2023.08.01.21
Files > Volume 8 > Vol 8 No 1 2023
Physicochemical characteristics and antioxidant capacity of Ecuadorian paramo flowers
Elena Coyago-Cruz 1* , Aida Guachamin 1
, Aida Guachamin 1 , Edwin Vera 2
, Edwin Vera 2 , Melany Moya3, Jorge Heredia-Moya 4
, Melany Moya3, Jorge Heredia-Moya 4 , Elena Beltrán 5
, Elena Beltrán 5
1 Universidad Politécnica Salesiana, Carrera de Ingeniería en Biotecnología de los Recursos Naturales, Sede Quito, Campus El Girón, Grupo de Investigación y Desarrollo en Ciencias Aplicadas a los Recursos Biológicos, Av. 12 de octubre N2422 y Wilson, Quito, 170109, Ecuador; [email protected] ; [email protected] .
2 Escuela Politécnica Nacional, Departamento de Ciencias de los Alimentos y Biotecnología, Facultad de Ingeniería Química, Av. 12 de octubre N2422 y Veintimilla, Quito, 170109, Ecuador; [email protected] .
3 Universidad Central del Ecuador, Facultad de Ciencias Médicas, Carrera de Obstetricia, Iquique, Luis Sodiro N14-121, Quito, 170136, Ecuador; [email protected] .
4 Universidad UTE, Facultad de Ciencias de la Salud Eugenio Espejo, Centro de Investigación Biomédica (CENBIO), Quito, 170527, Ecuador; [email protected].
5 Investigación de Alimentos (CIAL), Facultad de Ciencias de la Ingeniería e Industrias, Ingeniería de Alimentos, Quito, código postal, Ecuador; [email protected].
* Correspondence: [email protected]; Tel.:+593(995129321) (Quito, Ecuador)
Available from: http://dx.doi.org/10.21931/RB/2023.08.01.21
ABSTRACT
Ecuador is a megadiverse country with a wide variety of floral species that have been little studied. In this context, the study's objective was to evaluate the physicochemical characteristics and the antioxidant activity of several floral species of paramo of Pichincha Province in Ecuador. Thus, the weight, size, color, pH, soluble solids, moisture and ash of fresh flower was quantified. In addition, carotenoids, phenolic compounds and antioxidant activity were quantified in lyophilized powder. The results obtained showed that the flowers of Werneria nubigena were the longest (43,80 cm); Brugmansia x candida the widest (9,88cm) and heaviest (9,22g); Tristerix longebracteatus presented high soluble solids content (21,5 °Brix), Lupinus microphyllus high pH (14,00), Ceanothus maritimus high titratable acidity (0,26%), Castilleja integrifolia high ash content (6,42%) and Bidens ferulifolia high moisture content (95,73%). In addition, the highest ranges of total carotenoids and total phenolics were presented by yellow Bidens ferulifolia (24,81 µg β-carotene/g PS) and Fuchsia vulcania (531,77 mg EAG /g PS), respectively. Finally, it was found in Bomarea multiflora high values of antioxidant capacity (182,08 trolox eq. µmol/ g PS). These results suggest that the paramo flowers contain essential bioactive compounds that could be used for food, medicinal and cosmetic purposes.
Keywords: Bioactive compounds, carotenoids, phenolic compounds, Andean flowers
INTRODUCTION
Ecuador is divided into four regions: the coast, which the Pacific Ocean borders, the Andean highlands, which is hilly and volcanic; the Amazonian region, which is home to the Amazon Rainforest; and the insular region, which is made up of islands and archipelagos in the Pacific Ocean.
Due to its unique geography, it is a megadiverse country with a wide variety of habitats, enabling it to support a diversity of plant and animal species. Evolutionary processes have conferred characteristics on a continental and regional scale, geomorphology, soil type, fluctuating precipitation patterns, habitat fragmentation and temperature gradient. Thus, the paramo is a high mountain ecosystem located at an altitude of more than 2800 meters above sea level (masl) with cold and humid climates, frequent rainfall, strong winds and nearly constant cloud cover; however, above 4200 masl the vegetation is scarce, and the paramo is desert or has large sandbanks 1.
The paramos are biologically crucial due to the diversity and singularity of its flora and fauna species, which are endemic to the region. As a result, species diversity increases between 3000 and 3400 masl and diminishes as altitude increases 2. Asteraceae, Orchidaceae, Melastomataceae, Campanulaceae, Poaceae, Bromeliaceae, Gentianaceae, Cyperaceae, Ericaceae and Solanaceae, are the most abundant families of plants in Ecuador's paramo; the first seven of these have endemic species. The genera with the most significant number of species are Miconia, Stelis, Epidendrum, Baccharis, Calceolaria, Pleurothalis, Pentacalia and Tillandsia 3. Many of these plants have showy flowers mainly used for decoration and have not received much attention from researchers looking into bioactive compounds. However, certain flowers contain carotenoids, phenols, and alkaloids, among other organic molecules. This characteristic has contributed to the use of floral species in food and traditional medicine 4.
On the other hand, an antioxidant is any substance that delays or prevents the oxidation of oxidable substrates such as lipids, proteins, carbohydrates, and DNA 5. Some carotenoids and phenolic compounds are antioxidants, which can help to prevent disease 6. Table 1 shows the edible uses, analyzed parts, phenolic compounds, carotenoids and antioxidant activity by ABTs and DPPH method of some paramos floral species. Thus, it is shown that most of the paramo species were used in food as an infusion using flowers. In addition, in most cases, the concentration of total phenolic compounds was evaluated, except in Diplostephium hartwegii and Bidens ferulifolia. Only 5 of the 13 species studied bibliographically (Hypochaeris radicata, Taraxacum officinale, Fuchsia vulcania, Tropaeolum majus and Chuquiraga jussieuri) presented carotenoids studies, and antioxidant activity was measured in the majority of cases using the ABTS or DPPH assay. In this context, the objetive of this study was to evaluate the physicochemical characteristics and antioxidant activity of paramo flowers.
MATERIALS AND METHODS
Reagents and Standards
Ethanol HPLC grade and reagents of analytical quality such as hydrochloric acid, sodium hydroxide, dichloromethane, acetone, and sodium carbonate were purchased from Merck (Merck, Germany), while methanol HPLC grade from Pharmco (PHARMCO by Greenfield Global, California). Folin Ciocalteu reagent and all standard (β-Carotene, gallic acid, and Trolox) were obtained from Sigma-Aldrich (Merck, Germany).
