2022.07.03.45
Files > Volume 7 > Vol 7 No 3 2022
Abdulla L. Jiad1, May K. Ismael2 , Salwa S. Muhsin3 , Bahaa Abdullah Laftaah Al-Rubaii4*
1 Department of Anesthesia, Institute of Medical Technology, Baghdad, Iraq
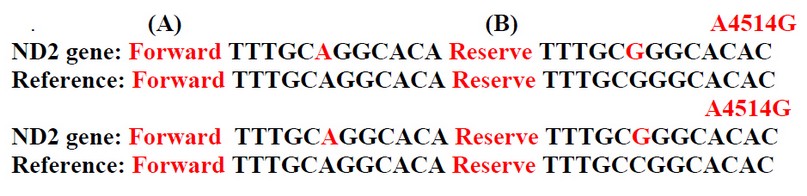

2,4 Department of Biology/ College of Science/ University of Baghdad
3Department of Community Health, Institute of Medical Technology, Baghdad, Iraq
*Corresponding author's email is: [email protected]
Available from: http://dx.doi.org/10.21931/RB/2022.07.03.45
ABSTRACT
A total of (90) blood samples were collected from male patients infected with Toxoplasmosis who recovered from COVID- 19 and attended Kamal Alsamiraai Hospital from 15 January to 15 September 2021. We measured anti-Toxoplasma antibodies (IgG and IgM) detected by ELISA, whereas Anti-COVID-19 antibodies (IgG and IgM) were estimated using Elisa and Afilias. The semen characteristics were also studied among fertile, healthy individuals (control group) and sub-fertile patients. Results showed that the mean sperm count was high among the control group (40.5±1.3x 106/ml) compared with that of the sub-fertile patients (10.3±1.75 and 8.8±1.9 x 106/ml for oligozoospermia, and oligoasthenozoospermia respectively), and it was the highest (44.7±1.4 x 106/ml) among asthenozoospermia patients. Compared to the control group, there were highly significant differences between anti-Toxoplasma IgG antibodies and anti-COVID-19 IgG antibodies (P<0.001). The mean level of Toxoplasma IgM was (11.74±8.90) and for control was (0.05±0.10), while the mean level of COVID-19 IgM was (1.91±1.06) and for control was (0.04±0.03) in sub-fertile patients. The mutation occurred in IL-IB gene A to G transgene at site 4514 of the IL-IB gene (sample code, 6383) and in the case of an invalid sample code, 2409 and 5097. In the alanine codon, the GCA codon has mutated into GCG. Also, G to A transgene occurred at site 4514 of the IL-IB gene. (sample code, 6750) In the case of an invalid sample code, it happened in 010081 and 009593. In the alanine codon, the ATG codon has mutated into ATA.
Keywords: ND2 Gene, sequence, Sub-fertile patients, COVID-19, Toxoplasmosis
INTRODUCTION
SARS CoV-2 is the riskiest virus known in the last few years because it mutates and resists treatments and possibly resists vaccines 1. Many complications accompany SARS-CoV-2 infection, including pneumonia, severe oxygen deficiency and blood clots, and other complications that may occur in various body parts 2. Several examinations must be done to determine the damage caused by infection with COVID- 19, which may reflect pathological conditions that may be risky, or there may be a malfunction in some parts of the body 3. When infected with COVID- 19, some men may be exposed to damage to sperm production, which is low or nonexistent, leading to male infertility 4, 5. There may be a partnership between COVID-19 and Toxoplasma in the occurrence of damage in the testis of men that may deprive them of their fertility due to the damage that occurs to the testicles or the sperm production channels 6. Infertility is determined by various means, including counting under a light microscope or by more sophisticated devices and the work of genetic sequencing of patients who suffer from infertility 7. Several phenomena are seen in male infertile patients, such as oligozoospermia, oligoasthenozoospermia asthenozoospermia; are all of which may be seen in persons with infertility 8. The MT-ND2 gene produces a 39 kDa protein composed of 347 amino acids. MT-ND2 is one of seven mitochondrial genes encoding subunits of the enzyme NADH dehydrogenase (ubiquinone) 9. Our study aimed to determine the ND2 gene in patients who recovered from COVID-19 and have Toxoplasmosis. The current study sheds light on infertile men exposed to COVID-19 who have recovered and been infected with Toxoplasma gondii.
