2021.06.04.22
Files > Volume 6 > Vol 6 No 4 2021
Role of Leptin with hypothyroidism in Iraqi diabetic type 2 patients
Sulaiman M. Hasan
Available from http://dx.doi.org/10.21931/RB/2021.06.04.22
ABSTRACT
In thyroid subjects, leptin hormone and thyroid-stimulating hormone levels connect; both are emphatically corresponding with adiposity. “Leptin hormone was essentially raised in the hypothyroid topic,” to levels like those seen in corpulent thyroid topic. This study aimed to determine leptin hormone levels, T3, T4, TSH , HbA1c %, FBG, lipid profile in diabetic and diabetic with hypothyroidism patients and compare the outcome with the healthy group. 90 samples were registered in this study with their ages ranging between (40 - 65) years that dole out into 3 groups as follows: thirty healthy groups (G1), thirty patients with diabetes (G2), and category three (G3) include diabetic patients and hypothyroidism as an associated disease. This study revealed a slightly significant elevated leptin in G3, contrasting to G2 and G1. Meantime no significant elevated spotted between G2 and G1. The conclusion could be from this investigation that leptin levels were changed in patients’ gatherings that might be utilized in checking and early analysis of thyroid dysfunction in these patients relying upon the significant connection for leptin chemical with T3, T4, and TSH.
Keywords: Leptin hormone, hypothyroidism, and diabetic type2.
INTRODUCTION
Diabetes mellitus (DM) and thyroid disorders are two endocrine problems that influence each other in various reactions. Thyroid chemicals add to the guideline of carbohydrates digestion and pancreatic capacity1,2. Sensible components for progression of T2DM in thyroid issue patients might be identified with the upset hereditary articulation of qualities related to physiological distortions prompting hindered glucose utilization by the expanded hepatic glucose yield, muscles, and glucose absorption rise from digestive system3. Leptin is a (146) amino acid protein chemical secreted by adipocytes in light of an increment in fat mass. It is by all accounts a vital atom in the input circle that directs energy balance. Leptin has a double activity: it diminishes hunger and builds energy utilization, making more fat scorched4. Leptin gives data to the sensory system on the measure of energy put away in the fat tissue. Serum leptin levels profoundly correspond with muscle to fat ratio mass in grown-ups, youngsters, and babies. Obese individuals have altogether higher flowing leptin than typical. In obesity, a diminished affectability to leptin happens (like insulin opposition in type 2 diabetes), bringing about powerlessness to recognize satiety despite high energy stores and high levels of leptin5. Thyroid hormones and leptin are two chemicals that manage power balance via central signaling mechanisms6. Aggravation of thyroid capacity is related to noticeable changes in energy consumption and body weight; furthermore, it has accordingly been the subject of much exploration to contemplate the shared jobs of thyroid hormones and leptin in this regard7. In thyroid subjects, leptin hormone and thyroid-stimulating hormone levels connect; both are emphatically corresponding with adiposity. “Leptin hormone was essentially raised in the hypothyroid topic” to levels like those seen in corpulent thyroid topic. “The information is steady with the speculation that leptin hormone and the pituitary-thyroid pivot interface in the thyroid state, and that hypothyroidism reversibly increments leptin focuses8-11.
MATERIAL S & METHODS
Ninety individuals with ages ranging between (40-65) years were joined up with this examination.
They separated into three gatherings as follows:-
1. Gathering (G1) that comprises 30 sound people as control bunch.
2. Gathering (G2) that consists of 30 diabetic patients.
3. Gathering (G3) that includes diabetic patients and hypothyroidism as an associated disease.
Blood samples were gathered from all gatherings after a time of fasting 12-14 hours. The study was conducted between December 2020 – April 2021 in the diabetic & endocrinology center in Al-Yarmouk Teaching Hospital / Iraq.
