2020.05.03.3
Files > Volume 5 > Vol 5 No 3 2020
INVESTIGATION / RESEARCH
Molecular identification of shiitake [Lentinula edodes Berk (Pegler)] and production of secondary metabolites with biotechnological potential
Byron Durán-Rivera1, Felipe Rojas-Rodas2, Wilber Silva-López3, Crhistian Gómez-Suárez4, Dagoberto Castro-Restrepo5*
Available from: http://dx.doi.org/10.21931/RB/2020.05.03.3
ABSTRACT
The shitake mushroom (Lentinula edodes) is the second most-consumed mushroom in the world; in Colombia, it is cultivated and commercialized on a small scale in some supermarkets. Little is known about the procedence, nutritional and medicinal properties of Shiitake produced in Colombia. In this study, four shiitake isolates grown in Colombia (LEUCO1, LEUCO2, LEUCO3, and LEUCO4) were sequenced in their ITS genes and evaluated for the production of three medicinal metabolites, eritadenine, ergotioneine and β-glucans (1,3-1, 6), using submerged culture. Genetic analysis revealed that all the isolates were close and related to the Japanese strain Cr62. LEUCO1 and LEUCO2 showed a distance of 0.000, as well as LEUCO3 and LEUCO4. All four isolates produced erythadenine in a range of 26.3-8.6 mg / L, with the best performance of LEUCO1 at 26.3 mg / L (p <0.05). Ergotioneine was produced with similar statistical yields in all the isolates with an average of 0.7 mg / g of dry weight biomass (DW). Β-glucans (1.3-1.6) were produced with yields of 5.6 - 3.8% of DW biomass, with the best values for LEUCO2 and the lowest for LEUCO4 (p <0.05). In conclusion, we identified low genetic diversity in the four isolates, corresponding to two haplotypes with minimal genetic difference between them, related to the Japanese strain Cr62, indicating that Colombian farmers cultivate almost the same strains of shitake. Secondary metabolites, eritadenine, β-glucans and ergotioneine were found in promising yields useful for the pharmaceutical and food industries. More studies should be conducted to improve the yield of shitake metabolites through new growing conditions for industrial production and to find metabolic pathways and related genes.
Keywords: Eritadenine, ergothioneine, β-Glucans, ITS genes, Shiitake mushroom.
INTRODUCTION
Gourmet shiitake mushroom has been consumed for more than a thousand years in eastern Asiatic countries and has been studied due to the essential medicinal properties 1. The mushroom is mainly cultivated in China, Korea, and Japan, and currently is the second more consumed mushroom in the world, after the mushroom (Agaricus spp) with 25 % of total mushroom world production 2. Most shiitake research has been focused on the medicinal properties, nutritional qualities, and contents of important bioactive substances beneficial to human health. Some known activities are antitumor, immunostimulatory, antibacterial, hypocholesterolemic, and antioxidant 3. Some of these shiitake substances are proposed to use in pharmaceutical and food industries 4.
In Colombia, shiitake cultivation initiated in the 80´s decade; since then, the mushroom is increasingly becoming more popular in country 5. Shitake is produced in the Andes under tropical weather conditions that are different from the regions where the mushroom is originated. In recent years, the consumption of shiitake has increased. The mushroom is sold in supermarkets as a fresh product and is used for food and medicine. However, little is known about the genetic origin of Colombian shiitake, genetic diversity, and parent strains, as well as the contents of bioactive compounds of shiitakes cultured under the tropical conditions of Colombia. Since the genetic instability has been frequently observed in edible mushrooms 6, environmental conditions might generate genetic and/or biochemical changes and differences in the secondary metabolite profile 7. These could influence the potential of Colombian shiitakes usability in biotechnological processes, considering that the mushroom potential to produce secondary metabolites has been already demonstrated 8.
Different studies have been carried out to establish the genetic characterization of shiitake mushrooms in different countries. Xiao et al. 9 sequenced the whole genome and made a comparison between cultivated and wild strains from China, to evaluate genetic diversity, and George et al. 10 used ITS sequences in shiitake to validate identifications of L. edodes strains based on morphology characters. ITS sequencing is very accepted in scientific literature to determine identity, genetic diversity, intraspecific variation with high confidence as a universal barcode marker for fungi kingdom, and prokaryotes in general 11. In the case of shiitake mushroom, molecular identification is essential because there are some other Lentinula species with similar morphological features, as the edible and tropical American native Lentinula boryana 12. Besides, genetic characteristics are determinant in the production of secondary metabolites 13.
Obtention of secondary metabolites of shiitake under submerged culture has become a critical biotechnological process for pharmaceutical and food uses 4. The purine alkaloid eritadenine [2(R), 3(R)-dihydroxy-4-(9-adenyl] butyric acid), is the primary, secondary metabolite responsible for the hypocholesterolemic action of shiitake, and has been proposed as pharmaceutical ingredient4. HPLC quantitation of eritadenine in carpophores of some shiitake strains has been made by Enman et al14 and Shulei et al15. Additionally, the production of eritadenine by submerged cultivation of shiitake mycelium have been described in different studies with unsatisfactory yields16,17. Recently, new culture techniques have improved the obtention of this metabolite8.