MATERIALS AND METHODS
A total of 90 blood samples were collected from male patients infected with Toxoplasmosis who recovered from COVID-19 and attended Kamal Alsamiraai Hospital from 15 January to 15 September 2021. From each participant, whole blood was dispensed in gel tubes, and serum was separated by centrifugation at 3000 rpm for 5 min; the obtained serum was divided into Eppendorf tubes and kept in the freezer at -20°C till used in further immunological tests. ELISA kit was used to detect Anti Toxoplasma antibodies (IgG and IgM), whereas Anti-COVID-19 antibodies (IgG and IgM) were estimated by Afilias technique depending on the manufacturer s instructions. Semen samples were obtained by masturbation near the semen analysis lab to reduce seminal exposure to temperature fluctuations and control the time from collection to analysis. To amplify the IL-IB gene, Genomic DNA was extracted from the sample PCR was carried out in a total volume of 50 μl reaction mixture, containing 10 μl of solution, DNA pre-mix buffer, and primers (20 pmol/reaction). PCR reactions have been optimized for each primer pair with various annealing temperatures. About 160 ngs of the total sperm's DNA have been used in 50 μL of the reaction mixtures; Automated DNA sequencing was performed using the Applied BioSystem Big DyeTM termination V 3.1 cycle sequencing kit. The mtDNA samples of different fertility groups were sequenced for the ND2 gene.
Statistical analysis
The SPSS (16) program and Microsoft office excel were applied for statistical analysis. Numeric data were expressed as mean (±) SEM (standard error of the mean), and the p-value <0.05 was regarded as significant 9.
RESULTS
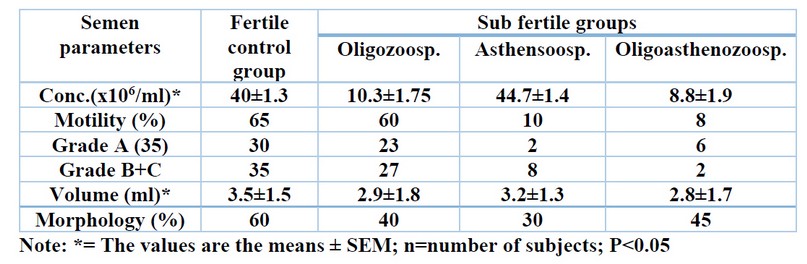
Table (1) shows the semen characteristics among fertile, healthy individuals (control group) and the sub-fertile group. The mean sperm count was high among the control group (40.5±1.3x 106/ml) as compared with that of the sub-fertile group, which was (10.3±1.75 and 8.8±1.9 x 106/ml for oligozoospermia, and oligoasthenozoospermia respectively), and it was the highest (44.7±1.4 x 106/ml) among asthenozoospermia patients.

Table 1. Comparison of semen characteristics between subfertile patients and the control group
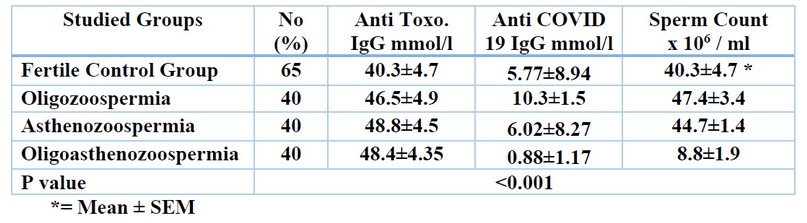
Results in the table (2) showed that there were highly significant differences in mean anti-Toxoplasma and anti-COVID-19 IgG antibodies and sperm count in comparison with patients' control group (P < 0.001).

