2022.07.03.54
Files > Volume 7 > Vol 7 No 3 2022
Immune response to the two doses of mRNA vaccine: A cross-sectional study from a single center located in Wasit province, Iraq
1 Department of Pharmacy, Kut University College, Alkut, Wasit, Iraq,52001,
2 Department of Pharmacy, Kut University College, Alkut, Wasit, Iraq,52001,
3 Department of Pharmacy, Kut University College, Alkut, Wasit, Iraq,52001,
*Correspondence: [email protected] ; Tel.: 009647810108111
Available from: http://dx.doi.org/10.21931/RB/2022.07.03.54
ABSTRACT
The pandemic controlling COVID-19 can cause the deaths of more than 4 million worldwide. Recently the production of the more effective vaccine genetically modified is the suitable solution for managing acute respiratory syndrome, SARS-CoV-2 was a talented emergency use license by FDA by a company of Pfizer-BioNTech (BNT162b2) mRNA the CRP and D dimer important blood markers for diagnosis and severity of this disease. The specific antibody SARS-CoV-2 IgG was the measurement titer after (14-20). After the intake first dose of vaccine by VIDAS® (BioMérieux, Marcy-L'Etoile, France), all volunteers have done screening examination by rapid test for detection not exposure to the previous infection, who had negative to SARS-CoV-2 IgG, after the second dose of vaccine, the specific IgG examination by using ELFA technique. CRP and D-dimer were used to monitor the blood markers to assess any side effects (side effects of the vaccine on the lung inflammatory and clotting system), measured after 48h of taking the vaccine in both doses. Ninety-four were included in this study (54 female and 40 male) and divided into two age groups. All the subjects had SARS-CoV-2 IgG after (10 and 20) days following the first dose and (10 and 20) days following the booster dose. The results were shown a non-significant difference in age between males and females. The mean value of the IgG titer against CoV antigen after 2nd dose of immunization is significantly higher than the corresponding value after the 1st dose. There is a non-significant correlation between the ages of immunized subjects with the IgG titer after receiving the second dose of the mRNA vaccine. The titer of IgG increased after the 2nd dose of vaccination while the C-reactive protein significantly decreased. The Pfizer mRNA vaccine gives an excellent active immune response to produce IgG after the booster dose, and results have not recorded any effect of the vaccine on the activity of blood inflammatory markers.
Keywords: COVID-19; immune response; mRNA vaccine; C-reactive protein, D-dimer, anti-SARS-CoV-2 IgG.
INTRODUCTION
On March 11, 2020, the World Health Organization (WHO) approved that COVID-19 is a pandemic disease. More than 187 million subjects were infected with SARS-CoV-2, and more than 4 million of them died 1; in addition, the number of infected persons and death still increased. Immunization against COVID-19 was introduced to reduce the complications and admissions into the hospitals and the spread of infection 2. The Food and Drug Administration (FDA) and European Medicines Agency [EMA] approved a few numbers of vaccines seeking a temporary solution for controlling the COVID-19 pandemic, by which few manufactured vaccines were considered genetically engineered methods. In contrast, the others used classical methods to trigger and stimulate the immune response against CoV antigen 3–6. The immunoglobulins formed by plasma cells in the body post-vaccination can prevent CoV infection by enhancing phagocytosis, neutralization, opsonizing, and complement activation leading to a reduction of viral load 7,8. IgG and IgM played distinct roles in the viral neutralization in post-infection and post-vaccination with CoV 9-12. mRNA technology-vaccine was introduced worldwide against CoV infection in December 2020 by Pfizer pharmaceutical company 13,14. FDA and EMA approved mRNA vaccine to immunize the adults using two doses of 0.3 mL each, intramuscularly and three weeks apart, and a third booster dose of 0.3 mL is recommended 15.
The nature of the mRNA-CoV vaccine is an RNA-LNP platform of nucleoside-modified RNA, which acts as a sensor of innate immunity. It contained the P2S antigen that can attach to angiotensin-converting enzyme receptor -2 (ACER2), leading to activation of innate immunity to produce the immunoglobulins against viruses. Furthermore, the expression of RNA in the cell allows the activation of CD8 T cell-mediated immunity against CoV 16,17.