The secondary metabolite ergothioneine (2-mercaptohistidine trimethylbetaine = ERG) is one of the main antioxidant substances in shiitake, with potent antioxidant properties for cells and DNA18, and is antinflamatory19. Different authors evaluated ergothioneine contents in many mushrooms20, 21. Additionally, other authors showed interest in establishing adequate cultivation parameters, to enhance the ergothioneine production, in shiitake mycelium22 and fruitbodies23.
β-Glucans (1,3-1,6) are anticancer substances conformed by glucose polymer chains, bonded in positions 1-3, and shortly ramified every 3 glucose bonded at 1,6 positions; they contribute significantly to the structure of cell walls in shiitake1. Contents of β-glucanes (1,3 – 1,6) have been reported by Nitschke et al24 in mycelium and carpophores of Shiitake, using a simple spectrophotometric Protocole quantitation with Congo red. Also, Bak et al1 quantitated α and β glucans in ten shiitake cultivars in mycelium and fruitbodies, although, the authors do not mention the conditions used to culture the mycelium. Nevertheless, in literature, it was not possible to find comparisons of β-glucanes (1,3 – 1,6) yields between different strains of shiitake, even more under submerged cultivation.
The comparison of yields of eritadenine, ergothioneine and β-glucanes (1,3 – 1,6) in various shiitake isolates, could be useful to select strains in biotechnological production of the metabolites. In this study were identified shiitake isolates cultivated in Colombia by sequencing ITS genes and evaluated the production for the secondary metabolites eritadenine, ergotioneine and β-Glucans (1,3-1,6) under submerged culture.
METHODS
This experiment was carried out at the laboratory of biotechnology, Universidad Católica de Oriente (Rionegro- Colombia), and institute Centro de la Ciencia e Investigación Farmacéutica (CECIF).
Shiitake cultures propagation: Carpophores of shitake were obtained at different local markets, from four different producers. These were used to obtain axenic cultures by extracting internal tissue with totipotent cells. Mycelium was propagated on malt extract agar (MEA) in Petri dishes and incubated at 24 °C by twelve days. The isolates obtained were labeled LEUCO1, LEUCO2, LEUCO3, and LEUCO4, and used for genetic characterization and obtention of secondary metabolites.
Genetic characterization of isolates: Identification of the isolates was carried out according to Sha et al11. DNA was extracted from every insolate using the Invitrogen Purelink Genomic DNA kit. DNA extracted was quantified by light absorption at 260nm (Nanodrop) and visualized in agarose gel. PCR was performed using the primers 5´TCCGTAGGTGAACCTGCGG3´ and 5´TCCTCCGCTTATTGATATGC3´ for ITS1 e ITS4 respectively. Both primers amplify fragments containing around 600 bp. Products of PCR were purified with BSA using the QIAquick PCR purification kit. Thereupon, samples were sequenced by the sanger/capillary method, reading both strands to assure reliability. The sequences were cleaned and ensamble using Cap3 and eBioX 1.5.1 software. Finally, sequences were analyzed through nucleotide BLAST® to determine the isolates taxonomically. Phylogenetic trees and distance matrixes were performed by using MEGA 6.0 software, neighbor-joining, and maximal verisimilitude with 500 replicates of bootstrap were used.
Fermentations: Liquid fermentations were set for all the isolates using the broth described by Enman et al16. Broths were composed of yeast extract (2 g/L), malt extract (20 g/L), and glucose (20 g/L) (Merck, Germany). Fermentations were carried out in 250 mL Erlenmeyer containing 100 ml of broth, inoculated with six 0.5 diameter disks of mycelium, from MEA solid cultures. After that, the flasks were incubated in an orbital shaker at 24°C, 120 rpm for 20 days. The biomass obtained was vacuum filtered with number 4 Whatman filter paper, then oven-dried at 40 °C for two days, and used to quantitate ergothioneine and B-glucans (1,3-1,6) in two separated portions. The fermentation broth was used to quantify the erititadenine.
Eritadenine quantitation. Eritadenine was quantitated using the HPLC method described by Enman et al16. An Agillent Technologies 1200 Series apparatus, equipped with a c18 column, was used. The mobile phase was composed of a gradient of acetonitrile, changing from 2% to 60% during the first 10 minutes and 0.1% trifluoroacetic acid (TFA) from 60% to 2%, from 10 to 11 minutes (Merck, Germany). The temperature was set at 23°C, with a detection wavelength at 260nm. The quantifications were determined using a calibration curve with eritadenine standard, as reference (Santa Cruz Biotechnology Inc.).