Plant Materials
The experimentation was authorized by the Ministerio del Ambiente of Ecuador under the framework contract MAE-DNB-CM-2017-0080-UTE, Project MAE-DNB-2019-0911-O. The collection used the International Code of Conduct for the collection and transfer of plant germplasm of the FAO 21, and the publications of Biodiversity International 22 were utilized in sampling 30 species of paramos in the Pichincha province (Table 2). The collection was carried out between February and July 2020.
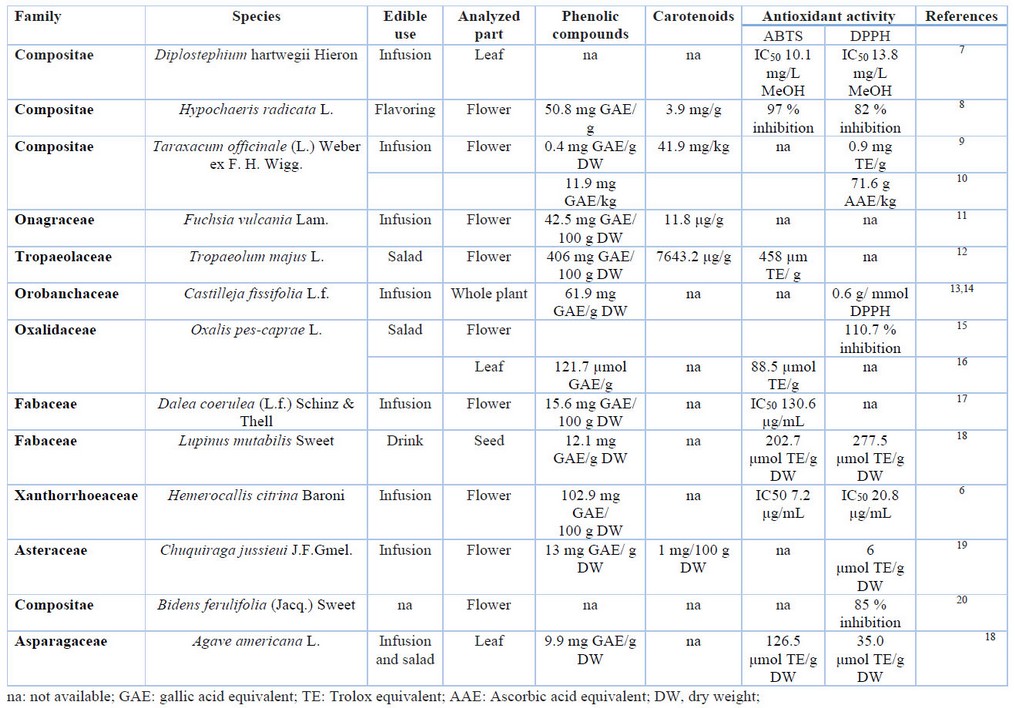
Table 1. Content of phenolic compounds, carotenoids and antioxidant activity of some paramo flowers
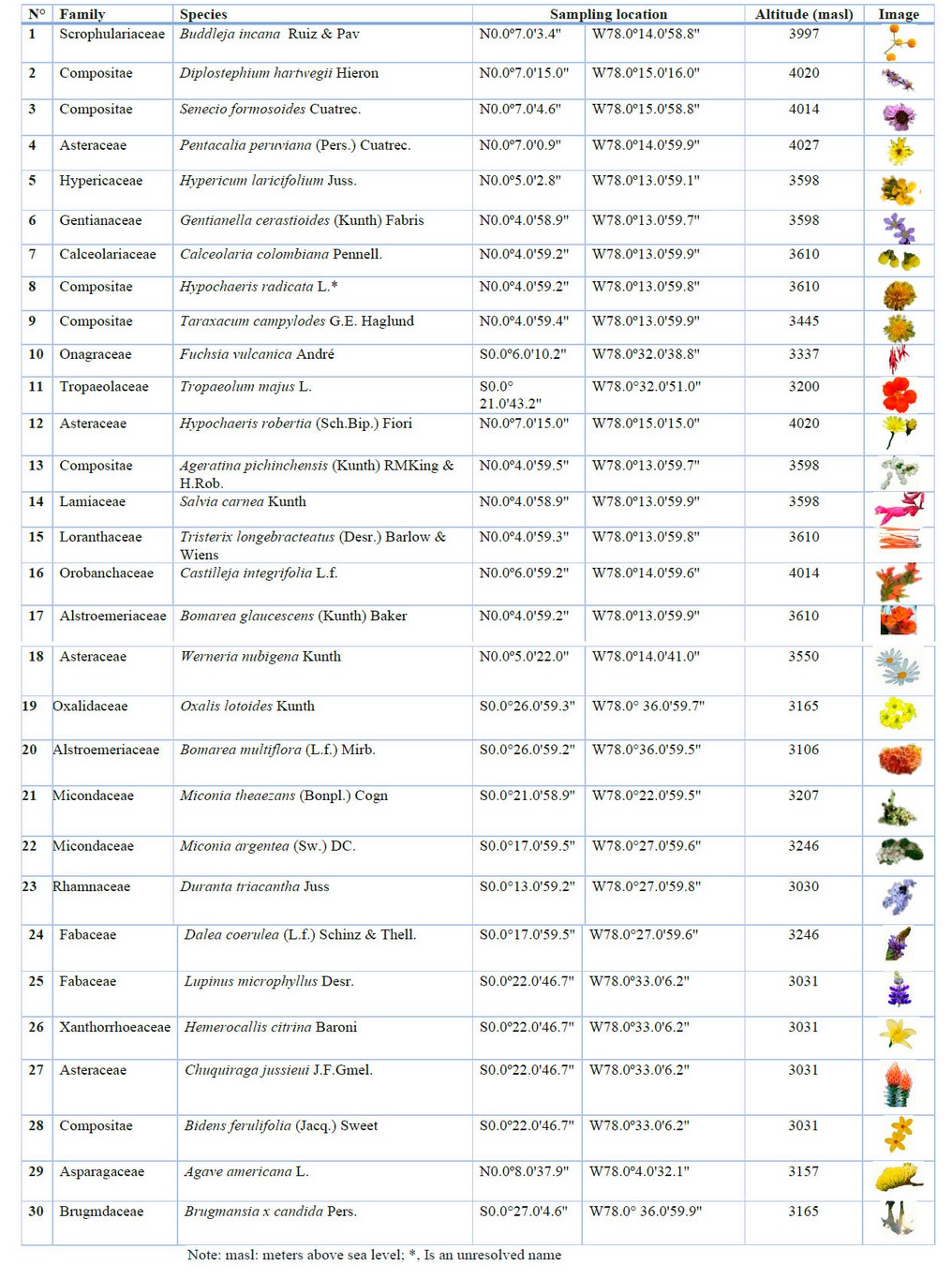
Table 2. Family, species, sampling location, altitude and flower image in the studio
Determination of physicochemical characteristics
The weight (g) of fresh flower petals was determined using a Mettler Toledo scale (Mettler Toledo, United States), as well as the equatorial diameter and longitudinal diameter (mm)11. Additionally, the soluble solids (°Brix) using a Hitech hand-held refractometer (Hitech RHB-32 ATC, United States)23, % total titratable acidity24, pH using a SevenMulti S47 automatic pH meter (Mettler Toledo, United States)25, % humidity in a Memmert Be 20 oven (Memmert GmbH+Co.KG, Spain)26,27, and % ash in a Thermolyne muffle (Thermo Fisher Scientific, United States)28 were quantified. The other fresh flowers were frozen at -80 °C and freeze-dried in a Christ Alpha 1-4 LDplus equipment (Martin Gwfriertrocknungsanlagen GmbH, Germany). The freeze-dried samples were ground and stored in amber bottles until analysis.