C-reactive protein (CRP) is an acute-phase protein marker of systemic inflammation and pathological conditions, e.g., autoimmune diseases and malignancies 18.
The serum levels of CRP are increased in COVID-19 patients, and higher levels are associated with poor outcomes 19,21
The SARS-CoV-2 virus activates the coagulation system. Activating the fibrinolytic pathway will dissolve the blood clots, thereby releasing the fibrin degradation product, D-dimer. 22 Therefore, the D-dimer assessment can serve as a prognostic marker of COVID-19 severity. 23 One study reported that patients with a significantly high D-dimer level are prone to poor outcomes 24,25 Patients of COVID-19 who have a D-dimer level exceeding 1000 ng/ml are at risk of a high mortality rate. 26
This study aims to assess IgG titer as an immune response index, CRP as a biomarker of inflammation, and D-dimer level as an indicator of coagulopathy, following immunization with mRNA coronavirus vaccine in Wasit province, Iraq.
MATERIALS AND METHODS
Ethical approval
The scientific and ethical committees at the Kut University College approved this study and registered it under the term of a cross-sectional study. The participants were informed about the study's objectives before signing the consent form.
Setting and design
This cross-sectional study was carried on in the Zahra Teaching Hospital in cooperation with the Department of Pharmacy at the Al-Kut University College in Wasit, Iraq, from April to October 2021.
Participants
The eligible subjects and patients are both sexes of age ≥ 20 years old. The subjects were recruited from Zahra Teaching Hospital in Wasit province. The inclusion criteria are subjects immunized with two doses of mRNA vaccine (BNT162b2, Pfizer BioNTech, Covid-19 vaccine) and healthy subjects without laboratory and clinical evidence of CoV infection. The criteria for exclusion are subjects with a previous history of CoV infection, any disorders of duration <4 weeks before conducting the study, non-steroidal or steroidal anti-inflammatory drugs, and autoimmune and rheumatic diseases. Ninety-four immunized individuals (54females and 40 males) fulfilled the above criteria and were included in this study.
Laboratory investigations.
A sample of blood was obtained from each patient after 24-48 hours of receiving the second dose of mRNA vaccine for determination of the C-reactive protein and D-dimer and within 10-20 days after the first and second doses of vaccine for determination of immunoglobulin G. The sera and plasma were separated from coagulated and uncoagulated blood (using sodium citrate as an anticoagulant) for determination the C-reactive protein and IgG, and d-dimer respectively. MINI-VIDAS (bioMérieux, France), a compact automated immunoassay system, was used to determine the abovementioned biomarkers. The principle of resolution of these markers is based on the enzyme-linked fluorescent assay. The immunoassay system specified cutoff values of 1 for serum IgG, 6mg/L for C-reactive protein, and 500ng/L for plasma D-dimer.
Statistical analysis
The results are expressed as numbers, percentages, median, and mean± SD. The data were analyzed using the Excel-10 Microsoft software program. The significant differences between immunized and non-immunized individuals were calculated using Chi-square for categorized data, a two-tailed independent-two sample. They paired t-tests for continuous data and Pearson (rho) correlation tests. A p-value of ≤0.05 is the cutoff significant level in two-tailed testing.
RESULTS
This study showed that there is a non-significant difference in age between males and females. Males responded to the mRNA vaccine by increasing the IgG titer, C-reactive protein, and D-dimer as female responders. No significant differences are observed between males and females. Plasma D-dimer level was not detected in 42 vaccinated subjects (<50ng/L), and none of the immunized subjects had a plasma D-Dimer of ≥500ng/L. Thirty patients (31.9 %) of immunized subjects (12 males and 18 females) have serum levels of C-reactive protein of ≥6mg/L. Then the mean value of the IgG titer against CoV antigen after 2nd dose of immunization is significantly higher than the corresponding value after the 1st dose, which amounted to 2.6 folds increment. Table 1.
