol. 2007;6:3.
22- Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, Lindgren CM, Perry JR, Elliott KS, Lango H, Rayner NW, Shields B. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science. 2007;316(5826):889-94.
Received: 30 January 2022 / Accepted: 10 June 2022 / Published:15 August 2022
Citation: Mohsen A M, AL-Azawi R S A, Naji H H, Kzar H H, Al-Gazally M E. Investigation of the Omentin-1 V109D gene polymorphism as a risk factor for the incidence of Polycystic Ovary Syndrome. Revis Bionatura 2022;7(3) 51. http://dx.doi.org/10.21931/RB/2022.07.03.51