Ergothioneine quantitation. Extraction and quantitation of ergothioneine were done according to Dubost et al25 with few modifications. The dry mycelium was ground to a fine powder with a mortar and pestle, then MEOH was added in a proportion of 1:5 (w/v), and incubated during 24 h at room temperature (18°C average) in darkness. The mixture was filtered, and the clear methanol extract was used directly for HPLC quantification. Samples were injected through a c18 column (4.6mm x 150 mm, Merck) mounted on an Agilent series 1200 apparatus, at 23°C, 1.0 mL/min with a MeCN gradient, starting with 2% during the first 5 min, changed to 80% from 5.1 to 10 min, then finally set again to 2% from 10 to 20 min. Values obtained were compared against a calibration curve constructed with the authentic standard substance (Santa Cruz Biotechnology Inc).
β-Glucans (1,3-1,6) quantitation. β-Glucans were analyzed in all the isolates by three successive extractions in the same sample, according to the procedure of Nitschke et al24. Extractions were as follows: 1) KOH fraction was obtained by extracting the pulverized-dried mycelium with 1M KOH, in a proportion of 40:1 (w/v) with stirring, during 20 min at 60°C. The extract was cooled down, filtered, neutralized with 6M HCl to pH=7.0, and adjusted to 100 ml with distilled water; 2) HCl fraction was obtained using the filtered cake from the last extract, suspended in 0.55 M HCl, and heated at 100°C for 1h. The extract was filtered and the filtrate neutralized with 6M NaOH to pH=7.0 and adjusted to 100 mL with distilled water; 3) NaOH fraction, was obtained from filter cake, extracted in 1M NaOH at 60°C for 20 min, then was cooled down, filtered, neutralized with 6M HCl to pH=7.0, and adjusted to 100 mL with dH2O.
The quantification of β-Glucans (1,3 →1,6) was performed using the spectrophotometric method with congo red, developed by Nitschke et al24. Into the spectrophotometric (spectroquant Pharo 400, Merck, Germany) cuvette, were put 700 µL of fungal extract, 700 µL of Citric acid Buffer (0,2M Citric acid/NaOH, pH=7, Merck, Germany), and 100 µL of Congo red solution (0.08 g in the citric acid buffer, Merck Germany). Absorbance was read at 553 nm, and the β-Glucans (1,3-1,6) concentration was determined using a calibration curve constructed with a series of solutions of standard schizophyllan (InvivoGen, USA). β-Glucans (1,3-1,6) found in the three extracts were added and expressed as total β-Glucans (1,3-1,6) in % of the dry weight of mycelium.
Statistics: every metabolite was studied by random experimental design with four treatments (isolates) by triplicate, then data were analyzed by ANOVA (Tukey test), or Kruskal-Wallis (Dunn posthoc test) with R studio 4.0.2 of 2020.
RESULTS AND DISCUSSION
Genetic characterization: we could establish the identity of four shiitake mushrooms of unknown origin cultivated in Colombia by sequencing two ITS genes. PCR products from the isolates were purified, visualized with electrophoresis and sequenced for ITS1 and ITS4 genes (Figure 1a). The sequences obtained were 649 bp long in all four isolates, identified as Lentinula edodes species, and were compared with the elite strain Cr62 from genebank (https://www.ncbi.nlm.nih.gov/genbank/). Genetic distance values were generated along with a Neighbor-Joining phylogenetic tree (Figure 1, B and C).
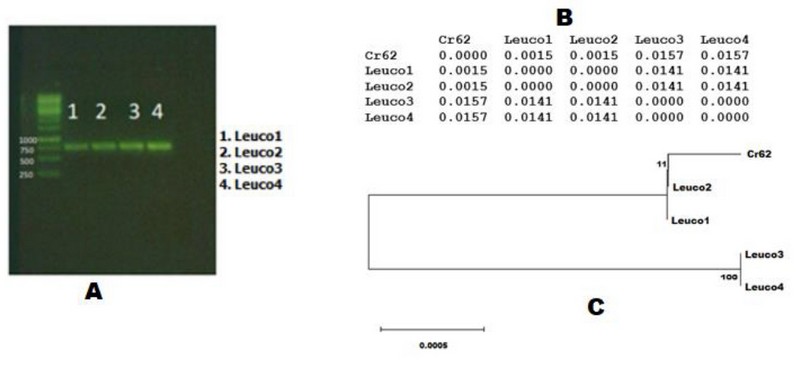
Figure 1. Genetic characterization to the isolates. A: purified products of PCR; B: Distances matrix between the isolates, and C: Phylogenetic tree for all four isolates together with Cr62 elite shiitake strain.
ITS sequences revealed low differences between isolates (matrix fig. 1 B). The sequences were gathered in two homolog groups (haplotypes) 1) LEUCO1-LEUCO2 and 2) LEUCO3-LEUCO4, with 0.000 distance in between, close to the elite strain Cr62. Distances between Cr62 and the isolates were equally low. The first group had a difference of 0. 15% with the strain Cr62, with one base substitution and one deletion. The second had a difference of 1.57%, nine bases substitution in a different order, and two deletions. Contradictorily, Sha et al11, found a variation of up to 138 bases in wild samples of the gourmet mushroom Thelephora ganbajun from china. These resulting distances confirm a low genetic diversity in the evaluated isolates. Additionally, Nilsson et al26 found from an exhaustive reviewing of sequences covering the fungi kingdom, an average intraspecific variability of 2.51% based on ITS sequences. Accordingly, Xiao et al9 found a reduced genetic diversity in Chinese shiitake cultivars compared to wild strains, probably caused by human selection and breeding.