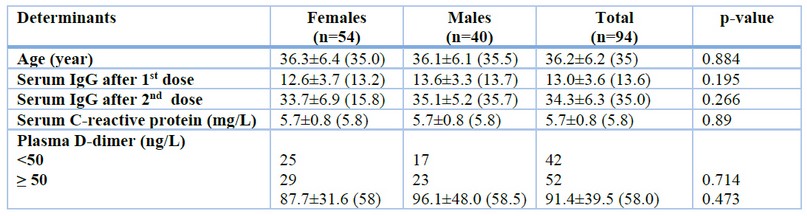
Table 1. Characteristics of the participants
The results are expressed as mean ± SD (median). P-value was calculated using an independent two-sample t-test for continuous data and a Chi-square test for categorized data to compare females with males.
There is a non-significant correlation between the age of immunized subjects with the titer of IgG (r=-0.03, p=0.770, df=92) after receiving the second dose of the mRNA vaccine. An inverse non-significant correlation (r=-0.150, p=0.150, df=92) between the IgG titer and C-reactive protein was observed. The titer of IgG after 1st dose of vaccination significantly and positively correlated with the titer of IgG (r=0.552, p<0.001, df=92) after receiving the 2nd dose of mRNA (Figure-1).

Figure 1. A significant positive correlation (r=0.552, p<0.001) between the titer of immunoglobulin G after 1st dose with after the 2nd dose of vaccination with mRNA.
Moreover, as the titer of IgG increased after the 2nd dose of vaccination, the C-reactive protein significantly decreased (r=-0227, p=0.028, df=92) (Figure 2).

Figure 2. A significant negative correlation (r=-0.227, p=0.028) between the responder's age to the 2nd dose of mRNA vaccine with the serum levels of C-reactive protein.
DISCUSSION
In this study, we will present in Iraq to report limited to a three case series and compassion with many published studies to evaluate immune response post-vaccination in different countries. This study applied by used blood sampling, which for (I) to detect anti-SARS-CoV-2 IgG as monitoring specific immune response to vaccination (II) assessment of active immunity by measurement titer of IgG after first and booster dose (III) monitoring of CRP and D dimer concentration after vaccination as monitoring for blood inflammatory factors and coagulation system activation. This result showed significant relationships between vaccination and non-vaccination (control groups) and significant effects between age and gender in both males and females in vaccination groups.
The age groups in this study will have a present good immune response after taking the first dose 30 μg doses of Pfizer mRNA BNT162b2 could be observed after 10 -20 days, and the same good active immune response after the booster dose after one month for the first dose the titer measurement after 10 -20 days from the second dose.
This study was compacted with published information by Vogel et al. 27 , the active immunity when taking (30 μg) in both doses of Pfizer vaccine, some study was shown that a more significant increase (about ∼3 orders of extender) in IgG could be recorded after 14 of first vaccination dose, whilst stronger stimulatory, recorded after seven days the booster dose. 28 in another different study the measurement of the IgG to 110 individuals taking two Pfizer vaccines and Moderna), the results of this study reported that most individuals developed IgG (∼2 orders greater than baseline) the examination of the sample after nine and twenty days after the first dose, while the titer elevated over three orders of grater between twenty-one and twenty-seven days. 29 After booster dose vaccination, the IgG antibody titer increased to reach 3.5 orders of more excellent comparison with baseline. Interestingly, the IgG titer was more robust in cases of the previous infection by covid19. Another study was published recently; the study referred to both Moderna and Pfizer vaccines giving the same quality of stimulation immune response when measuring all types of immunoglobulin classes, predominantly IgG, to spike protein after 60 days for a booster dose. 30
A recent study was conducted by Britton et al. to evaluate immune response in an elderly individual. By using mRNA vaccination only a single dose, the efficacy is about 63% to prevent infection by COVID-19. 31 When assessment using the single amount of vaccination, which has good results in the older age, which suggest taking the booster dose to give, prolong immune response in emerging spreading of viruses. 32
When the assessment of the previous infection of an individual by Covid 19 after administration first dose, the titer of IgG was highly reached to similarity with an individual who can take two doses of vaccination in about one to three weeks after the booster vaccine and in seventeen days when taking volunteers booster vaccination. 13, 33-35 The safety and 95% effectiveness against Covid-19 were recorded when a two-dose of 30 μg / dose was given in three weeks. 36
Circulating CRP level is determined only depending on the hepatic cell production rate considering the early marker of infection and inflammation and depends on the severity conditions of diseases. 37 The CRP concentration reflects very closely related to the severity of the disease. CRP can be used as prognostic modeling for an assessment of the management of treatment effectiveness. Clinical studies determine the change levels of inflammatory blood markers could be joint to the stage of acuteness and severity add to the mortality rate in COVID‐19 patients. 38
This study was inducted to explain the relationships between the Pfizer vaccine mRNA and the serum concentration of CRP as a blood inflammatory marker after both doses of vaccination; in both cases, the level of CRP was within the normal range with significant differences compared with the control groups. The results were explanations for not recording any vaccine effects on the level of CRP in the vaccination group compared with control groups.