Determination of bioactive compounds and antioxidant activity
Quantification of total carotenoids
Approximately 10 mg of lyophilized material and 300 µL of acetone:methanol: dichloromethane (1:1:2) were used for the microextraction of carotenoids. The mixture was stirred for one minute in an ultrasound model VWR (VWR International, EE.UU.), and the supernatant was recovered after centrifugation at 14000 rpm for 4 min in a MiniSpin microcentrifuge (Eppendorf, EE.UU.). This process was repeated until the solids showed no color. Finally, the combined extracts were dried in a rotary evaporator, re-dissolved in ethanol HPLC, and quantified in a Jasco V-730 spectrophotometer (Mettler Toledo, Ecuador), at a wavelength of 450 nm. The total carotenoids were expressed as µg equivalent of β-carotene / g of dry weight (DW) 4.
Quantification of total phenolic compounds
About 10 mg of lyophilized samples were extracted with 500 µL of an 80 % methanol solution that had been acidified with 0.1% hydrochloric acid. The mixture was homogenized in a Vortex Mixer VM 300 (Interbiolab Inc., Florida), shaken in a Fisher Scientific FS60 ultrasonic bath (Fisher Scientific, USA) for 3 min, centrifuged at 1400 rpm for 5 min at 4 °C in a MiniSpin series microcentrifuge (Eppendorf, Germany), and the supernatant was collected The extraction procedure was carried out in triplicate11,29. The recovered supernatant was filtered through a 0.45 µm PVDF filter to quantify phenolic compounds. In a 96-well plate, 20 µL of the methanolic extract was mixed with 100 µL of a 1:4 Folin-Cioacalteu solution, shaken, and allowed to standard for 4 min. Then, 75 µL of a sodium carbonate solution (100 g/L) was added to the mixture and shaken for 1 min. After two hours at room temperature, absorbance was measured at 750 nm using a BioTek Symergy H1 microplate reader (Agilent, United States). In addition, a solution of gallic acid between 10 to 200 mg/L was employed as a calibration curve. The results were reported as mg of gallic acid equivalent / g of dry weight (DW) 30.
Determination of antioxidant activity by ABTS
Approximately 20 mg of lyophilized powder were weighed and combined with 400 µL of Pharmco HPLC-grade methanol (Greenfield Global, California) and 400 µL of distilled water. The mixture was homogenized in a vortex, shaken in an ultrasonic bath for 3 min, and the supernatant was separated by microcentrifugation at 14000 rpm for 5 min at 4 °C. The resulting solid was dissolved in 560 µL acetone and 240 µL distilled water. The procedure was repeated to recover the supernatant that had been combined with the previous supernatant. The resulting combination was refrigerated until it was quantified 30.
The ABTS•+ radical was prepared by mixing a 1:1 solution of 7 mM ABTS Sigma-Aldrich (Merck, Germany) with 2.45 mM potassium persulfate Sigma-Aldrich (Merck, Germany) and letting itstand for 16 hours in the dark. After that, the ABTS•+ radical solution was diluted with absolute ethanol by a factor of about 1 to 10, or until an absorbance of 0.7 at 754 nm was obtained. On the other hand, a stock solution of 2.5 nM Trolox Sigma-Aldrich (Merck, Germany) was used to prepare the calibration curve, which was diluted by 75, 50, 25, and 12.5 %. For sample quantification, 20 µL of ABTS•+ radical solutions were added to a 96-well VWR Tissue culture plate (Novachen, USA) together with 10 µL of the final or standard supernatant. The measurement was taken at 270 nm using a spectrophotometer with a Thermo Scientific Multiskan GO microplate reader (Agilent Scientific Instruments, California) 31,32. Antioxidant activity was expressed as mmol trolox equivalent per gram dry weight (mmol TE/g PS).
