2022.07.03.43
Files > Volume 7 > Vol 7 No 3 2022
1 College of Education for Pure Science Ibn-Al-Haitham, University of Baghdad, Iraq; 0000-0001-8723-4675
2 College of Education for Pure Science Ibn-Al-Haitham, University of Baghdad, Iraq; [email protected] .
* Correspondence: [email protected]; Tel.: +964 750 628 7694
Available from: http://dx.doi.org/10.21931/RB/2022.07.03.43
ABSTRACT
Schiff base ligands were synthesised in this work, the first is (Z)-2-((Z)-3-((1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1H-pyrazol-4-yl)imino)-1,3-diphenylpropylidene) hydrazine-1-carboxamide, the second is (Z)-2-((1E,5Z,6E)-5-((1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1H-pyrazol-4-yl)imino)-1,7-bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-dien-3-ylidene)hydrazine-1-carbothioamide. The Schiff base ligands both were synthesised using the same method (the single pot reaction), using EtOH as the reaction medium and GAA as the catalyst in fixed temperature at 70 0C and reflux for 6 hrs. The starting materials of HL1 were semicarbazide,4-aminoantipyrine and dibenzoyl methane, while the starting materials of HL2 were TSC, 4-AAP and Curcumin.
The ligands proved to be bi-dentate ligands that coordinate from the azomethane groups. FT-IR, U.V-Visible, 1H and 13C- NMR, molar conductivity, and magnetic susceptibility were used to identify all the compounds produced. The metal ions used in preparing the complexes in this work were Co(II), Cu(II) and Cr(III). Schiff base and its complexes were evaluated for antibacterial activity against four bacterial strains of Gram-negative (Escherichia coli and Pseudomonas aeruginosa), Gram-positive (Staphylococcus aureus and Bacillus Subtilis) and two types of fungi (Candida albicans and Rhizopus Sporium), the results were positive for all compounds tested.
Keywords: Schiff base, Curcumin, 4-aminoantipyrine
INTRODUCTION
During the past few decades, the Schiff base metal complexes have been under attention due to their role in coordination chemistry, easy preparation, and the diversity of their structure. Schiff base is considered a bioactive compound; for example, these compounds behave as anti-cancer agents. The free ligands have a weaker biological effect when compared to their metal complexes 1,2. Antipyrines played an essential role in drug development fields; also, they helped in monitoring and observing patients with chronic liver illness (Hepatitis B virus (HBC), hepatitis C virus (HCV) and alcohol-related disease)3,4. 4-aminoantipyrine condensation with suitable compounds such as ketones, aldehydes, thiosemicarbazones, carbazones or compounds with a similar structure results in flexible ligands which can coordinate with various metal ions 5. β-diketones based Schiff base ligands have an important role in developing coordination chemistry; also, they play an important role in biochemical systems 6. In this study, the β-diketones used are Curcumin and dibenzoyl methane; both are known for their biological activity and behavior as antihyperglycemic, anti-inflammatory, anti-cancer, insecticidal, anti-Alzheimer's, gastroprotective, antifungal, antibacterial agents 7. Also significant are semicarbazones and thiosemicarbazones; they are made up of the scaffolds NH2–CO–NH–NH- and NH2–CS–NH–NH- respectively. Thiosemicarbazones' and semicarbazones' capacity to coordinate with transition and main group metallic cations is due to the extended delocalization of electron density over their skeleton, which is increased by substitution at the N (4)-position 8,9. The goal of this study was to synthesize and characterize Schiff bases with two azomethine groups derived from diketones (Curcumin and dibenzoylmethane), 4-amino antipyrine, semicarbazide and thiosemicarbazide, in order to test the ligand and its complexes' efficient biological activity against various types of bacteria and fungi.
MATERIALS AND METHODS
The device used to measure the melting point is (MPA160 Digi Melt); a device type (jew wary, model 4070) was used to measure the molar conductivity of the metal complexes. The FT-IR spectroscopy was evaluated using (Shimadzu FT-IR 8400S) spectrophotometer. The UV-Vis spectra of the ligands and their transition metal complexes were recorded in DMSO at 0.001M using (the UV 160A Shimadzu apparatus). Balance Johnson Mattey device was used to measure the magnetic susceptibility of the metal complexes, and NMR spectroscopy was recorded using device type (Inova 500). Finally, The Mass spectrum was recorded using (Sciex Esi) spectrometer.