Table 2. Correlation between sperm count & Anti Toxoplasma and COVID-19 IgGs among studied groups
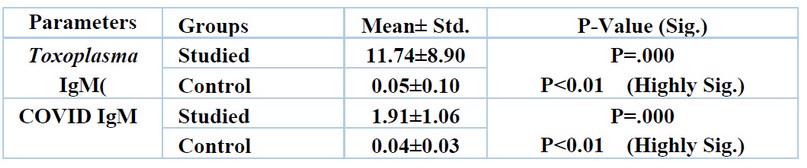
The mean level for Toxoplasma IgM was (11.74±8.90) mmol/l and for control IgM was (0.05±0.10), while the mean level for COVID IgM was (1.91±1.06) mmol/l and for control IgM was (0.04±0.03) mmol/l in sub-fertile patients with a highly significant difference (P<0.01), as shown in table (3).

Table 3. Comparison between the studied group and control group according to IgM level
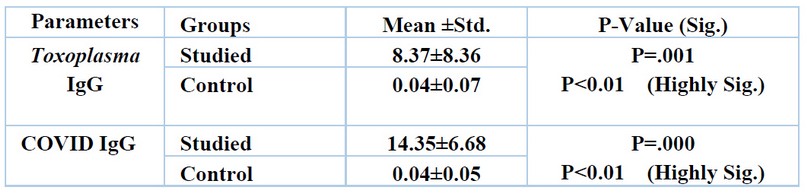
Table (4) showed that the mean level for Toxoplasma IgG was (8.37±8.36) and for control IgG was (0.04±0.07), while the mean level for COVID IgG was (14.35±6.68) and for control IgG was (0.04±0.05), in sub-fertile patients with a highly significant difference (P<0.01).

Table 4. Comparison between the studied group and control group according to IgG level
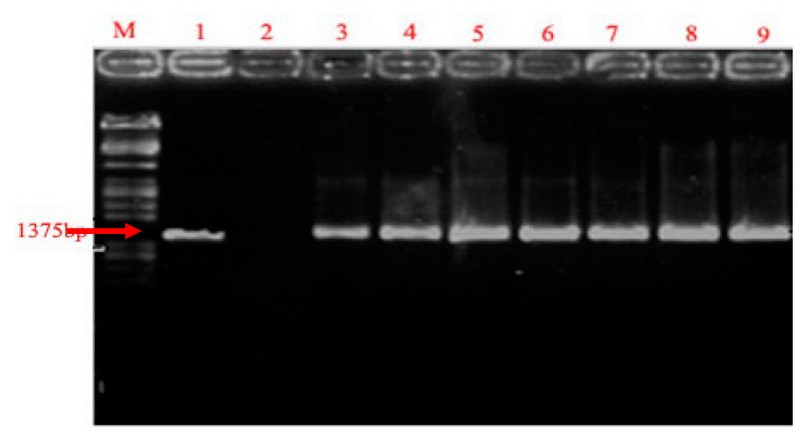
Detection of the IL-IB gene by PCR to determine the Normozoospermic, asthenozoospermic, and oligozoospermic, by using 1.2 gel for Electrophoresis under 300 UV using buffer solution TBE x 1.

Figure 1. Polymerase products of IL-IB gene at 1375bp. Lane M: ladder lane 1: Positive control for IL-IB), lane 2: negative control (sample without extracted DNA), lane 3-9: positive IL-IB gene. Using 1.2 gel for Electrophoresis under 300 UV and buffer solution TBE x 1.
Gene sequences:
Primer: Forward, TTCAAGGAGGACGGCAACAT, and Reverse, GTTCTGCTGGTAGTGGTCGG.
A to G 4514:
A to G transgene at site 4514 of the IL-IB gene. (sample code, 6383) and in the case of an invalid sample code, 2409 and 5097. In the alanine codon, where the GCA codon has mutated into GCG as shown in figure (2).