RESULTS AND DISCUSSION
Physicochemical properties (weight, flower height, flower width, pH, soluble solids, total titratable acidity, ash, and humidity)
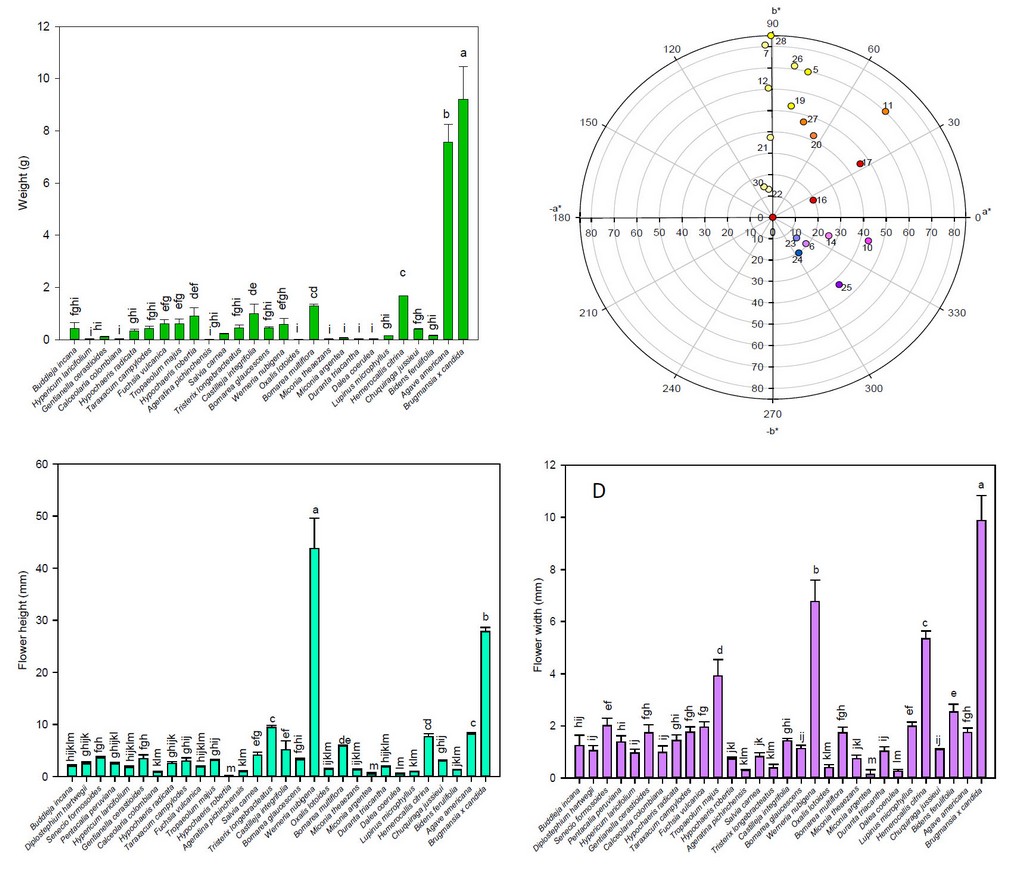
The weight (Figure 1-A), color (Figure 1-B), height (Figure 1-C) and width (Figure 1-D) of the flowers under study are shown in Figure 1. Thus, the weight of the flowers ranged from 0.01 (Ageratina pichinchensis) to 9.22 g (Brugmansia x candida). Thus, this study's weight of Tropaeolum majus (0.61 g) was lower than the same species reported by other authors (0.72 g) 33. In addition, as shown in Figure 1-B, most flowers are concentrated in the first quadrant with yellow to red colorations, followed by the fourth quadrant with violet to blue flowers.
The height of the flowers under study varied from 0.17 cm (Hypochaeris robertia) to 43.80 cm (Werneria nubigena), and the width from 0.14 cm (Miconia argentea) to 9.88 cm (Brugmansia x candida). Thus, the length and width of Buddleja globosa (2.00 and 1.24 cm, respectively), Diplostephium hartwegii (2.47 and 1.05 cm, respectively), Fuchsia vulcania (1.88 and 1.95 cm, respectively), and Agave americano (8.16 and 1.75 cm, respectively) showed comparable values with other studies, which presented values of 0.50 to 0.60 cm height and 0.30 to 0.40 cm width 34; 1.40 to 1.50 cm height and 0.12 to 0.16 cm width 35; 2.94 cm height and 0.50 cm width 36; and 11 cm height and 6.00 cm width 37, respectively. In addition, the length of Brugmansia x candida (27.84 cm) in this study showed comparable results with other authors 38.
Figure 1. Average values of weight, color, height and width of the flowers under study
Note: Vertical bars indicate the standard error. Different lowercase letters indicate homogeneous groups with Tukey, p < 0.05. 1, Buddleja incana; 2, Diplostephium hartwegii; 3, Senecio formosides; 4, Pentacalia peruviana; 5, Hypericum laricifolium; 6, Gentianella cerastioides; 7, Calceolaria colombiana; 8, Hypochaeris radicata; 9, Taraxacum campylodes; 10, Fuchsia vulcanica; 11, Tropaeolum majus; 12, Hypochaeris robertia; 13, Ageratina pichinchensis; 14, Salvia carnea; 15, Tristerix longebracteatus; 16, Castilleja integrifolia; 17, Bomarea glaucescens; 18, Werneria nubigena; 19, Oxalis lotoides; 20, Bomarea multiflora; 21, Miconia theaezans; 22, Miconia argentea; 23, Durantha triacantha; 24, Dalea coerulea; 25, Lupinus microphyllus; 26, Hemerocallis citrina; 27, Chuquiraga jussieui; 28, Bidens ferulifolia; 29, Agave americana; 30, Brugmansia x candida
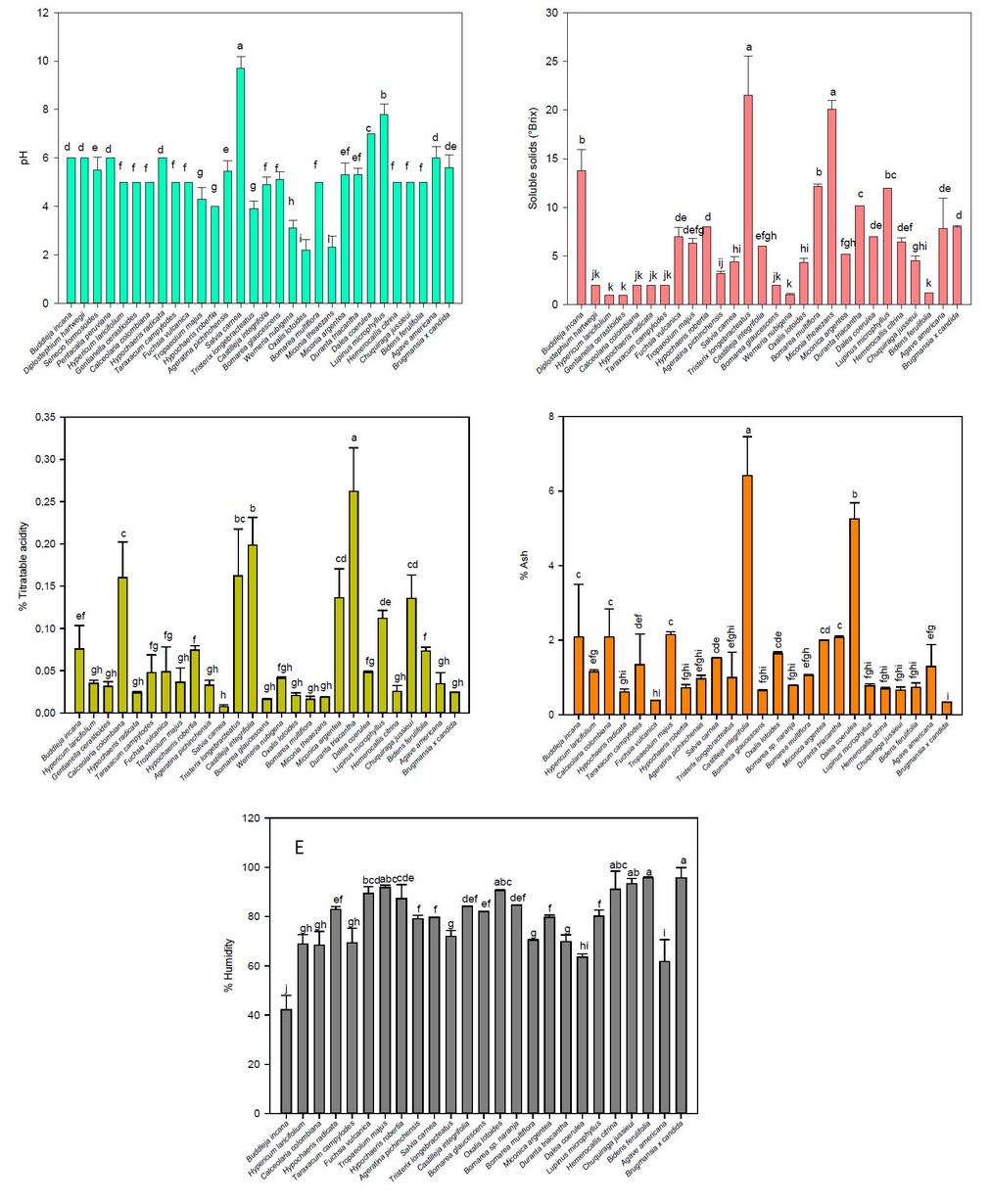