Synthesis of Schiff base ligand HL1
For the preparation of HL1 a mixture of 4-aminoantipyrine, semicarbazide and dibenzoylmethane was refluxed for six hrs. with stirring at a fixed temperature of 70 0C. after that, the solution was left to evaporate for a day the resulted ligand was a yellow-colored, needle-shaped crystal and odorless.
The structure was confirmed by the data collected from elemental analysis, FT-IR, 1 H-NMR and 13 C-NMR; the synthesis of the ligand and it complexes is illustrated in Figure 1.HL1 : (Z)-2-((Z)-3-((1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro1H-pyrazol-4-yl)imino)-1,3-diphenylpropylidene) hydrazine-1-carboxamide.Yield: 75 %, elemental analysis found% :69.49 C, 5.621 H, 18.11 N, 6.78 O, Calculated%: 69.51 C, 18.01 N, 5.66 H, 6.86 O. IR (KBr): 1624 (C=N), 1670 (C=O), 3398(NH), 3210 and 3175 (NH2). 1H-NMR (δ/ppm): 2.44 (s, 3H, C-CH3); 3.21 (s, 3H, N-CH3); 10.74 (s, N-H), 7.33-7.71(m,15H aromatic); 7.21 (s, NH2).13C-NMR δ/ppm: 150.69 and 161.85 (C=N), 124.86-137.60 (Caromatic), 157.67 and 159.69, 12.76 (C-CH3), 37.32 (N-CH3), 41.15 (CH2).

Figure 1. Synthesis route for HL1 and it metal complexes
Synthesis of Schiff base ligand HL2
Curcumin (0.368 gm, 1 mmol) was dissolved in 20 ml ethanol, then adding four drops of GAA as a reaction catalyst, 0.204 grams (1mmol) 4-aminoantipyrine dissolved in 20 mL ethanol and 0.09 gm (1mmol) thiosemicarbazide dissolved in 20 mL ethanol were all progressively added at the same time. This mixture was refluxed by stirring at 70 °C for 6 hours; TLC was used to monitor the reaction. A dark brown precipitate was isolated and recrystallized from hot ethanol. The structure was confirmed by the data collected from elemental analysis, FT-IR, 1 H NMR and 13 C NMR. The synthesis of the ligand is illustrated in Figure 2. [HL2]: (Z)-2-((1E,5Z,6E)-5-((1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1H-pyrazol-4-yl)imino)-1,7-bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-dien-3-ylidene)hydrazine-1-carbothioamide. Yield: 77%.Elemental analysis, calculated for C31H30N6O3S: C 65.71, H 5.34, N 14.83, O 8.47, S 5.66%. The analysis found: C 65.64, H 5.39, N 14.92, O 8.51, S 5.54 %. IR (KBr, cm −1 ): ν ( C = O ) 1670, ν ( C = N ) 1627, ν (N-H) 3441, ν (C=S) 817. 1 H NMR (δ/ppm): 2.44 (s, 3H, –CH3 –C); 3.21 (s, 3H,–CH3 –N); 10.69 (s, N-H), 7.33-7.51(m, 15H aromatic); 9.23 (s, NH2), 9.35 (O-H), 4.26 (s, 2H, NC-CH2-CN). 13C NMR δ/ppm: 150.69 and 161.85 (C=N), 124.86-137.60 (Caromatic), 157.67 and 159.69 (HC=CH), 12.76 (C-CH3) and 37.32 (N-CH3), 41.15 (CH2).
