2021.06.02.22
Files > Volume 6 > Vol 6 No 4 2021 > Vol 6 No 2 2021
Behavior of mean platelet volume in critical care patients with and without sepsis.

Pablo Andrés Vélez 1. Lucy Baldeón R 2,3 Jorge Luis Vélez-Paez1,2.
Available from: http://dx.doi.org/10.21931/RB/2021.06.02.22
ABSTRACT
The mean platelet volume is an anatomical biomarker that has shown its usefulness in various cardiovascular and metabolic pathologies; in sepsis, it has been positioning itself as an indicator of mortality, easily accessible and immediately applicable when reported in the routine blood count. This study demonstrates the mean platelet volume's biological behavior in critical patients with sepsis compared with non-septic patients. An observational, longitudinal, prospective, monocentric cohort study was conducted in 250 patients treated at the intensive care unit of the Pablo Arturo Suárez Hospital, Quito- Ecuador, from January 2019 January 2020. A group of patients with sepsis (n = 125) and without infectious pathologies (n = 125) were studied. The inclusion criteria were patients over 18 years of age of both genders, diagnosed with sepsis or septic shock using SEPSIS 3 criteria, and patients without septic pathology. The mean platelet volume (MPV) of days 1, 2, and 3 were studied. Septic patients had a mean APACHE (18.74 SD 9.52) higher than the non-septic ones (11.93 SD 7.01) (p = < 0.000). The MPV was consistently higher in patients with sepsis than non-septic patients, but it reached statistical significance on day 3 (9.13 SD 1.55 vs. 8.66 SD 1.34, p=0.042). The MPV on day 3 presented a significant area under the curve (AUC =0.580) (CI. 0.500-0.661), where the cut-off point according to Youden's index was positive for sepsis if MPV≥ 9.85 femtoliter (fL) with OR=3.30 and p-value= 0.005. Likewise, lactate on admission showed an AUC of 0.625 (CI. 0.555-0.694), with a cut-off point ≥of 1.15 mmol / L, OR=2.51, and p=0.007. Age and hypertension did not show a multivariate relationship with the presence of sepsis. It was shown that MPV is higher in patients with sepsis compared to non-septic ones. This observation reaches significance on day 3. Additionally, elevated lactate at admission was also associated with a septic state. On the other hand, platelet count did not show the expected behavior.
Keywords. Mean Platelet Volume, sepsis, mortality.
INTRODUCTION
Sepsis is defined as the unregulated response of the body to an infectious disease. Nowadays, this pathology is one of the main reasons for admission to the intensive care unit. There are still few strategies described to face it; however, it is known that an early antibiotic therapy, together with the adequate support of organ failure, are measures that reduce its pathophysiological impact and, therefore, its mortality 1.
Various biochemical markers (procalcitonin, C-reactive protein, and interleukin 6) and prognostic scales such as APACHE II and SOFA can determine this entity's severity. However, they are not always available in the daily clinical requirement, or their high economic value does not allow their routine use in low-income countries. Therefore, a new biomarker, such as the mean platelet volume (MPV), could become a practical and easily accessible tool to predict septic states.
Platelets are cell fragments from megakaryocytes, which are essential for blood clotting. The MPV is the measurement of platelet size and has an inverse relationship with the number of these; it is also an indicator of platelet activation 2, 3.
There is no fixed cut-off point for MPV because the values change and vary from population to population. They also vary according to the technique used to measure it (impedance or laser scattering), the type of anticoagulant used (EDTA or citrate), and the test time. However, it is established that values greater than 9.5 femtoliters (fL) are correlated with diseases related to inflammation, endothelial dysfunction, and pro-thrombotic states 1. Additionally, increased MPV has been reported to be associated with poor prognosis in patients with metabolic syndrome, diabetes mellitus, cardiovascular diseases, pulmonary embolism, smoking, and immuno-inflammatory diseases 2, 4, 5.
Van der Lelie et al. in 1983 reported that MPV increases in invasive infectious septic disease, but not in local infection and that this biomarker normalizes when controlling the disease 1. There are no studies that demonstrate that platelet size is higher in sepsis compared to other critical pathologies. Therefore, this study's objective is to demonstrate that mean platelet volume (MPV) is higher in critical patients with sepsis than non-septic patients.