Estimation of Leptin levels by Competitive Elisa Reveals12. The T3, T4, and TSH were determined by Enzyme-linked Fluorescent Immunoassay (ELFA) competition method with a final detection13. Whole blood was used in the determination of HbA1c. The HPLC method14 determined the HbA1c. Serum glucose was measured using kits from (Randox Company, United Kingdom) based on the PAP enzymatic determination of glucose15. TC 16, TG17, and high-density lipoprotein18 were estimated using the enzymatic method (Human Gesellschaft fur biochemical and Diagnostica mbH, Germany). The levels of LDL and VLDL were analyzed by using Friedewald equation19.
Statically study outcomes were communicated as mean ± SD. T-test was used for comparison among the three studied groups. The P-vales (< 0.05) no significant, (> 0.05) significant, (> 0.01) highly significant were considered statically.
RESULTS & DISCUSSION
Descriptive was introduced in Table (1), which shows the levels of HbA1c%, F.S.G, T3, T4, TSH, and leptin for every considered gathering. Table (1) showed a highly significant increase in patients’ gatherings (G2, G3) contrasting with control bunch in HbA1c% and FSG levels. Likewise, a significant rise in G3 contrasted with G2 was found.
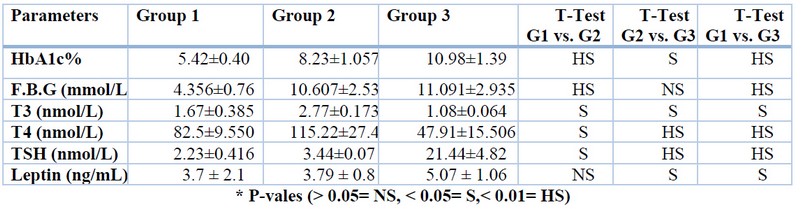
Table 1. HbA1c%, F.S.G, T3, T4, TSH, and leptin hormone levels for groups1, 2, 3.
Results uncovered a significant decrease in levels of T4 and T3 in G3 when contrasted with group one and group two. Furthermore, there is a highly significant increase in TSH level in group three contrasted with groups one and two.
Diabetic patients appear to impact thyroid capacity in the hypothalamic control of thyroid-stimulating hormone discharge and at peripheral tissue by changing T4 to T3. High glucose levels lead to decreased deiodinase enzyme concentration in the liver, decreased T3, rising levels of opposite T3, and low, typical, or undeniable levels of T420.
Results in Table 1 uncovered a significant rise in listening levels in G3, contrasting with G1and G2. These outcomes show that serum leptin is somewhat raised in subjects with moderate hypothyroidism, potentially because of the immediate activity of thyroid hormones.
Leptin is another hormone assuming a significant part in the guideline of power balance by tweaking food consumption, thermogenesis, just as lipid and glucose digestion. Concerning interactions between leptin and thyroid hormones, the impact of thyroid capacity and hypothyroidism, specifically on flowing leptin hormone levels, gave clashing information. Subsequently, diminishes, increments, or non-change in leptinaemia were accounted for in hypothyroid patients. This is the way might clarify disparate outcomes that leptin hormone levels fundamentally invert changes in lipid cluster, this boundary differing as indicated by sex, age, and span of hypothyroidism21.
Table (2) shows the levels of lipid profile for G1, G2, and G3. Results revealed a significant rise in lipid profile levels without HDL in groups two and three, contrasting to G1. Results likewise showed a significant rise in lipid profile without HDL in G3 contrasting to G2; Results also showed a significant decline in HDL levels in group two and group three contrasting with G1, while no significance was found in group two-three contrasting with group two in HDL levels.