Most studies conducted on patients with COVID-19 recorded a positive relationship between CRP and disease severity.
This study considered CRP biomarkers of disease intensity and outcome. Severe cases were shown with significantly higher CRP levels than less severe cases. 39-41 The meta-analysis study evaluated CRP concentration as a prospective blood marker to assess mortality of COVID-19 speculation, which recorded a high level of CRP. 42
When measured after 19 hours, the CRP is considered a remarkable benefit indicator of activity, extent, and severity of diseases in COVID-19 patients regardless of any pathological change present. 43 The CRP is used to assess specific treatment in the human condition when measured with activated complement. 44,45 In A retrospective study of MERS-CoV patients, the CRP remains at higher serum levels, which were confirmed related to the stage of severity and dead. 46,47 There is no relationship between CRP concentration level with some factors ex. Age, sex, and physical activity. 48 The blood golden stander of concentration of CRP is (< 10 mg/L); while the level higher rapidly after or wins in (6 - 8 h.), and the optimum peak is reached two days from the beginning of the disease. The CRP half‐life (19 h.) and the level reduction after healing accurse. 49
CRP distinguishes phosphocholine and binds with it when highly expressed on surface injury cells. 50 The calibration activates the classical complement pathway by enhancing phagocytic cell activity to clear MO. After the immune system can resolve inflammation and tissue damage, the CRP level falls, making a monitoring disease severity. 51
This study was measurement the concentration of D-dimer in the vaccination group after the first and second dose after 48 hours, the results have not recorded any effect of the vaccine on the concentration of D dimer, and all reading results up to the average engagement. Any change of concentration of coagulation factors, specifically of D-dimer during CONID 19 infection, reflected the prognosis and complications.
The study of TANG et al. found 25 the mean level concentration of D-dimer in dead patients later was significantly higher when contrasted with survivors. In contrast, in the study of WANG et al., 52 D dimer levels persisted in elevated and each patient's death compared to survivors. Add to this, the concentration level of D-dimer level show significantly elevated in severe cases in contrast to non-severe cases. 53,54.
CONCLUSIONS
In this study, we will present in Iraq to report limited to a three-case series with many published studies to evaluate immune response post-vaccination in different countries. The Pfizer mRNA vaccine gives an excellent active immune response to produce IgG. After the booster dose, results have not recorded any vaccine effect on the activity of blood inflammatory markers.
Funding: This research received no external funding.
REFERENCES
1. Chauhan S. Comprehensive review of coronavirus disease 2019 (COVID-19). Biomedical journal. 2020 Aug 1;43(4):334-40.
2. Zhu N, Zhang D, Wang W, et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. The New England journal of medicine. 2020;382(8):727-733.
3. Comirnaty. Summary of Product Characteristics. Available online: https://www.ema.europa.eu/documents/productinformation/ comirnaty-epar-product-information_en.pdf (accessed on March 25 2021).
4. COVID-19 Vaccine Moderna. Summary of Product Characteristics. Available online: https://www.ema.europa.eu/documents/ product-information/covid-19-vaccine-moderna-epar-product-information_en.pdf (accessed on March 25 2021).