MATERIALS AND METHODS
An observational, longitudinal, prospective, monocentric cohort study was conducted. The sample consisted of 250 patients treated in the intensive care unit of the Pablo Arturo Suárez Hospital in Quito, Ecuador, in the period January 2019 - January 2020. The sample consisted of two groups, patients with sepsis (n = 125) and patients with other non-infectious pathologies (n = 125). The inclusion criteria were patients of both genders, older than 18 years with a diagnosis of sepsis or septic shock who met the criteria of SEPSIS 3 [6] and patients with non-septic pathology such as preeclampsia, eclampsia, HELLP syndrome, acute pancreatitis, or cardiovascular diseases. Patients with neoplasms or acute bleeding states that can alter MPV were excluded. Patient data were coded, and the results of the routine tests requested were documented in the clinical history for the required variables (lactate, MPV, leukocyte count, platelet count, and procalcitonin) on days 1, 2, 3. The difference in MPV between patients with and without sepsis and the value that determined the progression to septic states was evaluated. The MPV was measured in a femtoliter by impedance in a Siemens machine, in venous blood collected in EDTA tubes. The data was coded and entered into a database, to which only the researcher has access. Analyzes were carried out with R and SPSS version 22 software. Qualitative variables were reported using tables and graphs representing absolute and relative values. Quantitative variables were represented as measures of central tendency and variability.
Inferential statistics were performed to compare the clinical characteristics and laboratory parameters between the sepsis and non-sepsis groups (bivariate analyzes). For categorical variables, the chi-square test was applied to compare proportions. For the analysis of quantitative variables, the assumption of normality was first verified. Thus, for variables that presented normality, a t-test was used. For the variables that do not meet the normality criteria, Mann Whitney test was used. Using the ROC Curve, the cut-off points for MPV and lactate significant in the bivariate analysis were determined, which were expressed as area under the curve (AUC). Besides, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and Odds Ratio (OR) were determined for each cut-off point. A multivariate logistic regression analysis was performed to determine the relationship of sepsis using the OR. Statistical significance was established for the value of p <0.05; the OR was considered significant, observing the limits of the 95% confidence interval. It was considered a risk factor if the lower limit was more significant than one.
ETHICAL CONSIDERATIONS
Researchers have followed the bioethical principles of human research. The data were obtained from medical records (secondary data), the identification of patients will not be disclosed, and they have been recorded to avoid their recognition. The authors obtained the authorization for publication.
RESULTS
The average age of patients with sepsis was 60.73 years, and 45.21 years of patients without sepsis. Differences in age between groups showed significance (p=0.000). In gender, no significant differences were observed; the predominant ethnic group was mestizo in sepsis and the non-infectious group (93.60% and 95.20%, respectively), without significant differences. Significant differences were observed for APACHE (p= 0.000), in patients with sepsis (18.74 points) and without sepsis (11.93 points). High blood pressure (HBP) presented significant differences between the groups (p= 0.011). The percentage of non-survivors was 26.40% in the sepsis group and 5.60% in the non-sepsis group, showing significance between groups (p= 0.000). The patients who required mechanical ventilation were 76.80% in the group of patients with sepsis and 52.80% in the patients without sepsis, showing significant differences (p=0.000). Patients who required sedation were 64.23% in the group sepsis and 35.54% in the group without sepsis, showing significant differences (p=0.000). The average days of hospitalization were 6.96 days in the sepsis group compared with 4.12 days without the sepsis group (p= 0.000) (see table 1).