The pH (Figure 2-A), soluble solids (Figure 2-B), titratable acidity (Figure 2-C), ash (Figure 2-D), and humidity (Figure 2-E) of the flowers under study are shown in Figure 2. Thus, the pH varied from 2.20 (Oxalis lotoides and Miconia theazans) to 9.7 (Salvia carnea); the soluble solids ranged from 1.00 (Hypericum laricifolium and Gentianella cerastioides) to 21.5 °Brix (Tristerix longebracteatus), while the total titratable acidity expressed as percentage of citric acid varied from 0.10 (Salvia carnea) to 0.26 (Duranta triacantha). The values of soluble solids (6.30 °Brix), pH (4.3), and total titratable acidity (0.04 %) in this study for Tropaeolum majus were similar to those reported by other authors (7.53 °Brix, 4.97 of pH, and 0.32 % of total titratable acidity, respectively) 39. In addition, edible flowers such as Diplostephium hartwegii, Taraxacum campylodes, Fuchsia vulcanica, Tropaelum majus, Dalea coerulea, Hemerocallis citrina, Chuquiraga jussieui, and Agave amaricana showed pH values between 4.3 and 7, indicating that the petals of these flowers may be susceptible to attack by microorganisms, causing organoleptic alterations, as suggested by otrer authors 40.
Figure 2. Average values of pH, soluble solids, titratable acidity, ash, and humidity of the flowers under study.Note: Vertical bars indicate the standard error. Different lowercase letters indicate homogeneous groups with Tukey, p < 0.05
In this study, the ranges for ash and moisture were 0.33 % (Brugmansia x candida) to 6.42 % (Castilleja integrifolia) and 42.24 % (Buddleja globosa) to 95.73 % (Bidens ferulifolia), respectively. As a result, the humidity of Fuchsia vulcania (81.26 %) was comparable to values reported by other authors 11. In contrast, the values of ash and humidity of Troapaeolum majus (21.5 % and 91.81 %, respectively) were similar to other studies (0.63 % and 89.93 %, respectively) 41. While the humidity of the Agave americana flowers in this study (61.73 %) was lower than that reported by other authors (86.62 %) 37. In turn, the ash and moisture values (0.66 % and 93.3 %, respectively) of the Chuquiraga jussieui flowers in this study differed from those found by other authors (5.08 % and 9.77 %, respectively) 42. In contrast, these values for Taraxacum campylodes (1.34 % and 69.31 %%, respectively) had a certain relationship with those reported by other authors (2.00 % and 5.9 %, respectively) 43.
Bioactive compounds and antioxidant activity quantification
Total carotenoids of the flowers under study are shown in Figure 3. Thus, the total carotenoids content ranged from 0.32 (Senecio formosoides) to 24.81 µg β-carotene/ g dried weight (DW) (Bidens ferulifolia). These results indicated that yellow and orange flowers, such as Bidens ferulifolia (24,81 µg β-carotene/ g DW), Hypericum laricifolium (22,18 µg β-carotene/ g DW), Oxalis lotoides (21,66 µg β-carotene/ g DW), and Buddleja globose (15,74 µg β-carotene/ g DW) showed the highest concentrations of total carotenoids, a result that was also reported by other authors 11,44,45.