5. COVID-19 Vaccine (ChAdOx1-S [Recombinant]). Summary of Product Characteristics. Available online: https://www.ema. europa.eu/documents/product-information/covid-19-vaccine-astrazeneca-epar-product-information_en.pdf (accessed on March 25 2021).
6. COVID-19 Vaccine (Ad26.COV2-S [Recombinant]). Summary of Product Characteristics. Available online: https://www.ema. europa.eu/documents/product-information/covid-19-vaccine-janssen-epar-product-information_en.pdf (accessed on March 25 2021).
7. Plotkin SA, Offit WA, KM PE. Plotkin's Vaccines. ed. (Philadelphia: Elsevier).
8. Dörner T. Radbruch A. Antibodies and B cell memory in viral immunity. Immunity. 2007;27(3):384-92.
9. Wang Z, Lorenzi JC, Muecksch F, Finkin S, Viant C, Gaebler C, Cipolla M, Hoffmann HH, Oliveira TY, Oren DA, Ramos V. Enhanced SARS-CoV-2 neutralization by dimeric IgA. Science translational medicine. 2021 Jan 20;13(577):eabf1555.
10. Klingler J, Weiss S, Itri V, Liu X, Oguntuyo KY, Stevens C, et al. Role of IgM and IgA Antibodies in the Neutralization of SARS-CoV-2. J Infect Dis. 2020.
11. Maeda K, Higashi-Kuwata N, Kinoshita N, Kutsuna S, Tsuchiya K, Hattori SI, Matsuda K, Takamatsu Y, Gatanaga H, Oka S, Sugiyama H. Neutralization of SARS-CoV-2 with IgG from COVID-19-convalescent plasma. Scientific reports. 2021 Mar 10;11(1):1-2.
12. Salazar E, Kuchipudi SV, Christensen PA, Eagar T, Yi X, Zhao P, Jin Z, Long SW, Olsen RJ, Chen J, Castillo B. Convalescent plasma anti–SARS-CoV-2 spike protein ectodomain and receptor-binding domain IgG correlate with virus neutralization. The Journal of clinical investigation. 2020 Dec 1;130(12):6728-38.
13. MJ EO, Juanes de Toledo B. Pfizer-BioNTech, la primera vacuna ARNm contra la COVID-19, parece segura y eficaz. N Engl J Med. 2020, 383:2603-15.
14. Walsh D. COVID-19: A Crisis and an Opportunity to Improve the Emergency Use Authorization Process. Minnesota Journal of Law, Science & Technology. 2021;22(2):169.
15. Emergency use authorization (eua) of the pfizer-biontech covid-19 vaccine to prevent coronavirus disease 2019 (covid-19). https://www.fda.gov/media/144413/download
16. Sahay G, Alakhova DY, Kabanov AV. Endocytosis of nanomedicines. Journal of controlled release. 2010 August 3;145(3):182-95.
17. Gee J, Marquez P, Su J, Calvert GM, Liu R, Myers T, et al. First Month of COVID-19 Vaccine Safety Monitoring - United States, December 14, 2020-January 13, 2021. MMWR Morbidity and Mortality Weekly Report. 2021 February 26;70(8):283-8.
18. Pepys MB, Hirschfield GM. C-reactive protein: a critical update. The Journal of clinical investigation. 2003 Jun 15;111(12):1805-12.
19. Smilowitz NR, Kunichoff D, Garshick M, Shah B, Pillinger M, Hochman JS, Berger JS. C-reactive protein and clinical outcomes in patients with COVID-19. European heart journal. 2021 Jun 14;42(23):2270-9.
20. Bonetti G, Manelli F, Patroni A, Bettinardi A, Borrelli G, Fiordalisi G, Marino A, Menolfi A, Saggini S, Volpi R, Anesi A. Laboratory predictors of death from coronavirus disease 2019 (COVID-19) in the area of Valcamonica, Italy. Clinical Chemistry and Laboratory Medicine (CCLM). 2020 July 1;58(7):1100-5.