Eritadenine assay: All four isolates produced eritadenine (8.6 - 26.3 mg/L). Best yields were attained by LEUCO1 (26.3 mg/L), with statistical differences with the rest of isolates (Table 1). Here, yields were higher than those published by other authors16,17 with maximal yields of 8.6 and 25 mg/L, using broths of complex nutritional composition, and lower than 88mg/L obtained in other experiments with alginate inmobilization8.
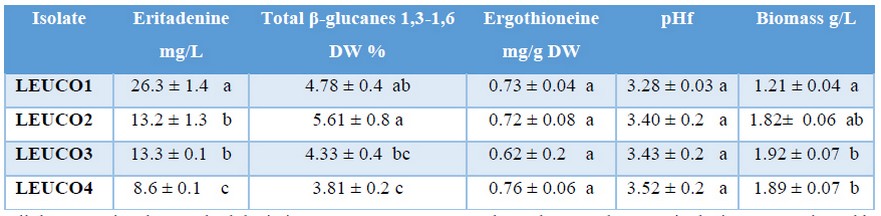
Table 1. Yields of metabolites on average after 20 days of submerged fermentation with shiitake mycelium.
Few reports exist in the literature related to the obtention of eritadenine by different shiitake strains in submerged culture. Enman et al14, found in carpophores of four shitake strains maximal yield of 6.33 mg/g, with no significant statistical differences, also Shulei et al15 reported 1.99 mg/g in carpophores, without mentioning the strain. Both authors did not report the methods used to produce the carpophores, then possibly, the differences were not only due to genetic differences but also to cultivation conditions, as samples were obtained from different producers. The latter probably used different environmental and nutritional conditions.
Here we have three groups of shiitake isolates (LEUCO1, LEUCO4, and LEUCO2-LEUCO3) that express eritadenine genes at different levels. Seemingly those differences in eritadenine expression do not have a clear relationship with the distances obtained (Fig 1), which could be a logical assumption as ITS genes do not encode for enzymes, then do not participate in eritadenine biosynthesis pathway. Accordingly, Schwessinger et al27 found gen expression variation of 6% in Puccinia striiformis, which occur in dikaryotic isolates (diploids) with sexual reproduction, as the shiitake isolates evaluated in the present research. Also, the production of purine precursors to eritadenine (AICAR and SAICAR) from the inosine monophosphate (IMP) pathway, are expressed in different levels by different strains of yeast Saccharomyces cereviciae, and those genes are activated in yeast by adding adenine to the nutritional medium28. As eritadenine pathway is not understood17, future research studies should be directed to identify genes related to eritadenine biosynthesis27.
β-glucans (1,3 – 1,6): All four isolates produced β-glucans in a range from 3,8% to 5,6 % of DW of mycelium. LEUCO2 produced the higher yields, without statistical differences with LEUCO1. On the other hand, LEUCO4 produced the lowest yields, without statistical differences with LEUCO3 (Table 1). LEUCO2 produced slightly more than twice β-glucans (1,3 – 1,6) the yields reported by Nitschke et al24. This author obtained 2,58 g/100 in shiitake mycelium, using the same extraction and quantitation protocols than this experiment. Nonetheless, the strain name was not reported, and the cultivation conditions were broths of yeast and malt extracts, at 80 rpm (different than used in this experiment).
Likewise, other studies reported differences in β-glucans contents in different cultivars of shiitake mushroom1 and in Agaricus subrufescens29. These differences in β-glucans (1,3 – 1,6) yields were probably due to the homeostasis between β-(1,3)-glucan synthase, the primary enzyme responsible for fungal cell wall construction30. And the β-glucans destructive counterparts β-(1,3)-glucanases31. In shiitake, various glucanases coding genes have been identified, which are upregulated in postharvest shiitake fruiting, leading to the autolysis of cell wall32,33.
In Lentinula edodes composition of the cell wall is well known, mainly consisting of polysaccharides like chitin and β-glucanes2. Those are synthesized by the enzyme β-(1,3)-glucan synthases that have a catalytic subunit FKS, plus a regulatory one RHO, both encoded by FKS and RHO genes34. However, there is inadequate knowledge about the transcriptional regulation of this enzymatic biosinthesis34. We hypothesize that nutrition and environmental conditions used in this experiment, especially the glucose concentration of the broth, produced a positive balance in favor of β-(1,3)-glucan synthase activity in LEUCO2 (the more productive isolate), but not for LEUCO4.