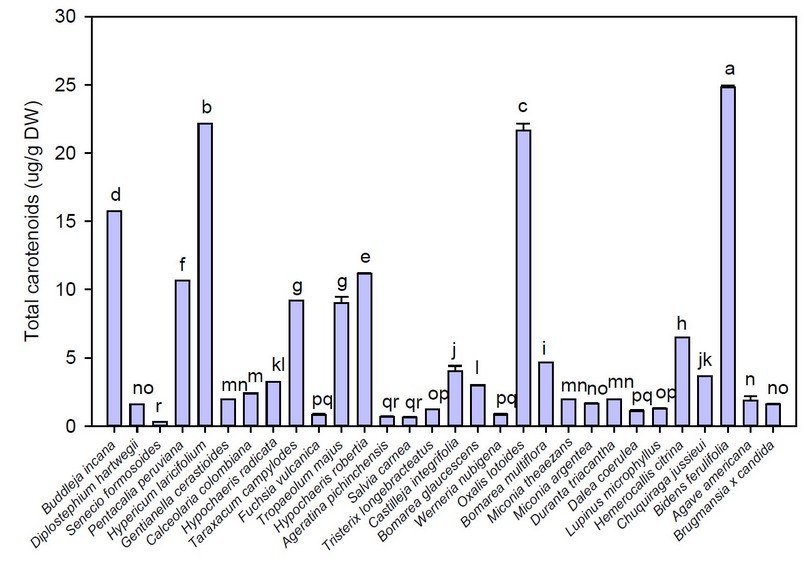
Figure 3. Average values of total carotenoids of the flowers under study
Note: Vertical bars indicate the standard error. Different lowercase letters indicate homogeneous groups with Tukey, p < 0.05
Total phenolics of the flowers under study are shown in Figure 4. Total phenolic compounds of the flowers under study varied from 72.13 (Agave americana) to 531.77 mg gallic acid equivalents (GAE)/g DW (Fuchsia vulcania). In this study, the highest phenolic compound concentrations were found in Fuchsia vulcania (531.77 mg GAE/g DW), Lupinus microphyllus (498.58 mg GAE/g DW), Miconica theaezans (469.27 mg GAE/g DW), Gentianella cerastioides (350.63 mg GAE/g DW), and Senecio formosoides (346.58 mg GAE/g DW). In contrast, F. magellanica in this study showed lower values than those reported by other authors (42.49 mg GAE/100 g DW) 11. Despite using the same extraction methodology, this difference may be due to the fact that this study sampled paramo species at altitudes higher than 3000 meters above sea level, whereas the comparison species were cultivated in four seasons and at sea level, conditions that modify the content of phenolic compounds 4.
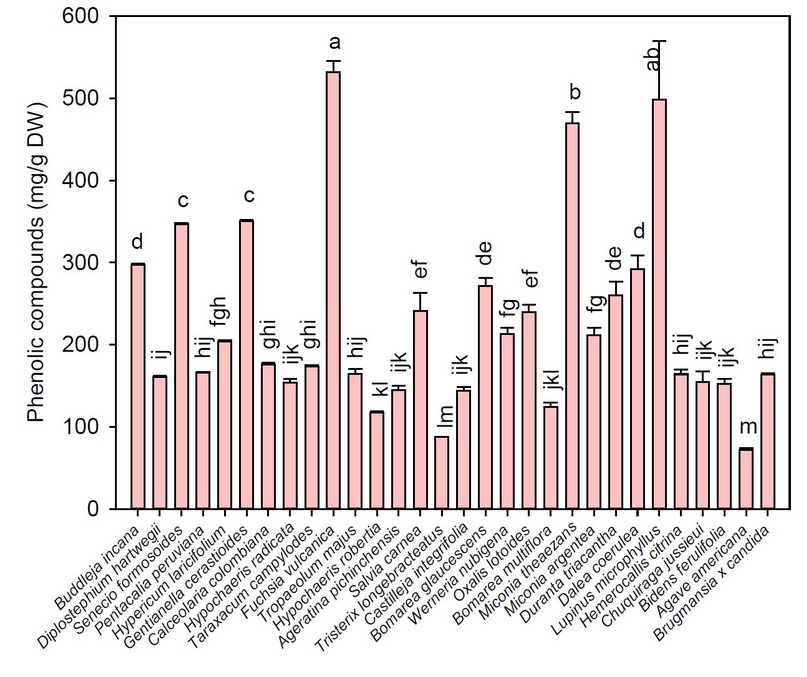
Figure 4. Average values of total phenolic compounds of the flowers under study
Note: Vertical bars indicate the standard error. Different lowercase letters indicate homogeneous groups with Tukey, p < 0.05
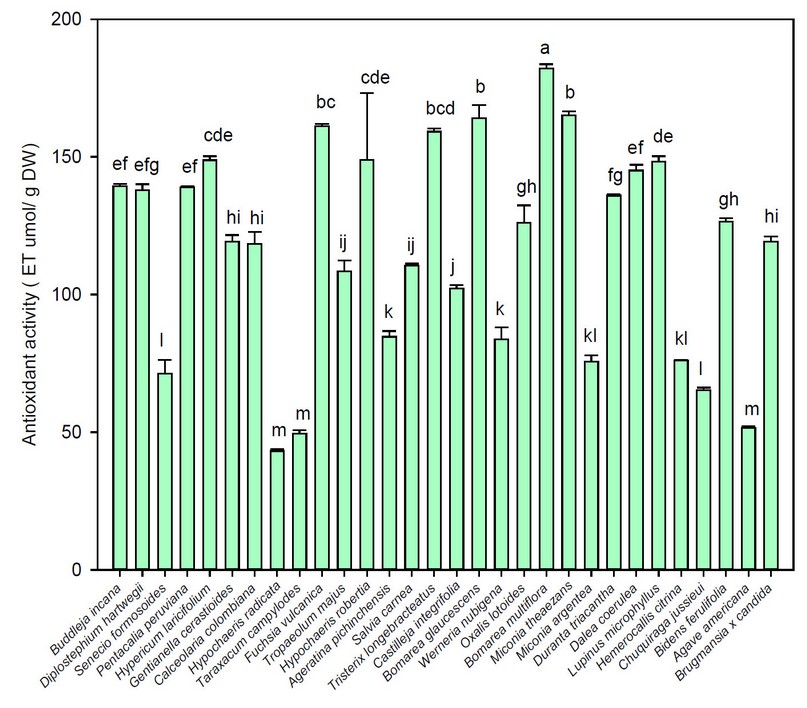
Figure 5. Average values of antioxidant activity of the flowers under study
Note: Vertical bars indicate the standard error. Different lowercase letters indicate homogeneous groups with Tukey, p < 0.05
Figure 5 depicts the antioxidant activity of the flowers under investigation. Thus varied from 43.13 trolox equivalent µmol / g DW (Hypochaeris radicata) to 182.08 trolox equivalent µmol / g DW (Bomarea multiflora). The highest antioxidant activity values were found in Bomarea multiflora (182.08 trolox equivalent µmol / g DW), Miconia theaezans (165.14 trolox equivalent µmol / g DW), Bomarea glaucescens (164.10 trolox equivalent µmol / g DW), and Fuchsia vulcania (161.08 trolox equivalent µmol / g DW). As a result, the inhibitory activity of Hypochaeris radicata in this study (26.39 % inhibition) was lower than the values reported by other authors (97.00 % inhibition) 46. Several studies have shown that this variation could be attributable to the method used for quantification47.