21. Yamada T, Wakabayashi M, Yamaji T, Chopra N, Mikami T, Miyashita H, Miyashita S. Value of leukocytosis and elevated C-reactive protein in predicting severe coronavirus 2019 (COVID-19): A systematic review and meta-analysis. Clinica chimica acta. 2020 Oct 1;509:235-43.
22. Adam SS, Key NS, Greenberg CS. D-dimer antigen: current concepts and future prospects. Blood, The Journal of the American Society of Hematology. 2009 Mar 26;113(13):2878-87.
23. Lippi G, Favaloro EJ. D-dimer is associated with the severity of coronavirus disease 2019: A pooled analysis. Thromb Haemost 2020; 120: 876-8.
24. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DS, Du B. Clinical characteristics of 2019 novel coronavirus infection in China. MedRxiv. 2020 Jan 1.
25. Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. Journal of thrombosis and haemostasis. 2020 Apr;18(4):844-7.
26. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The lancet. 2020 Mar 28;395(10229):1054-62.
27. Brouwer PJ, Brinkkemper M, Maisonnasse P, Dereuddre-Bosquet N, Grobben M, Claireaux M, de Gast M, Marlin R, Chesnais V, Diry S, Allen JD. Two-component spike nanoparticle vaccine protects macaques from SARS-CoV-2 infection. Cell. 2021 March 4;184(5):1188-200.
28. Krammer F, Srivastava K, Alshammary H, Amoako AA, Awawda MH, Beach KF, Bermúdez-González MC, Bielak DA, Carreño JM, Chernet RL, Eaker LQ. Antibody responses in seropositive persons after a single dose of SARS-CoV-2 mRNA vaccine. New England Journal of Medicine. 2021 April 8;384(14):1372-4.
29. Wang Z, Schmidt F, Weisblum Y, Muecksch F, Barnes CO, Finkin S, Schaefer-Babajew D, Cipolla M, Gaebler C, Lieberman JA, Oliveira TY. mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants. Nature. 2021 Apr;592(7855):616-22.
30. Britton A, Slifka KM, Edens C, Nanduri SA, Bart SM, Shang N, Harizaj A, Armstrong J, Xu K, Ehrlich HY, Soda E. Effectiveness of the Pfizer-BioNTech COVID-19 vaccine among residents of two skilled nursing facilities experiencing COVID-19 outbreaks—Connecticut, December 2020–February 2021. Morbidity and Mortality Weekly Report. 2021 March 19;70(11):396.
31. Lippi G, Henry BM. How will emerging SARS-CoV-2 variants impact herd immunity?. Annals of translational medicine. 2021 Apr;9(7).
32. Manisty C, Otter AD, Treibel TA, McKnight Á, Altmann DM, Brooks T, Noursadeghi M, Boyton RJ, Semper A, Moon JC. Antibody response to first BNT162b2 dose in previously SARS-CoV-2-infected individuals. The Lancet. 2021 March 20;397(10279):1057-8.
33. Prendecki M, Clarke C, Brown J, Cox A, Gleeson S, Guckian M, Randell P, Dalla Pria A, Lightstone L, Xu XN, Barclay W. Effect of previous SARS-CoV-2 infection on humoral and T-cell responses to single-dose BNT162b2 vaccine. The Lancet. 2021 Mar 27;397(10280):1178-81.
34. Gobbi F, Buonfrate D, Moro L, Rodari P, Piubelli C, Caldrer S, Riccetti S, Sinigaglia A, Barzon L. Antibody response to the BNT162b2 mRNA COVID-19 vaccine in subjects with prior SARS-CoV-2 infection. Viruses. 2021 Mar;13(3):422.
35. Saadat S, Tehrani ZR, Logue J, Newman M, Frieman MB, Harris AD, Sajadi MM. Binding and neutralization antibody titers after a single vaccine dose in health care workers previously infected with SARS-CoV-2. Jama. 2021 Apr 13;325(14):1467-9.
36. Food and Drug Administration. Guidance for industry: emergency use authorization for vaccines to prevent COVID-19. October 2020 (https://www.fda .gov/ media/ 142749/ download).