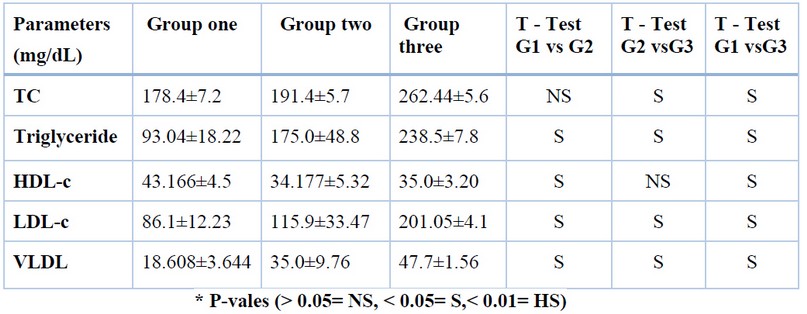
Table 2. Lipid profile levels for groups 1,2,3.
CONCLUSIONS
The conclusion could be from this investigation that leptin levels were changed in patients’ gatherings that might be utilized in checking and early analysis of thyroid dysfunction in these patients relying upon the significant connection for leptin chemical with T3, T4, and TSH.
REFERENCE
1. Kim, Mee Kyoung, Seung-Hyun Ko, Bo-Yeon Kim, Eun Seok Kang, Junghyun Noh, Soo-Kyung Kim, Seok-O. Park et al. “2019 Clinical practice guidelines for type 2 diabetes mellitus in Korea.” Diabetes & metabolism journal 43, no. 4 (2019): 398.
2. Fleiner, H.F.; Bjøro, T.; Midthjell, K.; Grill, V.; Åsvold, B.O. Prevalence of Thyroid Dysfunction in Autoimmune and Type 2 Diabetes: The Population-Based HUNT Study in Norway, The Journal of Clinical Endocrinal Metabolism. 2016, 101, 669-677.
3. Chen, Rong-Hsing, Huey-Yi Chen, Kee-Ming Man, Szu-Ju Chen, Weishan Chen, Po-Len Liu, Yung-Hsiang Chen, and Wen-Chi Chen. “Thyroid diseases increased the risk of type 2 diabetes mellitus: A nation-wide cohort study.” Medicine 98, no. 20 (2019).
4- Ibrahim, Maher K., A. H. Al-Samarrai, and Khudhair A. Khudhair. “Association between leptin hormone and thyroid hormone levels in hypothyroid, hyperthyroid and euthyroid subjects.” Front Biomed Sci 1 (2016): 39-44.
5. Betry, C., M. A. Challan-Belval, A. Bernard, A. Charrie, Jocelyne Drai, M. Laville, Charles Thivolet, and E. Disse. “Increased TSH in obesity: Evidence for a BMI-independent association with leptin.” Diabetes & metabolism 41, no. 3 (2015): 248-251.
6. Considine, Robert V. “Human leptin: an adipocyte hormone with weight-regulatory and endocrine functions.” In Seminars in vascular medicine, vol. 5, no. 01, pp. 15-24. Copyright© 2005 by Thieme Medical Publishers, Inc., 333 Seventh Avenue, New York, NY 10001 USA, 2005.
7. Wang, Lai, Yvonne Y. Shao, and R. Tracy Ballock. “Leptin synergizes with thyroid hormone signaling in promoting growth plate chondrocyte proliferation and terminal differentiation in vitro.” Bone 48, no. 5 (2011): 1022-1027.
8. Feldt-Rasmussen, Ulla. “Thyroid and leptin.” Thyroid 17, no. 5 (2007): 413-419.
9. Yoshida, Tadashi, Toshiaki Monkawa, Matsuhiko Hayashi, and Takao Saruta. “Regulation of expression of leptin mRNA and secretion of leptin by thyroid hormone in 3T3-L1 adipocytes.” Biochemical and biophysical research communications 232, no. 3 (1997): 822-826.
10. Escobar-Morreale, Héctor F., Francisco Escobar del Rey, and Gabriella Morreale de Escobar. "Thyroid hormones influence serum leptin concentrations in the rat." Endocrinology 138, no. 10 (1997): 4485-4488.