Ergothioneine assay. All isolates produced similar ergothioneine yields, ranging from 0,62 to 0,75 mg/g DW of mycelium, with an average of 0.7 mg/g, to all the isolates. These concentrations did not show significant statistical differences between isolates but were higher than the concentrations reported by Tepwong et al22, who found 0.5 mg/g for Shiitake mycelium pellets in submerged culture. And seventeen times higher than the yields reported by Jang et al23 who used shiitake submerged cultures supplemented with various nitrogen sources. Unexpectedly, the isolates used in this experiment contained eight times more ergothioneine than the fruiting body of the popular Ganoderma lucidum21.
Few reports in literature compare ergothioneine content in different strains of edible mushrooms related to Shiitake. Contrary to this research, Chen et al20 found different ergothioneine concentrations in the mycelium of three strains of Cordyceps militaris, and in three commercial carpophores of Pleurotus ostreatus from China, Korea, and Japan. However, the authors do not report cultivation methods to obtain carpophores or mycelium; then, it is possible to speculate that differences are related to the possible different cultivation conditions. Hence, to obtain accurate comparisons, fungal cultures for chemotaxonomic analysis should be grown in the same medium and incubation conditions, to ensure that differences correspond to fungal genetic diversity and not to environmental conditions35.
Nitrogen source is critical for ergothioneine biosynthesis, amino acids like cysteine, methionine, and histidine are precursors of the substance36. By adding methionine to liquid broths, ergothioneine yields are increased up to twice, compared to fermentation without the addition of aminoacids37. This is due to histidine is a precursor of ergothioneine22,37. Then the low ergothioneine yields obtained in this experiment were probably due to the lack of precursor amino acids in the yeast extract used. For instance, Lin et al38 found in submerged cultures of Pleurotus citrinopileatus a higher ergothioneine yield 10.65mg/g, by using tryptone instead of yeast extract. Probably tryptone contains 1.6% of methionine, while yeast extracts only 0.8%. In this sense, we hypothesized that the use of nitrogen sources rich in the amino acids mentioned above might improve ergothioneine yields produced in the shiitake isolates of this experiment.
Biomass production and final pH obtained in fermentations. All four isolates produced biomass after twenty days of fermentation in a range from 1.21 to 1.92 g/L. LEUCO2, LEUCO3, and LEUCO4 produced the higher biomass (1.82, 1.92, and 1.89 g/L respectively) without significant statistical differences between them. At the same time, LEUCO1 produced the lowest value (1.21g/L), statistically similar to LEUCO2. Accordingly, shiitake strains show different utilization of nutritional components in liquid broths, which affects biomass yields. This was demonstrated by López-Peña et al39, who found two high biomass producing strains 3.73 g/L and 8.79 g/L, from six evaluated. In this sense, the differences between the isolates could be attributed to incubation temperature (24°C). Probably this temperature was not optimal for the isolate LEUCO1 and LEUCO2. According to Lee et al40, every strain has an optimum growth temperature. For instance, Quaicoe et al41 found different radial growth in three shiitake strains at 20°C, cultured in potato dextrose agar (PDA).
On the other hand, final pH had statistically similar values in fermentations of all isolates, ranging from 3.28 to 3.52. Different authors found similar pH at the end of fermentations, Enman et al17, obtained a final pH of 3.5-3.6 for shiitake under submerged culture, with eritadenine yields of 25 mg/L. The same authors established that optimal pH for eritadenine production could be in a range from 3 to 4. Likewise, Quaiqoe et al41 established a pH range from 3 to 4 as optimal for shiitake mycelium growth at in vitro culture. According to the above authors, our four shiitake isolates reduced the pH to the expected values of optimal growth. Nonetheless, there is not enough evidence about the effect of pH on eritadenine, ergothioneine, and β-glucans yields. Even more, for eritadenine production with shiitake, variables that produce good biomass growth are not necessarily the same to produce high metabolite yields17.
Finally, after 20 days of fermentation, all isolates produced biomass in the form of well-defined pellets. The 120-rpm agitation promotes this growth morphology. Similarly, other studies obtained pellets of shiitake mycelium under submerged culture, at the same agitation values [42,37]. Growth morphology insides on metabolites production, shiitake pellets produce higher yields of ergothioneine compared to cultures with no pellets formation.
CONCLUSION.
In this investigation, we identified low genetic diversity in the four shiitake isolates examined, which corresponds to the most commonly produced cultivars in colombia. These findings allow us to conclude that the four isolates formed two haplotypes LEUCO1-LEUCO2 and LEUCO3-LEUCO4 with little genetic difference between them, but related to the elite Japanese strain Cr62. All strains produced the pharmaceutical metabolites eritadenine in a promising concentration for industrial use: β-glucans (1-3, 1-6) and ergothioneine. LEUCO1 was more suitable for the production of eritadenine, while LEUCO1, 2, and 3 were more suitable for producing β-glucans. Furthermore, all four isolates produced the same amounts of ergotioneine. Finally, biomass was better produced by LEUCO2, 3 and 4. Further research could be aimed at improving shiitake metabolite yields and optimizing culture and incubation systems, for industrial production, and finding metabolic pathways and genes related.
ACKNOWLEDGMENTS.