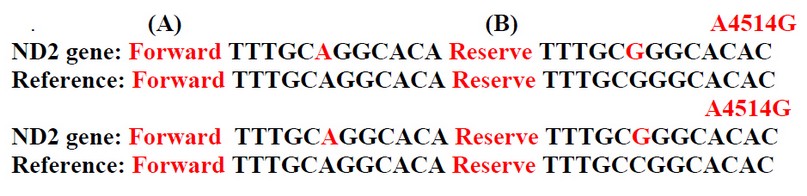
Figure 2. The occurrence of genetic mutation A: A to G, B: G to A G to A 4580
G to A transgene at site 4514 of the IL-IB gene. (sample code, 6750) and in the case of a weak sample code, 010081 and 009593. In the alanine codon, where the ATG codon has mutated into ATA.

Figure 3. A: The occurrence of genetic mutation in IL-IB gene A, B, C and D: G to A
DISCUSSION
SARS CoV-2 is a dangerous virus that causes severe conditions that may lead to death, cause infertility in men, and may be associated with toxoplasmosis 10. The phenotypes identified in infertile patients who tested positive for COVID-19 and cured and who had Toxoplasmosis were asthenozoospermic, oligozoospermic and oligoasthenozoospermic among those who shared viral and cellular parasitic infections. These finding matched the results of (Abobaker, A. and Raba, A. A. 2020) 11, who reported that the rate of infertility in men due to infection with COVID-19 or the so-called SARS-2 were asthenozoospermic at 42%, oligozoospermic 33% and oligoasthenozoospermic, 40% 12. There was a significant difference between IgM antibodies of COVID-19 and the control group, where the mean IgM value was (1.91±1.06) compared with the control group (0.04±0.05) in sub-fertile patients. Hou, H. et al. (2020) reported a high increase in anti- IgM and IgG antibodies in COVID-19 infections compared to the healthy group, as it deviated from its normal levels with a significant standard measure 13. Also, there was an increase in the anti-IgM and IgG antibodies in Toxoplasmosis compared to the control group in sub-fertile men with Toxoplasmosis. This was explained by (Hlaváčová, J. et al., 2021), who stated that the identification of antibodies to Anti Toxoplasma antibodies increases in most patients, especially those who have infertility due to this parasite 13.
A to G transgene is found at site 4514 of the IL-IB gene (sample code, 6383) and in the case of an invalid sample code, 2409 and 5097. In the alanine codon, the GCA codon is mutated into GCG 14. These results were in harmony with (Alkhuriji, A. F. et al., 2020), who reported that the genetic mutation that occurs on the interleukin-beta and interleukin-6 genes due to Toxoplasma or what is shared by a virus that may be SARS-2, for example, changes the genetic sequence G to C, or vice versa along these genes. Sterility occurs in both men and women 15. G to A transgene is found at site 4514 of the IL-IB gene (sample code 6750) and in the case of an invalid sample code 010081 and 009593. In the alanine codon, the ATG codon is mutated into ATA. This SNP is a synonymous substitution that occurred in the third position of the methionine codon, changing the codon from ATG to ATA. These findings agreed with another finding 16, which found that the mutation induces azoospermia in sub-fertile men, as the mutation was (2653G → T, in exon 29 16. In this study, it has been proven that with the participation of COVID-19 and Toxoplasmosis, men with infertility were exposed to infection with COVID-19 and cured of the disease. Still, they had Toxoplasmosis, so they had a double infection, which led to a genetic mutation in the sperm, leading to infertility 17.
CONCLUSION
We concluded that the mutation occurred in IL-IB gene A to G transgene at site 4514 of the IL-IB gene (sample code, 6383) and in the case of a weak sample code, 2409 and 5097. In the alanine codon, the GCA codon has mutated into GCG. Also, G to A transgene occurred at site 4514 of the IL-IB gene.
Acknowledgment: Thanks going for all who support us.
Conflict between authors: No conflict
Funds: self by authors
REFERENCES
1. Excler JL, Saville M, Berkley S, Kim JH. Vaccine development for emerging infectious diseases. Nature medicine. 2021 Apr;27(4):591-600..