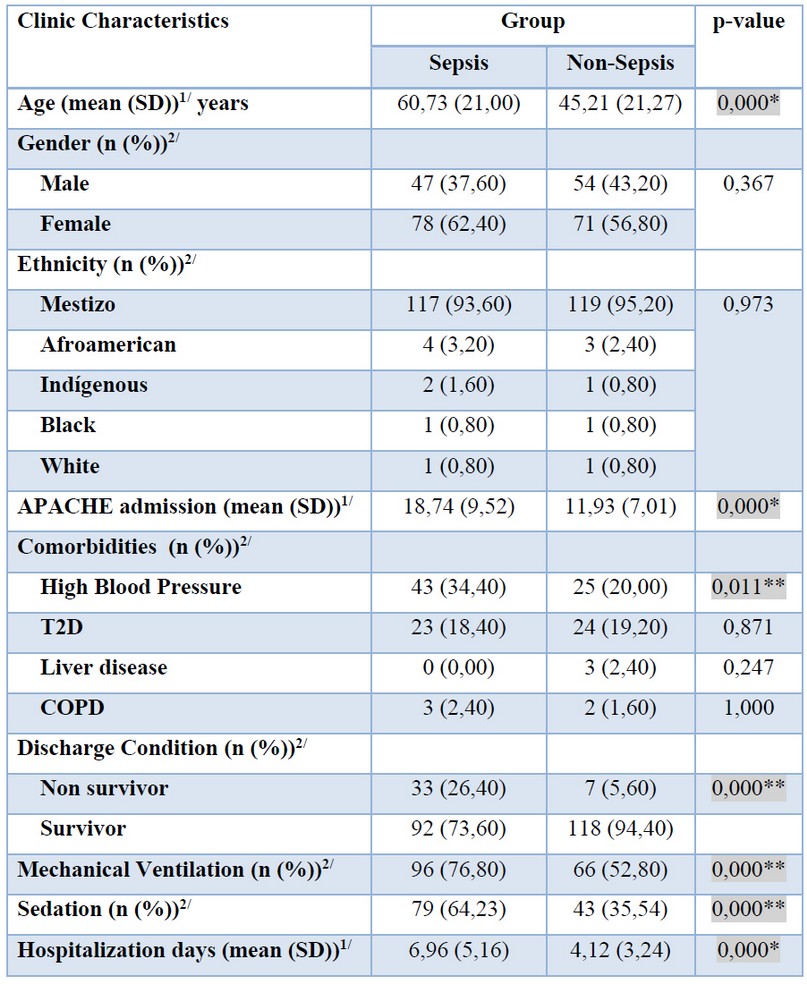
Table 1. Distribution of demographic and clinical characteristics by sepsis and non-sepsis group.
When comparing laboratory parameters such as MPV in the sepsis (3 9.13 fL) and non-sepsis (8.66 fL) groups, significant differences were observed on day 3 (p= 0.042) and at discharge (p=0.035). Differences were also observed in leukocyte values on day 1 (p=0.007) and on day 3 (p= 0.001). Mean lactate in the sepsis group was 2.85 mmol/L and 2.05 mmol/L in the non-sepsis group, showing significant differences (p=0.005). The mean of procalcitonin in the sepsis group was 26.21 ng/ml and 3.37 ng/ml in the non-sepsis group showing significant differences (p-value 0.000) (see table 2)

Table 2. Comparison of laboratory parameters by sepsis and non-sepsis group.
Also, a graph was made to compare MPV values in survivors and non-survivors patients with and without sepsis, in whom platelet size superiority is evidenced in all measurements. (Figure 1)

Figure 1. MPV kinetics from day 1 to day 3 in septic patients that survive and no-survive.
For the variables MPV on day 3, at discharge, and lactate at admission, which were significant in comparing the sepsis and non-sepsis groups, the cut-off point was determined using the ROC curve.
The MPV at day 3 presented a significant area under the curve (AUC) of 0.580 (CI 0.500-0.661), where the cut-off point according to Youden's index was positive for sepsis if MPV ≥9.85 fL. Similarly, lactate on admission was significant with AUC 0.625 (CI 0.555-0.694), with cut-off points 1.15 and 1.25 to predict sepsis. Of these points, the one that showed relevance in the statistical tests and was positive for sepsis was lactate ≥1.15 mmol / L. (see Figure 2.)
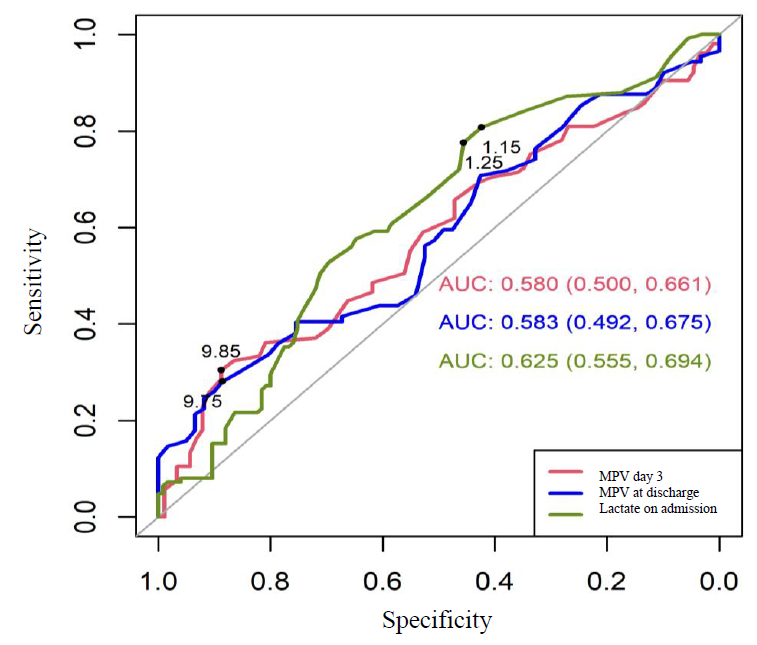
Figure 2. ROC curve for MPV at day 3 and lactate on admission to estimate sepsis.