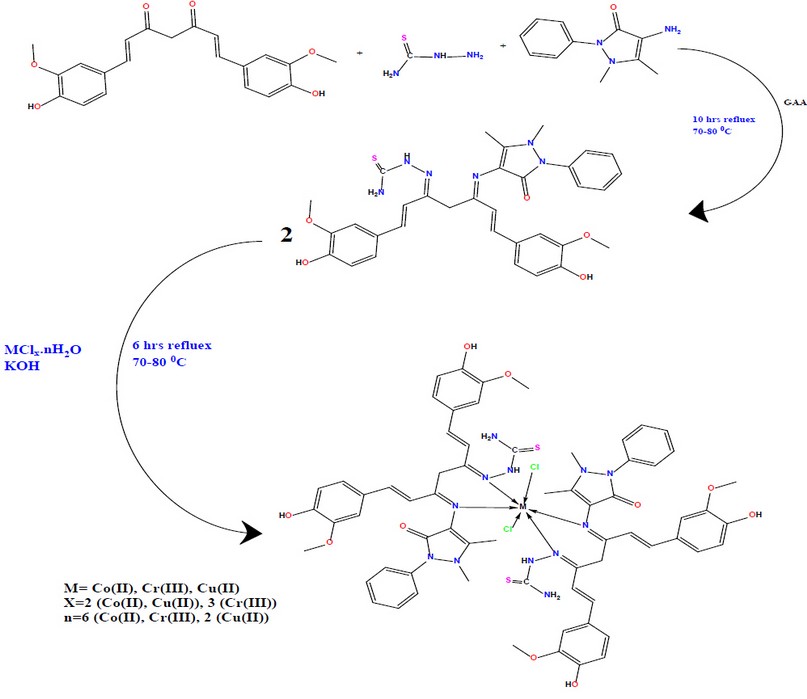
Figure 2. Synthesis route for HL2 and it metal complexes
Synthesis of metal complexes
a) [HL1] complexes: complexes were prepared by dissolving 0.466 gm (1mmol) of HL1 ligand in 10 ml ethanol, followed by adding an equivalent of KOH (0.5 mmol) of the metal salt (CoCl2.6H2O, CuCl2.2H2O, CrCl3.2H2O) dissolved in 3 ml ethanol which was added as dropwise on the mixture of the ligand with KOH, the mixture was refluxed for 6hrs with continuous stirring at fixed temperature 70-80 0C. The mixture was filtered, and the participants washed several times with cold ethanol and then recrystallized from hot ethanol. Figure 1 depicts the suggested geometry of the recrystallized structures.
b) [HL2] complexes: In 20 mL of ethanol, 0.627 gm (1mmol) of the Schiff base (ligand) was dissolved, and the equivalent of KOH was added; the solution of the metal chloride (0.5 mmol) was added dropwise addition with continual stirring and reflux for 6 hours at fixed temperature 70-80 0C, (3 mmol) of CoCl2.6H2O, CuCl2.2H2O, CrCl3.2H2O (dissolved in 3 mL ethanol) was added progressively to the mixture. The residues that formed were separated, rinsed with cold ethanol multiple times, and then recrystallized from hot ethanol. The proposed geometry of the recrystallized structure is shown in Figure 2.
RESULTS
All complexes were generally synthesized by reacting the metal salts with the Schiff base in a 1:2 mole ratio (metal salts: Schiff base). All of the complexes were colored solid compounds. Table 1 shows that the analytical findings corresponded with the proposed formula of a 1:2 metal: Schiff base molar ratio. The complexes synthesized using HL1 were soluble in dimethylformamide and dimethyl sulfoxide, and it has partial solubility in chloroform, ethanol, methanol and water. In contrast, the complexes of HL2 were soluble in water, DMSO and DMF and had a partial solubility in ethanol and methanol. Some of the physical and chemical characteristics are shown in Table 1.
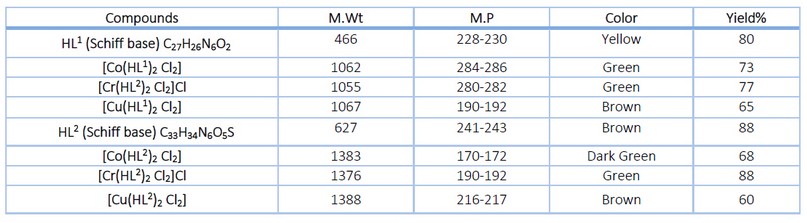
Table 1. Some of the physical and chemical characteristics of the synthesized compounds
FT-IR spectra
The spectrum of Schiff base ligands showed a band at 1624 cm-1 that referred to the imine group in HL1 and 1627 cm-1 for HL2 ligand, also a band appeared at 1670 cm-1 that assigned to the carbonyl group (C=O), the thione band was assigned at 817 cm-1 for HL2. The NH indicating band is set for HL1 at 3441 cm-1 and 3417 cm-1 for HL2, and the OH group band of [HL2] appears at 3525 cm-1 10-13. Otherwise, the spectrum of the complexes showed shifting in the imine band, and the shifting range was 1600-1624cm-1; the carbonyl group and the thione group did not show any noticeable shifting, which means the two groups did not participate in the coordination with the metal ions. Also, new bands for stretching frequencies of M-N have been allocated at 460-420 cm-1 14. Another band was observed at a range of 243-260 cm-1, indicating the formation of the M-Cl coordination bond 11. The data are listed in Table 2, and the FT-IR spectrum is shown in Figures 3 and Figure 4.