37. Marnell L, Mold C, Du Clos TW. C-reactive protein: ligands, receptors and role in inflammation. Clinical immunology. 2005 Nov 1;117(2):104-11.
38. Nicholls JM, Poon LL, Lee KC, Ng WF, Lai ST, Leung CY, Chu CM, Hui PK, Mak KL, Lim W, Yan KW. Lung pathology of fatal severe acute respiratory syndrome. The Lancet. 2003 May 24;361(9371):1773-8.
39. Sadeghi-Haddad-Zavareh M, Bayani M, Shokri M, Ebrahimpour S, Babazadeh A, Mehraeen R, Moudi E, Rostami A, Barary M, Hosseini A, Bijani A. C-reactive protein as a prognostic indicator in COVID-19 patients. Interdisciplinary Perspectives on Infectious Diseases. 2021 April 23;2021.
40. Liu F, Li L, Xu M, Wu J, Luo D, Zhu Y, Li B, Song X, Zhou X. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. Journal of clinical virology. 2020 Jun 1;127:104370.
41. Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W, Tian DS. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clinical infectious diseases. 2020 Jul 28;71(15):762-8.
42. Sahu BR, Kampa RK, Padhi A, Panda AK. C-reactive protein: a promising biomarker for poor prognosis in COVID-19 infection. Clinica chimica acta. 2020 October 1;509:91-4.
43. Linkins LA, Takach Lapner S. Review of D‐dimer testing: good, Bad, and Ugly. International Journal of Laboratory Hematology. 2017 May;39:98-103.
44. Matsubara T, Yamakawa K, Umemura Y, Gando S, Ogura H, Shiraishi A, Kushimoto S, Abe T, Tarui T, Hagiwara A, Otomo Y. Significance of plasma fibrinogen level and antithrombin activity in sepsis: a multicenter cohort study using a cubic spline model. Thrombosis Research. 2019 Sep 1;181:17-23.
45. Zhou B, She J, Wang Y, Ma X. Utility of ferritin, procalcitonin, and C-reactive protein in severe patients with 2019 novel coronavirus disease.
46. Gao H, Yao H, Yang S, Li L. From SARS to MERS: evidence and speculation. Frontiers of medicine. 2016 Dec;10(4):377-82.
47. Taneja R, Batra P. Biomarkers as point of care tests (POCT) in neonatal sepsis: A state of science review. Journal of neonatal-perinatal medicine. 2021 Jan 1;14(3):331-8.
48. Bilgir O, Bilgir F, Calan M, Calan OG, Yuksel A. Comparison of pre-and post-levothyroxine high-sensitivity c-reactive protein and fetuin-a levels in subclinical hypothyroidism. Clinics. 2015;70:97-101.
49. Young B, Gleeson M, Cripps AW. C-reactive protein: a critical review. Pathology. 1991 Jan 1;23(2):118-24.
50. Gao Y, Li T, Han M, Li X, Wu D, Xu Y, Zhu Y, Liu Y, Wang X, Wang L. Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19 [published online March 17, 2020]. J Med Virol. doi.;10.
51. Ballou SP, Kushner I. C-reactive protein and the acute phase response. Advances in internal medicine. 1992;37:313-36.
52. Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. Jama. 2020 Mar 17;323(11):1061-9.
53. Park SY, Kim JH, Kim HJ, Seo B, Kwon OY, Chang HS, Kwon HS, Kim TB, Kim H, Park CS, Moon HB. High prevalence of asthma in elderly women: findings from a Korean national health database and adult asthma cohort. Allergy, asthma & immunology research. 2018 July 1;10(4):387-96.
54. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The lancet. 2020 Feb 15;395(10223):497-506.
Received: 3 April 2022 / Accepted: 12 June 2022 / Published:15 August 2022
Citation: Kudhair Abdullabass H, Hamza Al-ShammariA, AL-Zuhairy S A S. Immune response to the two doses of mRNA vaccine: A cross-sectional study from a single center located in Wasit province, Iraq. Revis Bionatura 2022;7(3) 54. http://dx.doi.org/10.21931/RB/2022.07.03.54