The following values of sensitivity and specificity for sepsis were observed for the cut-off points determined by the ROC curve. MPV on day 3 presented a sensitivity of 30.48%, a specificity of 88.76%, PPV of 76.20%, NPV of 52.00%, the OR was significant for the cut-off point. Patients with MPV at day 3 ≥9.85 fL are 3.46 times more likely to present sepsis than those with values lower than this cut-off point.
Lactate on day 3 presented a sensitivity of 80, 80%, a specificity of 42, 40%, PPV of 58, 40%, NPV of 68, 80%, the OR was significant for the cut-off point. Patients with lactate on admission ≥1, 15 mmol/L are 3.10 times more likely to present sepsis than those with values lower than this cut-off point.

Table 3. Sepsis predictor parameters by cut-off points for MPV at day 3 and lactate on admission.
Logistic regression was used to determine the multivariate relationship of sepsis with the cut-off points of the MPV on day 3 and lactate at admission and the clinical characteristics of age (median 55 years) and comorbidity due to HBP, which were significant in the bivariate analysis.
The results showed that the MPV (≥9.85 fL) on day 3 (p = 0.005) and lactate at admission ≥1.15 mmol /L presented a multivariate relationship with sepsis. Patients with MPV on day 3 ≥9.85 fL are 3.30 times more likely to present sepsis, while patients with lactate on admission ≥1.15 mmol / L are 2.51 times more likely to present sepsis. Age and HBP did not show a relationship with the presence of sepsis.
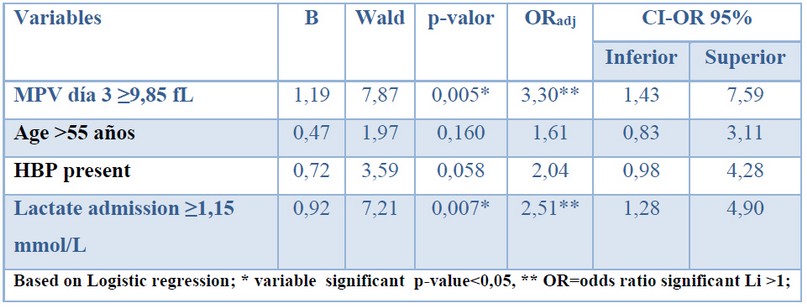
Table 4. A multivariate relationship for sepsis.
DISCUSSION
This study shows that platelet MPV is higher in critically ill patients with sepsis than non-septic patients. Similarly, it was observed that a higher lactate value on admission to intensive care was related to progression to septic states, establishing significant cut-off points and ORs for both variables.
In systemic processes with a high probability of organic impact, the coagulation system is frequently activated 6,7. It has been shown that coagulation and platelet activation occur in early stages in pathologies with a different pathophysiological context, such as sepsis, ischemic heart disease, brain infarction, preeclampsia, and eclampsia 1, 4, 8.
The increase in platelet size and decrease in its count is due to pro-inflammatory cytokines, thrombopoietin, and other substances that stimulate the massive production of spherical and poorly functional young platelets due to the action of spherical and poorly functional young platelets to the severity of the septic shock. All this process leads to the formation of thrombi, almost always culminating in unfavorable outcomes 9, 10
Several studies have demonstrated the predictive utility of MPV in sepsis 9, 11-24 however;, there is little reported bibliography that compares platelet kinetics in critically ill patients with and without sepsis, a critical situation to be demonstrated since several non-infectious diseases can also potentially increase platelet size.