Figure 3. FT-IR spectrum of HL1

Figure 4. FT-IR spectrum of HL2.
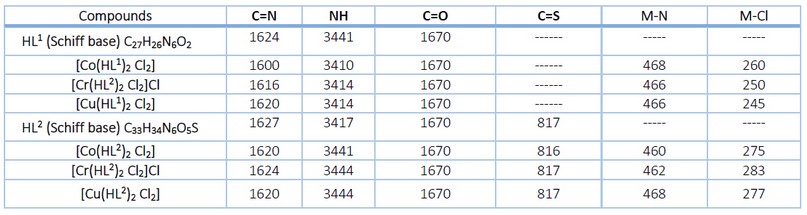
Table 2. FT-IR selected data of the ligands and their metal complexes
Electronic spectra
The ligands' absorption spectra revealed peaks at 270-290 nm due to the π →π* and n→π* peaks around 340 nm; Figure 3 shows the ranges of the ligands. The complexes revealed intra-ligand fields at 267-289 and 340-347 nm; this slight shift is considered to approve the ligand→metal coordination, charge transfer peaks at 363 nm 13-15, and d-d transition peaks as follows; The electronic spectra of ligand's HL1 metal complexes confirmed an octahedral geometry of the synthesized complexes where the peaks exhibited was like the following; Co(II) complex exhibited a peak at 782nm that is related to the electronic transition 4T1g→4T2g, Cr(III) complex showed a peak at 779 nm which refers to the electronic changes 4A2g(F)→4T2g(F) , The Cu(II) complex d-d transitions display two peaks at the d-d region at 778nm and 760 nm which are related to 2B1g→2A2g 13-17. The HL2 complexes spectral data were as follows; Co(II) complex displayed peaks at the d-d region at 700 nm related to the electronic transition 4T1g(F) →4A2g(F), the complex's magnetic moment equals 3.88 BM, the data suggested an octahedral geometry. The Cr(III) complex spectra exhibited a peak at 803 nm related to 4A2g→4T2g; the electronic spectrum and the magnetic moment of the complex, which equals 2.91 BM confirm an octahedral geometry structure. According to the electronic spectrum data collected, an octahedral geometry was suggested for the Cu(II) complex, where peaks were detected at 626 nm and 682 nm that are referring to 2Eg→2T2g and 2B1g→2A1g, and the magnetic moment of the complex is 2.15 BM 13-17.
The molar conductivity of the ligands and its complexes measured in (S.cm2 mol-1) were as following: HL1 (5.7),[Co (HL1)2Cl2] (0.7), [Cr (HL1)2Cl2]Cl (29), for [Cu (HL1)2Cl2] (3.6), HL2 (9.3), [Co (HL2)2Cl2] (23), [Cr (HL2)2Cl2]Cl (33.14) and for [Cu (HL2)2Cl2] (22).
Also, the magnetic susceptibility were measured and the results were: [Co (HL1)2Cl2] (3.962), [Cr (HL1)2Cl2]Cl (4.05), [Cu (HL1)2Cl2] (2.23), [Co (HL2)2Cl2] (3.88), [Cr (HL2)2Cl2]Cl (3.91) and for [Cu (HL2)2Cl2] (2.15).