CONCLUSIONS
Ecuador, due to its great biodiversity, hosts a great variety of floral species with potential medicinal and nutritional properties. As a result, different species had high values of weight (9.22 g Brugmansia x candida), height (43.80 cm Werneria nubigenea), width (9.88 Brugmansia x candida), pH (9.7 Salvia carnea), soluble solids (21.5 °Brix Tristerix longebracteatus), titratable acidity (0.26 % Ceanothus maritimus), ash (6.42 % Castilleja integrifolia, moisture (95.73 % Bidens ferulifolia), total carotenoids (24.81 µg of β-carotene / g of DW Bidens ferulifolia), total phenolic compounds (531.77 mg of GAE / g of DW Fuchsia vulcania), and antioxidant activity (182.08 µmol Eq trolox / g of DW Bomarea multiflora). Present results could contribute to the development of new products in the field of medicine, cosmetics and food.
Author Contributions: formal analysis, Coyago Elena; investigation, Coyago Elena.; resources, Coyago Elena.;writing—original draft preparation, Aida Guachamin, Coyago Elena, Michael Villacís; writing—review and editing, Coyago Elena, Vera Edwin, Moya Melany, Jorge Heredia-Moya; project administration, Coyago Elena.; funding acquisition, Coyago Elena. All authors have read and agreed to the published version of the manuscript.
Funding: This research was carried out under the framework contract MAE-DNB-CM-2017-0080-UTE. Project MAE-DNB-2019-0911-O and was financed by the Ecuadorian Corporation for the Development of Research and the Academy (CEDIA) within the CEPRA-XII-2019-Flores Andinas Project "Physical-chemical characterization and bioactivity tests of Andean floral species with nutritional potential and preventive effect of certain human diseases".
Conflicts of Interest: "The authors declare no conflict of interest."
REFERENCES
1. MAE. Sistema de clasificación de los ecosistemas del Ecuador continental. Subsecretaría de Patrimonio Natural https://www.ambiente.gob.ec/wp-content/uploads/downloads/2012/09/LEYENDA-ECOSISTEMAS_ECUADOR_2.pdf (2012).
2. Hofstede, R. Los páramos andinos ¿Qué sabemos? Estado de conocimiento sobre el impacto del cambio climático en el ecosistema páramo. Tiempos de Crisis sistémica (UICN, Quito, Ecuador, 2014). doi:10.2307/j.ctvpv50bh.8.
3. Rangel-Ch., J. R. Colombia diversidad biótica XVI Patrones de riqueza y de diversidad de las plantas con flores en el bioma de páramo. (Universidad Nacional de Colombia. Facultad de Ciencias, Instituto de Ciencias Naturales, 2018).
4. Coyago-Cruz, E., Corell, M. & Meléndez-Martínez, A. Estudio sobre el contenido en carotenoides y compuestos fenólicos de tomates y flores en el contexto de la alimentación funcional. (Punto Rojo Libros, S.L., 2017).
5. Venereo, J. Daño oxidativo, radicales libres y antioxidantes. Rev. Cuba. Med. Mil. 31, 126–159 (2002).
6. Wang, J. et al. Ethyl acetate fraction of Hemerocallis citrina Baroni decreases tert-butyl hydroperoxide-induced oxidative stress samage in BRL-3A cells. Oxid. Med. Cell. Longev. 2018, 1–13 (2018).
7. Rodriguez, O. & Torrenegra, R. Flavonoids, terpenes and the anti-Oxidant activity of Diplostephium phylicoides (HBK.) Wedd. Indian J. Sci. Technol. 11, 1–7 (2018).
8. Senguttuvan, J. & Subramanium, P. Micropropagation of medicinal herb, Hypochaeris radicata L. via indirect organogenesis. Res. Biotechnol. 5, 1–9 (2014).
9. Pădureţ, S., Amariei, S., Gutt, G. & Piscuc, B. The evaluation of dandelion (Taraxacum officinale) properties as a valuable food ingredient. Rom. Biotechnol. Lett. 21, 11569–11575 (2016).
10. Kucekova, Z., Mlcek, J., Humpolicek, P. & Rop, O. Edible flowers - antioxidant activity and impact on cell viability. Cent. Eur. J. Biol. 8, 1023–1031 (2013).
11. Meléndez-Martínez, A. et al. Screening for innovative sources of carotenoids and phenolic antioxidants among flowers. Foods 10, 1–23 (2021).
12. Garzón, G. A. & Wrolstad, R. E. Major anthocyanins and antioxidant activity of Nasturtium flowers (Tropaeolum majus). Food Chem. 114, 44–49 (2009).
13. Ulloa, C., Álvarez, S., Jørgensen, P. & Minga, D. Guía de 100 Plantas silvestres del páramo del Parque Nacional Cajas. Guia de 100 plantas silvestres del paramodel Parque Nacional de Cajas (Ministerio del Ambiente, 2008).
14. Mejía-Giraldo, J., Henao-Zuluaga, K., Gallardo, C., Atehortúa, L. & Puertas-Mejía, M. Novel in vitro antioxidant and photoprotection capacity of plants from high altitude ecosystems of Colombia. Photochem. Photobiol. 92, 150–157 (2016).
15. Purohit, S., Rana, S., Idrishi, R., Sharma, V. & Ghosh, P. A review on nutritional, bioactive, toxicological properties and preservation of edible flowers. Futur. Foods 4, 1–14 (2021).
16. Gonçalves, J., Oliveira, G. & Almeida, L. Compostos bioativos em flores comestíveis. Biológicas & Saúde 9, 11–20 (2019).
17. De la Cruz, M. A. et al. Compuesto fenólicos y actividad antioxidante de extractos hidroalcohólicos provenientes de la flores de Dalea Coerulea (L.F.) Schinz & Thell. in VII Congreso latinoamericano de plantas medicinales ‘Plutarco Naranjo’ 207 (2019).
18. Chirinos, R., Pedreschi, R., Rogez, H., Larondelle, Y. & Campos, D. Phenolic compound contents and antioxidant activity in plants with nutritional and/or medicinal properties from the Peruvian Andean region. Ind. Crops Prod. 47, 145–152 (2013).
19. Guerrero Bonilla, D. et al. Bioactive compounds and antioxidant capacity of Chuquiraga jussieui J.F.Gmel from the highlands of Ecuador. Nat. Prod. Res. 34, 2652–2655 (2019).