11. Pinkney, Jonathan H., Steven J. Goodrick, Jonathan Katz, Andrew B. Johnson, Stafford L. Lightman, Simon W. Coppack, and Vidya Mohamed‐Ali. “Leptin and the pituitary–thyroid axis: a comparative study in lean, obese, hypothyroid and hyperthyroid subjects.” Clinical endocrinology 49, no. 5 (1998): 583-588.
12. Soliman, Ashraf T., Magdi Omar, Hala M. Assem, Ibrahim S. Nasr, Mohamed M. Rizk, Wael El Matary, and Rania K. El Alaily. “Serum leptin concentrations in children with type 1 diabetes mellitus: relationship to body mass index, insulin dose, and glycemic control.” Metabolism-Clinical and Experimental 51, no. 3 (2002): 292-296.
13. Anderson, Rouven, Ralf Mueller, Sven Reese, and Astrid Wehner. “Evaluation of an automated enzyme-linked fluorescent assay for thyroxine measurement in cat and dog sera.” Journal of Veterinary Diagnostic Investigation 29, no. 3 (2017): 278-286.
14. Little, Randie R., Curt Rohlfing, and David B. Sacks. “The National Glycohemoglobin Standardization Program: over 20 years of improving hemoglobin A1c measurement.” Clinical chemistry 65, no. 7 (2019): 839-848.
15. Heo, Nam Su, Han Pill Song, Sang Moon Lee, Hye-Jin Cho, Hae Jin Kim, Yun Suk Huh, and Moon Il Kim. Rosette-shaped graphitic carbon nitride acts as a peroxidase mimic in a wide ph range for fluorescence-based determination of glucose with glucose oxidase. Microchimica Acta 187, no. 5 (2020): 1-11.
16. Talbot, Charlotte PJ, Jogchum Plat, Andreas Ritsch, and Ronald P. Mensink. “Determinants of cholesterol efflux capacity in humans.” Progress in lipid research 69 (2018): 21-32.
17. Kiss, Lóránd, Gabriella Fűr, Péter Mátrai, Péter Hegyi, Emese Ivány, Irina Mihaela Cazacu, Imre Szabó et al. “The effect of serum triglyceride concentration on the outcome of acute pancreatitis: systematic review and meta-analysis.” Scientific reports 8, no. 1 (2018): 1-14.
18. Ebtehaj, Sanam, Eke G. Gruppen, Stephan JL Bakker, Robin PF Dullaart, and Uwe JF Tietge. “HDL (High-Density Lipoprotein) Cholesterol Efflux Capacity Is Associated With Incident Cardiovascular Disease in the General Population: A Case-Control Study From the PREVEND Cohort.” Arteriosclerosis, thrombosis, and vascular biology 39, no. 9 (2019): 1874-1883.
19. Ephraim, Richard KD, Emmanuel Acheampong, Swithin M. Swaray, Enoch Odame Anto, Hope Agbodzakey, Prince Adoba, Bright Oppong Afranie et al. “Developing a modified low-density lipoprotein (M-LDL-C) Friedewald’s equation as a substitute for direct LDL-C measure in a Ghanaian population: a comparative study.” Journal of lipids 2018 (2018).
20. Shivanada, N. Manipal Manual of clinical biochemistry 3rd Edition. Medical publishers (p) LTD. 2007.
21.Cettour-Rose, P., C. Theander-Carrillo, C. Asensio, Mark Klein, T. J. Visser, A. G. Burger, C. A. Meier, and Francoise Rohner-Jeanrenaud. “Hypothyroidism in rats decreases peripheral glucose utilisation, a defect partially corrected by central leptin infusion.” Diabetologia 48, no. 4 (2005): 624-633.
Received: 14 August 2021
Accepted 17 September 2021
Sulaiman M. Hasan
Ph.D, biochemistry, Collage of Dentistry, Al-Farahidi University/Iraq.
*E-mail of the corresponding author: [email protected]
Orcid: https://orcid.org/0000-0002-5117-2027