Our findings indicate that the platelet is more significant in patients with sepsis than those without sepsis, but this difference is significant at day 3, marking a significant cut-off point of ≥9.85 fL. Simultaneously, we observed statistical significance in the ROC curve with relevant AUC for lactate at admission with a cut-off point of ≥1.15 mmol / L, which significantly marked septic states' progression. Although no bibliography contrasts these results in patients without sepsis, in patients with sepsis, some studies, for example, two large meta-analyses 25,26 show that the MPV increases at 72 hours, with cut-off points that differ in these studies and is associated with septic states and even higher mortality. When the host's inflammatory response has not been controlled, it has a strong impact, inducing greater clinical severity and unfavorable outcomes.
Although age and HBP were significant variables in the bivariate analysis, they did not present a multivariate relationship, meaning that they do not intervene in the outcome of presenting or not presenting septic states. Something striking was that the platelet count, which is a point to be evaluated in sepsis-induced multi-organ dysfunction, which is more severe than the lower number of platelets, was not significantly different from non-septic pathologies. This observation was unexpected and can be explained by the enrollment of patients with hypertensive pathology of pregnancy (preeclampsia, eclampsia, and HELLP) in the non-septic group; these pathologies also decrease the platelet count due to a different pathophysiological mechanism 27, 28.
Our findings' relevance lies in the fact that we were able to show significant differences in MPV between patients with and without sepsis. This study is the basis for the execution of future studies investigating this biomarker in specific populations; Furthermore, taking into account that this biomarker is routinely reported in the common blood count, it makes it desirable for its immediate applicability.
Among the study's limitations, we can name the potential increase in MPV when using EDTA, which generates an increase in size not attributable to the underlying pathology since it is a post-analytical phenomenon and enhanced by the delay in the processing of the sample. Taking into account that this phenomenon could affect both groups (septic and non-septic); and that K3 tubes were also used, which preserve platelet morphology, and that the processing time was less than 30 minutes (samples from intensive care unit), we consider that this bias plays a minor role in our results. Agustino et al. described similar findings and gave little relevance to this bias in their studies that compared EDTA with citrate 29.
Another essential point to take into account is that, in our work, a high MPV was associated with septic states, but there is no generalized cut-off point reported. Perhaps ethnicity is an important variable to consider since the values reported in eastern countries show MPV cut-off points higher than 11 fL 30,31,32 compared to western countries like ours, where we obtained a cut-off point somewhat lower but no less significant. Besides, our study was carried out in the city of Quito at 2800 meters above sea level. Evidence shows that at high altitudes, hypoxia in non-acclimatized people could generate hyperreactivity and more significant platelet aggregation in response to adenosine diphosphate (ADP), which could also increase other cytometric indicators such as MPV, which indicates the need for more studies to clarify this theory 33,34.
CONCLUSION
This study demonstrated that the mean platelet volume is higher in patients with sepsis than in non-septic patients, findings that are accentuated and become significant on day 3. Likewise, lactate measured at the admission time is a biomarker whose elevation is associated with septic states. On the contrary, the platelet count did not show the expected behavior.
REFERENCES
1. J.Van der Lelie and A. E. G. K.Von dem Borne, "Increased mean platelet volume in septicemia," Journal of Clinical Pathology, vol. 36, no. 6, pp. 693–696, 1983
2. Gutierrez-Romero A, Gutierrez-Grobe Y, Cariilo-Esper R. Volumen Plaquetario Medio: el tamaño sí importa. Med Int Mex. 2013; 29: 3017-310.
3. Greco E, Lupia E, Bosco O. Platelets and Multi-Organ Failure in Sepsis. Int. J. Mol. Sci. 2017, 18, 2200; doi:10.3390/ijms18102200
4. Takatoshi K, Yusuque Y, Keita T. Changes in the Mean Platelets Volume Levels after bloodstream infections have prognostic value. Intern Med. 52: 1487-1493, 2013.
5. Pabón P, Ballesteros F, Moríñigo JL. Influencia del volumen plaquetario medio sobre el pronóstico a corto plazo del infarto agudo de miocardio. Revista Española de Cardiología. 1998. 51 (10): falta números de páginas.
6. Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016; 315: 801–10. https://doi.org/10.1001/jama.2016.0287 PMID: 26903338
7. Ates S, Okzus H, Dogu B. Can mean platelet volume and mean platelet volume/platelet count ratio be used as a diagnostic marker for sepsis and systematic inflammatory response syndrome?. Saudi Med J. 2015 Oct;36(10):1186-90. doi: 10.15537/smj.2015.10.10718.
8. Yun S, Sim F, Goh R. Platelet Activation: The Mechanisms and Potential Biomarkers. BioMed Research International Volume 2016, Article ID 9060143, http://dx.doi.org/10.1155/2016/9060143.
9. Machlus KR, Thon JN, Italiano JE. Interpreting the developmental dance of the megakaryocyte: a review of the cellular and molecular processes mediating platelet formation. Br J Haematol. 2014; 165: 227–236. https://doi.org/10.1111/bjh.12758 PMID: 24499183
10. Nishimura S, Nagasaki M, Kunishima S, Sawaguchi A, Sakata A, Sakaguchi H, et al. IL-1α induces thrombopoiesis through megakaryocyte rupture in response to acute platelet needs. J Cell Biol. 2015; 209: 453–466. https://doi.org/10.1083/jcb.201410052 PMID: 25963822.
11. Hampton T. Platelets' role in adaptative immunity may contribute to sepsis and shock. JAMA, abril 2018, vol 319, num 13, págs 1311. doi:10.1001/jama.2017.12859 https://jamanetwork.com/journals/jama/article-abstract/2677421
12. Monares-Zepeda E, Ríos-Ayala MA. Clinical evaluation of hematic cytometry in critical ill patient. The intensivist point of view. Rev Mex Patol Clin Med Lab. 2019; 66 (2): 100-106.
13. Pattraporn Tajarernmuang, Arintaya Phrommintikul, Atikun Limsukon, Chaicharn Pothirat, and Kaweesak Chittawatanarat, "The Role of Mean Platelet Volume as a Predictor of Mortality in Critically Ill Patients: A Systematic Review and Meta-Analysis," Critical Care Research and Practice, vol. 2016, Article ID 4370834, 8 pages, 2016. https://doi.org/10.1155/2016/4370834.
14. Patel SR, Hartwig JH, Italiano JE. The biogenesis of platelets from megakaryocyte proplatelets. J Clin Invest. 2005; 115: 3348–3354. https://doi.org/10.1172/JCI26891 PMID: 16322779
15. Thon JN, Montalvo A, Patel-Hett S, Devine MT, Richardson JL, Ehrlicher A, et al. Cytoskeletal mechanics of proplatelet maturation and platelet release. J Cell Biol. 2010; 191: 861–874. https://doi.org/10.1083/jcb.201006102 PMID: 21079248
16. Machlus KR, Thon JN, Italiano JE. Interpreting the developmental dance of the megakaryocyte: a review of the cellular and molecular processes mediating platelet formation. Br J Haematol. 2014; 165: 227–236. https://doi.org/10.1111/bjh.12758 PMID: 24499183
17. Nishimura S, Nagasaki M, Kunishima S, Sawaguchi A, Sakata A, Sakaguchi H, et al. IL-1α induces thrombopoiesis through megakaryocyte rupture in response to acute platelet needs. J Cell Biol. 2015; 209: 453–466. https://doi.org/10.1083/jcb.201410052 PMID: 25963822.
18. Grunewald T, et al. Tamaño de plaquetas predice mortalidad en sepsis. Revista Hospimedica. Disponible en: www.hospimedica.es/cuidados-criticos/articles/294742277/tamano-de-plaquetas-predice-mortalidad-en-sepsis.html.
19. Vélez JL, Bucheli R, Ramírez V. Volumen medio plaquetario: predictor de mortalidad en sepsis de pacientes críticos. Revista Metrociencia. 2015; 23: 40-43.
20. Vélez JL, Jara A, Vélez P. ¿Es el volumen medio plaquetario un predictor de mortalidad en sepsis de pacientes críticos? Revista de la Facultad de Ciencias Médicas. 2017; 42 (1); 84-90.
21. Vélez JL. ¿El volumen medio plaquetario es un es un predictor de mortalidad en pacientes sépticos? Revisión de la literatura.Rev Med Hered. 2018; 29:116-119. http://www.scielo.org.pe/pdf/rmh/v29n2/a10v29n2.pdf
22. Sànchez-Calzada A, Navarro JL, Delgado L. Mean platelet volume a marker of sepsis in patients admitted to intensive therapy. Intensive Care Medicine Experimental. 2015; 3(suppl 1):A871.