20. Shymanska, O., Vergun, O., Fishchenko, V. & Rakhmetov, D. Antioxidant potential of some Asteraceae Bercht. & J. Presl. representatives. Agrobiodiversity Improv. Nutr. Heal. Life Qual. 70–77 (2020) doi:10.15414/agrobiodiversity.2020.2585-8246.070-77.
21. FAO. Texto concertado del proyecto de código internacional de conducta para la recolección y transferencia de germoplasma vegetal. (1993).
22. Guarino, L., Ramanatha Rao, V. & Goldberg, E. Collecting plant genetic diversity: technical guidelines. 2011 update. (2011).
23. US-ISO-2173:2003. Fruit and vegetable products. Determination of soluble solids. Refractometric method. Int. Organ. Stand. 1, 17 (2009).
24. US-ISO750:1998. Fruit and vegetable products — Determination of titratable acidity. Int. Organ. Stand. 1, 4 (2009).
25. ISO1842:1991. Fruits, vegetables and derived products-Sampling and methods of test. Part 5: Determination of pH. East African Stand. 5, (2000).
26. Garcia-Mogollon, C., Alvis-Bermudez, A. & Dussán-Sarria, S. Validación del método de microondas para determinar humedad en ñame espino (Dioscorea Rotundata Poir). Inf. Tecnol. 28, 87–94 (2017).
27. INEN1953. Gelatina pura comestible. Determinación de la pérdida por calentamiento. Instituto Ecuatoriano De Normalización vol. 1 4 (2012).
28. ISO-5520. Fruits, vegetables and derived products - Determination of alkalinity of total ash and of water-soluble ash. Int. Stand. ISO 1, 13 (1981).
29. Periago, M. J., Martínez-Valverde, I., Chesson, A. & Provan, G. Phenolic compounds, lycopene and antioxidant activity in commercial varieties of tomato (Lycopersicum esculentum). J. Sci. Food Agric. 82, 323–330 (2002).
30. Bobo-García, G., Davidov-Pardo, G., Arroqui, C. & Marín-Arroyo, M. Intra-laboratory validation of microplate methods for total phenolic content and antioxidant activity on polyphenolic extracts, and comparison with conventional spectrophotometric methods. SCI 46, 29–33 (2014).
31. Kuskoski, E. M., Asuero, A. G., Troncoso, A. M., Mancini-Filho, J. & Fett, R. Aplicación de diversos métodos químicos para determinar actividad antioxidante en pulpa de frutos. Food Sci. Technol. 25, 726–732 (2005).
32. Rivero-Pérez, M. D., Muñiz, P. & González-Sanjosé, M. L. Antioxidant profile of red wines evaluated by total antioxidant capacity, scavenger activity, and biomarkers of oxidative stress methodologies. J. Agric. Food Chem. 55, 5476–5483 (2007).
33. Nunes, E. et al. Determinação do ponto de colheita de flores de Tropaeolum majus L. Rev. Bras. Agropecuária Sustentável 8, 37–43 (2018).
34. Navas, L. Flora de la cuenca de Santiago de Chile, Tomo III. (Ediciones de la Universidad de Chile, 1979).
35. Bohorquez, R. Determinación de actividad antioxidante de extractos de hojas de Diplostephium phylicoides (Kunth) Wedd. (Universidad de Ciencias Aplicadas y Ambietales, 2016).
36. Smith-Ramirez, C. Los picaflores y su recurso floral en el bosque templado de la isla de Chiloé, Chile. Rev. Chil. Hist. Nat. 66, 65–13 (1993).
37. Semuli, M. Nutritional composition, descriptive sensory analysis and consumer acceptability of products developed from Agave americana flowers. (University of the Free State, 2014).
38. Álvarez, L. Borrachero , cacao sabanero o floripondio ( Brugmansia Spp .) un grupo de plantas por redescubrir en la biodiversidad latinoamericana. Cult. y Drog. 13, 77–93 (2008).
39. Cardoso, I., Botrel, N., De-Barros, E., Madeira, N. & De-Castro, R. Caracterização de flores da capuchinha (Tropaeolum majus L.) armazenadas em diferentes temperaturas. in XI Brazilian Meeting on Chemistry of Food and Beverages V Simpósio de Engenharia e Ciência de Alimentos, Brasil 1–6 (2016).
40. Vásquez, G. La Contaminación de los alimentos, un problema por resolver. Rev. Salud Uis 35, 48–57 (2003).
41. Navarro-González, I., González-Barrio, R., García-Valverde, V., Bautista-Ortín, A. B. & Periago, M. J. Nutritional composition and antioxidant capacity in edible flowers: Characterisation of phenolic compounds by HPLC-DAD-ESI/MSn. Int. J. Mol. Sci. 16, 805–822 (2015).
42. Palma, M. & Tamayo, E. Caracterización de la chuquiragua (Chuquiraga jussieui) con perspectivas agroindustriales. (Universidad de las Américas, 2019).
43. Palate, K. Estudio de la composición nutricional de flores comestibles Diente de león (Taraxacum officiale), Tronadora (Tecoma stans), Mastuerzo (Tropaeolum majus), Girasol (Helianthus annuus) para potenciar su consumo. (Universidad Técnica de Ambato, 2021).
44. Coyago-Cruz, E. Estudio sobre el contenido en carotenoides y compuestos fenólicos de tomates y flores en el contexto de la alimentación funcional. (Universidad de Sevilla, Departamento de Ciencias Agroforestales, 2017).
45. Lu, C. et al. Flower color classification and correlation between color space values with pigments in potted multiflora chrysanthemum. Sci. Hortic. (Amsterdam). 283, 110082 (2021).
46. Ko, H., Eom, T., Song, S., Jo, G. & Kim, J. Tyrosinase and α-glucosidase inhibitory activities and antioxidant effects of extracts from different parts of Hypochaeris radicata. Korean J. Med. Crop Sci. 25, 139–145 (2017).
47. Takahashi, J., Guilherme, F., Fidelis, M., Borges, L. & Sande, D. Edible flowers: bioactive profile and its potential to be used in food development. Food Res. Int. 129, 108868 (2020).
Received: September 26, 2022 / Accepted: October 15, 2022 / Published:15 February 2023
Citation: Coyago-Cruz E, Guachamin A , Vera E, Moya M, Heredia-Moya J, Beltrán E . Physicochemical characteristics and antioxidant capacity of Ecuadorian paramo flowers. Revis Bionatura 2023;8 (1)21. http://dx.doi.org/10.21931/RB/2023.08.01.21