Table 3. Electronic spectral data of the ligands and their complexes
Mass spectroscopy
In the Schiff base ligands, mass spectrum Figure 4 the HL1 spectrum has a well-defined molecular ion peak at m/z = 467 amu, which correlates (M + 1) with the Schiff base ligand's molecular formula (C27H26N6O2). The spectra of HL1 ligand reveal a succession of peaks at m/z 409, 332, 290,199, 184. 105, 70 and 56, amu, which correspond to the fragments. The strength of these peaks indicates the pieces' stabilities. At the same time, the Mass of HL2 exhibited peaks at m/z+=627 amu attributed to [M+1] of the ligands molecular formula (C33H34N6O5S). Also, it revealed peaks at 594.1, 490.1, 449.1, 390.1, 367, 339, 227, 164, 136, 77, and 54 amu that are attributed to the fragments 18.

Figure 5. This is a Figureure.: (a) mass spectrum of HL1 (b) mass spectrum of HL2
The 1H and 13C -NMR spectra of the ligands
According to a literature review, NMR spectroscopy is required to determine the structure of various compounds; the NMR spectra were recorded in DMSO-d6 (dimethyl sulfoxide) using TMS (tetramethyl silane) as standard.
For HL1 the signals were at 10.74ppm (NH), 7.21 ppm (NH2), 5.16 ppm (N=C-CH2-C=N), 7.33-7.71 ppm (H aromatic) and the signals referring to the CH3 is overlapped by the DMSO signal. While for HL2 the signs were 9.23 ppm (NH2), 9.35 ppm (OH), 10.69 ppm (NH), 7.33-7.51 ppm (H aromatic), 4.26 ppm (N=C-CH2-C=N). A slight downfield shift was observed in the signals of significant complexes 19-21. The spectra are shown in Figure 6 and Figure 7.
The 13C-NMR For HL1, the signals of the aromatic carbons are located at 124.86-137.6 ppm, the carbon of the imine (C=N) group signal at 150.69 and 161.85 ppm, the signal of the carbonyl group was located at 157.67 and 159.69 ppm, finally, for HL1 the signals of the aliphatic carbons were located at 12.76 (C-CH3) and 37.32 ppm (N-CH3) and 41.15 ppm (CH2) [19-21]. When speaking of HL2 signals, they were explained as the following 179.61ppm indicates the thione (C=S) group, the aromatic carbons are located at 124-137 ppm, while the aliphatic carbons signals are located at 12.76 (CH3) and 41.5 ppm indicating the CH2 group that is located between the imine groups, and finally, the carbons of the imine group have two signals 155.18, and 156.71 ppm and the deference in signals results from the deference in the electronic environment surrounding the group 19-21. The spectra are shown in Figure 8 and Figure 9.

Table 4. NMR spectral data of the ligands

Figure 6. 1HNMR spectrum of the first Schiff base HL1

Figure 7. 1HNMR spectrum of the first Schiff base HL2

Figure 8. 13CNMR spectrum of the first Schiff base HL1

Figure 9. 13CNMR spectrum of the first Schiff base HL2
Biological activity studies
The effect of the synthesized compounds was evaluated using the agar diffusion technique on four types of bacteria (E. Coli, P. aeruginosa, S aureus and B. Subtilis) and two types of fungi (C. albicans and Rhizopus sporium). The inhibition zone (IZ) of 1x10-3 M of the compounds was compared to the IZ of Ceftriaxone antibiotic as standard 22. The bacteria and fungi were chosen for their recognized impact on various illnesses; they exhibit varying resistance to medicines and medicinal compounds. The ligand and its complexes demonstrated a reasonable zone of inhibition.