23. Ates S, Okzus H, Dogu B. Can mean platelet volume and mean platelet volume/platelet count ratio be used as a diagnostic marker for sepsis and systematic inflammatory response syndrome?. Saudi Med J. 2015 Oct;36(10):1186-90. doi: 10.15537/smj.2015.10.10718.
24. Fogagnolo A, Taccone F. Predictive values of platelets morphological indices analysis in septic and non-septic critically ill patients. Minerva Anestesiol-14528. 23 February, 2020
25. Pattraporn Tajarernmuang, Arintaya Phrommintikul, Atikun Limsukon, Chaicharn Pothirat, and Kaweesak Chittawatanarat, "The Role of Mean Platelet Volume as a Predictor of Mortality in Critically Ill Patients: A Systematic Review and Meta-Analysis," Critical Care Research and Practice, vol. 2016, Article ID 4370834, 8 pages, 2016. https://doi.org/10.1155/2016/4370834.
26. J.L. Vélez-Paez, et al. Volumen plaquetario medio como predictor de la mortalidad en pacientes con sepsis: revisión sistemática y metanálisis. Infectio 2020; 24(3): 162-168.
27. Cines DB, Devine LD. Thrombocytopenia in pregnancy. Hematology Am Soc Hematol Educ Program. 2017; 1: 144-151.
28. Myers B. Diagnosis and management of maternal thrombocytopenia in pregnancy. British Journal of Hematology. 2012; 158: 3-15
29. Agustino, AM. et al. Recuento de plaquetas y volumen plaquetario medio en una población sana. Rev Diagn Biol [online]. 2002, vol.51, n.2 [cited 2019-11-17], pp.51-53. Available from: <http://scielo.isciii.es/scielo.php?script=sci_arttext&pid=S0034-79732002000200002&lng=en&nrm=iso>. ISSN 0034-7973
30. Gao Y, Li L, Li Y, Yu X, Sun T, Lan C. [Change of platelet parameters in septic shock patients]. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2014;26(1):28-32.
31. Z. Zhang, X. Xu, H. Ni, and H. Deng, "Platelet indices are novel predictors of hospital mortality in intensive care unit patients," Journal of Critical Care, vol. 29, no. 5, pp. 885.e1–885.e6, 2014.
32. S. Zhang, Y. L. Cui, M. Y. Diao,D. C. Chen, and Z. F. Lin, "Use of platelet indices for determining illness severity and predicting prognosis in critically ill patients," ChineseMedical Journal, vol.128, no. 15, pp. 2012–2018, 2015.
33. Figallo M. Estudios sobre plaquetas. Tesis Doctoral. Lima: Universidad Peruana Cayetano Heredia. 1972.
34. Rocke AS, Paterson GG, Barber MT, et al. Thromboelastometry and Platelet Function during Acclimatization to High Altitude [published correction appears in Thromb Haemost. 2018 Apr;118(4):801. MacInnis, Martin [added]; Main, Shona [corrected to Main, Shona E]; Horne, Elizabeth H [corrected to Horn, Elizabeth H]]. Thromb Haemost. 2018;118(1):63-71. doi:10.1160/TH17-02-0138.
Received: 10 November 2020
Accepted: 15 February 2021
Pablo Andrés Vélez 1. Lucy Baldeón R 2,3 Jorge Luis Vélez-Paez1,2.
1. Servicio de Medicina Crítica-Hospital Pablo Arturo Suárez.
2. Facultad de Ciencias Médicas-Universidad Central del Ecuador.
3. Instituto de Investigación en Biomedicina Universidad Central del Ecuador.
ORCID
Pablo Andrés Vélez Páez 0000-0002-6392-3895 Correo: [email protected]
Jorge Luis Vélez Páez https://orcid.org/0000-0002-6956-4475 Correo: [email protected]
Lucy Baldeón R. https://orcid.org/0000-0002-0447-0136 Correo:
Correspondece: Dr. Pablo Andrés Vélez Páez Facultad de Ciencias Médicas – Universidad Central del Ecuador Iquique N14-121 y Sodiro – Itchimbía, El Dorado. Teléfonos: (593-02) 2528 690 Correo electrónico: [email protected]