Authors want to be thankful to investigación & Desarrollo at Universidad Catolica de Oiriente, to CECIF (Centro de la ciencia y la investigación farmacéutica). Both institutions suported entusiastically the research, and to Dr. Carlos Eduardo Giraldo Sánchez who colaborated us with the genes sequences analysis.
REFERENCES
1. Bak WC, Park JH, Park YA, Ka KH (2014) Determination of Glucan Contents in the Fruiting Bodies and Mycelia of Lentinula edodes Cultivars. Mycobiology 42(3): 301-304. doi: 10.5941/MYCO.2014.42.3.301
2. Jiang T, Wang Q, Xu S, Jahangir MM, Ying T (2010) Structure and composition changes in the cell wall in relation to texture of shiitake mushrooms (Lentinula edodes) stored in modified atmosphere packaging. J Sci Food Agric. 90:742–749. https://doi.org/10.1002/jsfa.3876
3. Bisen PS, Baghel RK, Sanodiya BS, Thakur GS, Prasad GBKS (2010) Lentinus edodes: A Macrofungus with Pharmacological Activities. Curr Med Chem. 17: 2419-2430. https://www.ncbi.nlm.nih.gov/pubmed/20491636
4. Enman J, Patra A, Ramser K, Rova U, Berglund KA (2011) Solid State Characterization of Sodium Eritadenate. Am. J. Anal. Chem 2:164-173. DOI: 10.4236/ajac.2011.22019
5. Romero-Arenas O, Martínez GM, Damián MA, Ramírez B, López-Olguín F (2015) Producción del hongo Shiitake (Lentinula edodes Pegler) en bloques sintéticos utilizando residuos agroforestales. Rev. Mex. Cienc. Agríc 6(6): 1229-1238. http://www.scielo.org.mx/pdf/remexca/v6n6/v6n6a7.pdf
6. Mallick P, Sikdar SR (2015) Genome instability in fruit body derived lines generated from fruiting pfle somatic hybrid lines and development of hybrid strain specific scar marker in edible mushroom. J. Hort. Res 23(2): 111-120. DOI: 10.2478/johr-2015-0022
7. Torres S, cabrera-pardo J, Alonso f, Bustos E, Pérez C, Palfner G, Hernández V, Uriarte E, Becerra J (2016) Changes in secondary metabolites profiles and biological activity of the fresh fruiting bodies of stereum hirsutum exposed to high-dose uv-b radiation. J. Chil. Chem. Soc 61(4): 3224-3227. http://dx.doi.org/10.4067/S0717-97072016000400015
8. Duran-Rivera B, Moreno-Suarez J, Rojas RF, Valencia KM, Castro-Restrepo D (2018) Enhancement of eritadenine production using three carbon sources, immobilization and surfactants in submerged culture with shiitake mushroom (Lentinula edodes) (Berk.) Singer). Afr. J. Food Sci 12(12): 374-382. DOI: 10.5897/AJFS2017.1654
9. Xiao Y, Cheng X, Liu J, Li C, Nong W, Bian Y, Kit MC, Shan HK (2016) Population genomic analysis uncovers environmental stressdriven selection and adaptation of Lentinula edodes population in China. Sci. Rep. 6(36789):1-12. https://doi.org/10.1038/srep36789
10. George PL, Sripathi VR, Nyaku ST, Sharma GC, Kantety RV (2016) DNA-based identification of Lentinula edodes strains with species-specific primers. Afr. J. Biotechnol 15(7):191-198. DOI: 10.5897/AJB2015.15089
11. Sha T, Xu J, Palanichamy GM, Zhang H-B, Li T, Zhao Z-W, Zhang Y-P (2008) Genetic diversity of the endemic gourmet mushroom Thelephora ganbajun from southwestern China. Microbiology 154: 3460–3468. DOI: 10.1099/mic.0.2008/020495-0
12. Gaitán-Hernández R, Salmones D (2015) Uso de residuos lignocelulósicos para optimizar la producción de inóculo y la formación de carpóforos del hongo comestible Lentinula boryana. Rev. Mex. Cienc. Agríc 6(7):1639-1652. DOI: 10.29312/remexca.v6i7.556
13. Pandit SS, Lohmar JM, Ahmed S, Etxebeste O, Espeso EA, Calvo AM (2018) UrdA Controls Secondary Metabolite Production and the Balance between Asexual and Sexual Development in Aspergillus nidulans. Genes 9(570): 1-23. DOI: 10.3390/genes9120570
14. Enman J, Rova U, Berglund K (2007) Quantification of the Bioactive Compound Eritadenine in Selected Strains of Shiitake Mushroom (Lentinus edodes). J Agric Food Chem 55: 1177-1180. DOI:10.1021/jf062559+
15. Shulei W, Jingyu L, Qingjiu T, Yangfang L, Shuai Z, Yan Y, Jingsong Z (2011) Determination of Eritadenine in Lentinula edodes Fruit bodies Using HPLC. Acta edulis fungi 18(2): 52-56. http://en.cnki.com.cn/Article_en/CJFDTOTAL-SYJB201102014.htm
16. Enman J, Hodge D, Berglund KA, Rova U (2008) Production of the Bioactive compound Eritadenine by Submerged Cultivation of Shiitake (Lentinus edodes) Mycelia. J Agric Food Chem 56: 2609–2612. DOI:10.1021/jf800091a