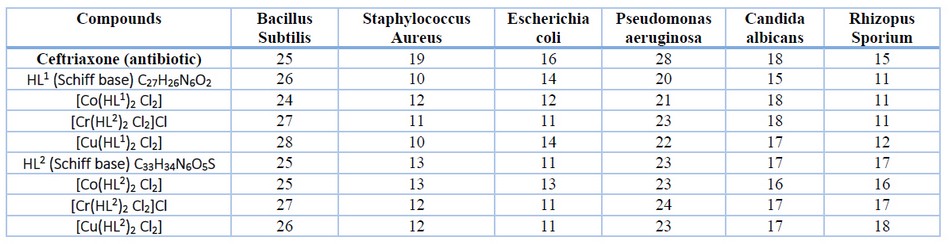
Table 5. Biological activity data of the ligands and their metal complexes
DISCUSSION
Schiff compounds were synthesized were characterized using multiple techniques, and were approved to have better biological activity than the antibiotic used as a standard and the physical activity
CONCLUSIONS
The Schiff base ligand was synthesized by reacting dibenzoyl methane 1mmol with 4-aminantipyrine 1mmol and semicarbazide 1mmol (HL1). The imine group atoms (N) performed it as bidentate coordinates. The Schiff base ligand (HL2) was synthesized by reacting curcumin 1mmol with 4-aminantipyrine 1mmol and thiosemicarbazide 1mmol. It turned out to be a bidentate ligand coordinate from the imine group. All complexes were produced based on available data such as FT-IR, UV-Visible, 13C,1H-NMR, molar conductivity, and magnetic susceptibility to adopt an octahedral geometric form. When tested for biological effectiveness against four types of bacteria and two types of fungi, all compounds gave a positive result; the synthesized compounds showed adequate inhibition in various ranges against the bacterium spices and the effect on the fungi spices.
Author Contributions: The experimental part of the research, measurements and preparation of the research draft were done by Reham H. Najem. The work was supervised and corrected by professor Hasan A. Hasan.
Acknowledgments: I gratefully acknowledge Pro. Hasan A. Hasan and Pro. Mohamad J. Al-Jeboori for the many invaluable insights and guidance until I could finish my research.
Conflicts of Interest: The authors declare no conflict of interest
REFERENCES
1. Uddin N, Rashid F, Ali S, Tirmizi SA, Ahmad I, Zaib S, Zubair M, Diaconescu PL, Tahir MN, Iqbal J, Haider A. Synthesis, characterization, and anti-cancer activity of Schiff bases. Journal of Biomolecular Structure and Dynamics. 2020 Jul 23;38(11):3246-59.
2. Mbugua SN, Sibuyi NR, Njenga LW, Odhiambo RA, Wandiga SO, Meyer M, Lalancette RA, Onani MO. New Palladium (II) and Platinum (II) complexes based on pyrrole schiff bases: synthesis, characterization, X-ray structure, and anti-cancer activity. ACS omega. 2020 Jun 19;5(25):14942-54.
3. Wissel PS. Dose-dependent suppression of hepatic cytochrome P-450 content by doxorubicin and mitomycin-C: correlation with antipyrine biotransformation. Life sciences. 1988 Jan 1;42(11):1139-45.
4. Grieco A, Matera A, Di Rocco P, Marcoccia S, Giancaterini A, Alfei B, Addolorato G, Varlese F, Gasbarrini G. Plasma levels of fibronectin in patients with chronic viral and alcoholic liver disease. Hepato-gastroenterology. 1998 Sep 1;45(23):1731-6.
5. Sarı A. Thermal Energy Storage and Applications Using Phase Change Materials. InChair of the Conference 2017 (p. 3).
6. Eshaghi Malekshah R, Salehi M, Kubicki M, Khaleghian A. New mononuclear copper (II) complexes from β-diketone and β-keto ester N-donor heterocyclic ligands: structure, bioactivity, and molecular simulation studies. Journal of Coordination Chemistry. 2018 Apr 3;71(7):952-68.
7. Goel B, Tripathi N, Bhardwaj N, Sahu B, Jain SK. Therapeutic Potential of Genus Pongamia and Derris: Phytochemical and Bioactivity. Mini Reviews in Medicinal Chemistry. 2021 May 1;21(8):920-51.
8. Cheng W, Xiao T, Qian W, Lu T, Zhang T, Tang X. Design, synthesis and insecticidal activity of novel semicarbazones and thiosemicarbazones derived from chalcone. Natural Product Research. 2021 Nov 2;35(21):3801-9.
9. Ali I, Saleem K, Wesselinova D, Haque A. Synthesis, DNA binding, hemolytic, and anti-cancer assays of Curcumin I-based ligands and their ruthenium (III) complexes. Medicinal chemistry research. 2013 Mar;22(3):1386-98.