2. Jain U. Effect of COVID-19 on the Organs. Cureus. 2020 ;12(8). : e9540.
3. Woods JA, Hutchinson NT, Powers SK, Roberts WO, Gomez-Cabrera MC, Radak Z, Berkes I, Boros A, Boldogh I, Leeuwenburgh C, Coelho-Júnior HJ. The COVID-19 pandemic and physical activity. Sports Medicine and Health Science. 2020;2(2):55-64.
4. Huang C, Ji X, Zhou W, Huang Z, Peng X, Fan L, Lin G, Zhu W. Coronavirus: a possible cause of reduced male fertility. Andrology. 2021;9(1):80-7.
5. Khalili MA, Leisegang K, Majzoub A, Finelli R, Selvam MK, Henkel R, Mojgan M, Agarwal A. Male fertility and the COVID-19 pandemic: systematic review of the literature. The world journal of men's health. 2020;38(4):506.
6. Cariati F, D'Argenio V, Tomaiuolo R. The evolving role of genetic tests in reproductive medicine. Journal of translational medicine. 2019;17(1):1-33.
7. Wang F, Yang W, Ouyang S, Yuan S. The Vehicle Determines the Destination: The Significance of Seminal Plasma Factors for Male Fertility. International Journal of Molecular Sciences. 2020;21(22):8499..
8. Yang S, Huo Y, Wang H, Ji J, Chen W, Huang Y. The spatio-temporal features of chicken mitochondrial ND2 gene heteroplasmy and the effects of nutrition factors on this gene. Scientific reports. 2020;10(1):1-9.
9. Serdar CC, Cihan M, Yücel D, Serdar MA. Sample size, power and effect size revisited: simplified and practical approaches in pre-clinical, clinical and laboratory studies. Biochemia medica. 2021;31(1):27-53.
10. Batiha O, Al‐Deeb T, Al‐zoubi EA, Alsharu E. Impact of COVID‐19 and other viruses on reproductive health. Andrologia. 2020;52(9):e13791.
11. Abobaker A, Raba AA. Does COVID-19 affect male fertility?. World journal of urology. 2021;39(3):975-6.
12. Hou H, Wang T, Zhang B, Luo Y, Mao L, Wang F, Wu S, Sun Z. Detection of IgM and IgG antibodies in patients with coronavirus disease 2019. Clinical & translational immunology. 2020;9(5):e1136.
13. Hlaváčová J, Flegr J, Řežábek K, Calda P, Kaňková Š. Association between latent toxoplasmosis and fertility parameters of men. Andrology. 2021;9(3):854-862.
14. Girard, Ch. Akerib, Ch. C. , Villeneuve, A. M. Suppression of him-14(it44ts) by a transgene insertion expressing GFP::COSA-1, Micro-publication biology: 8(24): 2021.
15. Alkhuriji AF, Al Omar SY, Babay ZA, El-Khadragy MF, Mansour LA, Alharbi WG, Khalil MI. Association of IL-1β, IL-6, TNF-α, and TGFβ1 gene polymorphisms with recurrent spontaneous abortion in polycystic ovary syndrome. Disease Markers. 2020;2020.:6076274.
16. Sha Y, Zheng L, Ji Z, Mei L, Ding L, Lin S, Wang X, Yang X, Li P. A novel TEX11 mutation induces azoospermia: a case report of infertile brothers and literature review. BMC Medical Genetics. 2018;19(1):1-7.
17. Flegr J. Toxoplasmosis is a risk factor for acquiring SARS-CoV-2 infection and a severe course of COVID-19 in the Czech and Slovak population: a preregistered exploratory internet cross-sectional study. Parasites & Vectors. 2021;14(1):1-1.https://doi.org/10.1186/s13071-021-05021-9.
Received: 13 March 2022 / Accepted: 25 July 2022 / Published:15 August 2022
Citation: Abdulla L. Jiad , May K. Ismael , Salwa S. Muhsin , Bahaa Abdullah Laftaah Al-Rubaii . ND2 Gene Sequencing of Sub fertile Patients Recovered from COVID-19 in Association with Toxoplasmosis. Revis Bionatura 2022;7(3) 45. http://dx.doi.org/10.21931/RB/2022.07.03.45