17. Enman J, Hodge D, Berglund KA, Rova U (2012) Growth promotive conditions for enhanced eritadenine production during submerged cultivation of Lentinus edodes. J. Chem. Technol. Biotechnol 87(7): 903–907. https://doi.org/10.1002/jctb.3697
18. C¸oyan K, Numan BM, Baspinar N, Taspinar M, Aydos S (2012) Ergothioneine attenuates the DNA damage of post-thawed Merino ram sperm. Small Ruminant Res 106:165–167. https://doi.org/10.1016/j.smallrumres.2012.02.002
19. Laurenza I, Colognato R, Migliore L, Del Prato S, Benzi L (2008) Modulation of palmitic acid-induced cell death by ergothioneine: evidence of an anti-inflammatory action. Biofactors 33:237-47. https://doi.org/10.1002/biof.5520330401
20. Chen SY, Ho KJ, Hsieh YJ, Wang LT, Mau JL (2012) Contents of lovastatin, g-aminobutyric acid and ergothioneine in mushroom fruiting bodies and mycelia. LWT-Food Sci Technol 47: 274-278. https://doi.org/10.1016/j.lwt.2012.01.019
21. Lee WY, Park E-J, Ahn JK, Ka K-J (2009) Ergothioneine Contents in Fruiting Bodies and Their Enhancement in Mycelial Cultures by the Addition of Methionine. Mycobiology 37(1): 43-47. doi: 10.4489/MYCO.2009.37.1.043
22. Tepwong P, Giri A, Sasaki F, Fukui R, Ohshima T (2012a) Mycobial enhancement of ergothioneine by submerged cultivation of edible mushroom mycelia and its application as an antioxidative compound. Food Chem 131: 247–258. https://doi.org/10.1016/j.foodchem.2011.08.070
23. Jang Y, Park J, Ryoo R, Park Y, Ka K-H (2016) Ergothioneine Contents of Shiitake (Lentinula edodes) Fruiting Bodies on Sawdust Media with Different Nitrogen Sources. Kor. J. Mycol 44(2): 100-102. DOI: http://dx.doi.org/10.4489/KJM.2016.44.2.100
24. Nitschke J, Modick H, Busch E, von Rekowski RW, Altenbach H-J, Mölleken H (2011) A new colorimetric method to quantify b-1,3-1,6-glucans in comparison with total b-1,3-glucans in edible mushrooms b-1,3-glucans in edible mushrooms. Food Chem 127:791–796. DOI: 10.1016/j.foodchem.2010.12.149
25. Dubost NJ, Beelman RB, Royse DJ (2007) Influence of selected cultural factors and postharvest storage on ergothioneine content of common button mushroom Agaricus bisporus (J. Lge) Imbach (Agaricomycetideae). Int. J. Med. Mushrooms 9:163–176. DOI: 10.1615/IntJMedMushr.v9.i2.70
26. Nilsson RH, Kristiansson E, Ryberg M, Hallenberg N, Larsson KH (2008) Intraspecific ITS Variability in the Kingdom Fungi as Expressed in the International Sequence Databases and Its Implications for Molecular Species Identification. Evol Bioinform 4:193–201. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2614188/
27. Schwessinger B, Sperschneider J, Cuddy W, Garnica D, Miller M, Taylor J, Dodds P, Figueroa M, Parck R, Rathjen J (2018) A Near Complete Haplotype-Phased Genome Of The Dikariotic Wheat Stripe Rust Fungus Puccinia striiformis f. sp. tritici Reveals High Interhaplotype Diversity. Mbio 9(1): e02275-17. DOI: 10.1128/mBio.02275-17
28. Rebora K, Laloo B, Daignan-Fornier B (2006) Revisiting Purine-Histidine Cross-Pathway Regulation in Saccharomyces cerevisiae: A Central Role for a Small Molecule. Genetics 170: 61–70. DOI:10.1534/genetics.104.039396
29. Clemente TRC, Andrade CM, Oliveira LLC, Barros V-BV, de Souza DE (2013) Measurement of ß-glucan and other nutritional characteristics in distinct strains of Agaricus subrufescens mushrooms. African journal of Biotechnology. 12(43): 6203-6209. http://dx.doi.org/10.5897/AJB2013.13024
30. Orlean P (2012) Architecture and Biosynthesis of the Saccharomyces cerevisiae Cell Wall. Genetics 192(3): 775–818. DOI: 10.1534/genetics.112.144485
31. Bauermeister A, Giese E-C, Borsato D, Rezende M-I, Dekker F-H, Barbosa A-M (2017) Filamentous Fungal Biomass as Inducer of Extracellular Β-1,3-Glucanases and Β-Glucosidases by Aureobasidium Pullulans 1WA1”. ECMi 5(4): 138-147. https://www.ecronicon.com/ecmi/pdf/ECMI-05-0000142.pdf
32. Sakamoto Y, Nakade K, Sato S, Yoshida K, Miyazaki K, Natsume S, Konno N (2017) Lentinula edodes Genome Survey and Postharvest Transcriptome Analysis. Appl Environ Microbiol 83(10): e02990-16. DOI: 10.1128/AEM.02990-16
33. Konno N, Nakade K, Nishitani Y, Mizuno M, Sakamot Y (2014) Lentinan Degradation in the Lentinula edodes Fruiting Body during Postharvest Preservation Is Reduced by Downregulation of the exoβ-1,3-Glucanase EXG2. J Agric Food Chem 62: 8153−8157. DOI: dx.doi.org/10.1021/jf501578w.