10. Danilescu O, Bourosh PN, Petuhov O, Kulikova OV, Bulhac I, Chumakov YM, Croitor L. Crystal Engineering of Schiff Base Zn (II) and Cd (II) Homo-and Zn (II) M (II)(M= Mn or Cd) Heterometallic Coordination Polymers and Their Ability to Accommodate Solvent Guest Molecules. Molecules. 2021 Jan;26(8):2317.
11. Tashi L, Singhaal R, ul Nisa Z, Devi S, Sheikh HN. Asparagine modified downconversion NaGdF 4: Dy 3+/Tb 3+ nanophosphor for selective and sensitive detection of Cu (ii) ion. New Journal of Chemistry. 2021;45(34):15392-404.
12. Gultekin E, Bekircan O, Kolcuoğlu Y, Akdemir A. Synthesis of new 1, 2, 4‐triazole–(thio) semicarbazide hybrid molecules: Their tyrosinase inhibitor activities and molecular docking analysis. Archiv der Pharmazie. 2021 Aug;354(8):2100058.
13. Ali AM, Al-Noor TH. Synthesis, spectroscopic study, biological activity and dyeing application of curcumin-Schiff base with various metal ions complexes. InIOP Conference Series: Materials Science and Engineering 2021 Feb 1 (Vol. 1046, No. 1, p. 012006). IOP Publishing.
14. Radha VP, Chitra S, Jonekirubavathi S, Chung IM, Kim SH, Prabakaran M. Transition metal complexes of novel binuclear Schiff base derived from 3, 3′-diaminobenzidine: synthesis, characterization, thermal behavior, DFT, antimicrobial and molecular docking studies. Journal of Coordination Chemistry. 2020 Mar 18;73(6):1009-27.
15. Al-Qazzaz AH, Al-Jeboori MJ. New metal complexes derived from mannich ligands: Synthesis, spectral investigation and biological activity. Biochem. Cell. Arch. 2020;20:4207-16.
16. Hussien AK, Yousif EI, Hasan HA, Ahmed RM. Metal Complexes Derived from Mixed Azo-Linked Schiff-Base Ligandwith Dithiocarbamate Derivative: Formation, Spectral Characterization and Biological Study. Oriental Journal of Chemistry. 2018;34(3):1492.
17. Chandra S, Gautam A. Spectroscopic and biological approach in the characterization of Cr (III), Mn (II) and Co (II) complexes with a novel hexaazamacrocyclic ligand derived from semicarbazide. Journal of the Serbian Chemical Society. 2009;74(12):1413-22.
18. Naureen B, Miana GA, Shahid K, Asghar M, Tanveer S, Sarwar A. Iron (III) and zinc (II) monodentate Schiff base metal complexes: Synthesis, characterisation and biological activities. Journal of Molecular Structure. 2021 May 5;1231:129946.
19. Saritha TJ, Metilda P. Synthesis, spectroscopic characterization and biological applications of some novel Schiff base transition metal (II) complexes derived from curcumin moiety. Journal of Saudi Chemical Society. 2021 Jun 1;25(6):101245.
20. Naureen B, Miana GA, Shahid K, Asghar M, Tanveer S, Sarwar A. Iron (III) and zinc (II) monodentate Schiff base metal complexes: Synthesis, characterisation and biological activities. Journal of Molecular Structure. 2021 May 5;1231:129946.
21. Kheirkhahi M, Shaabani B, Samadi Kafil H. Calix [4] arene-based thiosemicarbazide Schiff-base ligand and its transition metal complexes: synthesis and biological assessment. Journal of the Iranian Chemical Society. 2021 Dec;18(12):3429-41.
22. Ali AM, Al-Noor TH. Synthesis, identification, antibacterial, and dyeing applications of complexes of hexadentate (N4O2 donor) Schiff base ligands derived from Curcumin with some transition and non–transition metal cations. InIOP Conference Series: Materials Science and Engineering 2021 Feb 1 (Vol. 1046, No. 1, p. 012005). IOP Publishing.
Received: 3 March 2022 / Accepted: 23 May 2022 / Published:15 August 2022
Citation: Hasan R H. and Hasan H A. Synergism antibacterial activity for novel synthesized Schiff base ligands and semi-thiosemicarbazones with ß-diketones and 4-aminoantipyrine. Revis Bionatura 2022;7(3) 43. http://dx.doi.org/10.21931/RB/2022.07.03.43