34. Chai R, Qiu C, Liu D, Qi Y, Gao Y, et al (2013) β-Glucan Synthase Gene Overexpression and β-Glucans Overproduction in Pleurotus ostreatus Using Promoter Swapping. PLoS ONE 8(4): e61693. https://doi.org/10.1371/journal.pone.0061693
35. Frisvad JC, Andersen B, Thrane U (2008) The use of secondary metabolite profiling in chemotaxonomy of filamentous fungi. Mycol Res. 112: 231–240. DOI:10.1016/j.mycres.2007.08.018
36. Pluskal T, Ueno M, Yanagida M (2014) Genetic and Metabolomic Dissection of the Ergothioneine and Selenoneine Biosynthetic Pathway in the Fission Yeast, S. pombe, and construction of an Overproduction System. PLOS ONE 9(5): 1-12. DOI: 10.1371/journal.pone.0097774
37. Tepwong P, Giri A, Ohshima T (2012b). Effect of mycelial morphology on ergothioneine production during liquid fermentation of Lentinula edodes. Mycoscience 53:102–112. https://doi.org/10.1007/S10267-011-0145-0
38. Lin S-Y, Chien S-C, Wang S-Y, Mau J-L (2015) Submerged Cultivation of Mycelium with High Ergothioneine Content from the Culinary- medicinal Golden Oyster Mushroom, Pleurotus citrinopileatus (Higher Basidiomycetes). Int J Med Mushrooms 17(8): 749–761. DOI: 10.1615/IntJMedMushrooms.v17.i8.50
39. López-Peña D, Gutiérrez A, Esqueda M (2013) Cinética de crecimiento y composición química del micelio de Lentinula edodes cultivado en medio líquido suplementado con extractos de madera de vid. Rev. Mex. Mic 37: 51-59. http://www.scielo.org.mx/pdf/rmm/v37/v37a7.pdf
40. Lee S, Bae H, Kim N, Hwang S (2008) Optimization of Growth Conditions of Lentinus edodes Mycelium on Corn Processing Waste Using Response Surface Analysis. J Biosci Bioeng 105(2): 161-163. DOI: 10.1263/jbb.105.161
41. Quaicoe EH, Amoah C, Obodai M, Odamtten GT (2014) Nutrient requirements and environmental conditions for the cultivation of the medicinal mushroom (Lentinula Edodes) (Berk.) in Ghana. IJSTR 3(12): 45-50. http://www.ijstr.org/final-print/dec2014/Nutrient-Requirements-And-Environmental-Conditions-For-The-Cultivation-Of-The-Medicinal-Mushroom-lentinula-Edodes-berk-In-Ghana.pdf
42. Ting LTC, Tsao HH, Wang AY, Chang CA (2007) Pressurized Water Extraction of Polysaccharides as Secondary Metabolites from Lentinula edodes. J Agric Food Chem 55: 4196-4201. DOI: 10.1021/jf070035j
Received: 7 July 2020
Accepted: 7 agost 2020
Byron Durán-Rivera1, Felipe Rojas-Rodas2, Wilber Silva-López3, Crhistian Gómez-Suárez4, Dagoberto Castro-Restrepo5 *
1. Universidad Catolica de Oriente, Unidad de Biotecnología Vegetal, Rionegro, Antioquia, Colombia, postal code 054040 https://orcid.org/0000-0002 - 4592-6873
2. Universidad Catolica de Oriente, Unidad de Biotecnología Vegetal, Rionegro, Antioquia, Colombia, postal code 054040 (https://orcid.org/0000-0003-3959-5761)
3. Centro de Ciencia Básica, Grupo de óptica y Espectroscopía, Universidad Pontificia Bolivariana, Circular primera N° 70 – 01- Campus Laureles 050031. (https://orcid.org/0000-0003-4536-8565)
4. CECIF, Centro de la Ciencia y la Investigación Farmacéutica, Sabaneta, Antioquia, Colombia, postal code 055450
5. Universidad Catolica de Oriente, Unidad de Biotecnología Vegetal, Rionegro, Antioquia, Colombia, postal code 054040 (https://orcid.org/0000-0002-6599-9332)
* Corresponding author: Investigació[email protected] ; cel. 3136524137
