2022.07.02.49
Files > Volume 7 > Vol 7 No 2 2022
2 Department of Biology, College of Science, University of Misan, Misan, Iraq;
* Correspondence: [email protected].
Available from: http://dx.doi.org/10.21931/RB/2022.07.02.49
ABSTRACT
Eucalyptus oil is used in a wide range of medicinal and other uses, and the current study aims to estimate the median lethal dose LD50 of eucalyptus oil of Eucalyptus camaldulensis and estimate the pathological effect of eucalyptus oil in mice. Eucalyptus oil was obtained in the study by extracting the leaves of the plant Eucalyptus camaldulensis using hydrodistillation. Toxicity testing was performed by dividing mice into groups that you treated with different doses of eucalyptus oil 1200, 1600, 2000, and 2400 mg/kg. The current study showed that the average lethal dose, the LD50, was 1820 mg/kg. The study suggested the emergence of clinical signs in mice when consuming eucalyptus oil in high doses, such as slow movement, suffocation, and convulsion, compared with the control group and the group that administered a lower dose, It was observed that high doses of eucalyptus oil could cause damage to the lung, liver, kidneys, and heart, as the lung was the organ most affected by the oil, followed by the liver, kidneys, and then the heart, and it was less affected by eucalyptus oil, and this indicates that eucalyptus oil, when administered orally in high doses, causes clear toxicity effects. Eucalyptus oil is a safe medicine for external use.
Keywords. Eucalyptus oil, LD50; Administrated; Lung; Liver; Heart; Kidney; Mice.
INTRODUCTION
The eucalyptus plant is a tall plant native to Australia and includes nearly 700 species 1. Eucalyptus is a genus belonging to the Myrtaceae family. It is a fragrant evergreen flowering tree cultivated in all regions, including Iraq 2. The common consensus was that all herbal treatments and natural items were safe to use 3, that essential oils are safe for human usage since they are maintained as natural as possible, are risk-free, and have no side effects 4. Essential oils are extracted using various physical and chemical methods, including solvent extraction (hydrodistillation, pressing) 5. Aldehydes, alcohols, ketones, saturated and unsaturated hydrocarbons, terpenes, phenolic oxides, and esters are the components that make up essential oils 6. Essential oils are simple to extract and degrade in the environment, making them environmentally beneficial 7. Essential oils have gained widespread popularity due to their widespread use, and they have been dubbed "aromatherapy." 8. Eucalyptus essential oil has been utilized in the culinary business as a flavoring agent in the food industry 9. Eucalyptus oil is an essential oil derived from the eucalyptus tree that contains 1,8-cineole 10 and α-pinene 11. It's also employed in the fragrance and medicinal industries 12. It is also used to treat respiratory infection; the eucalyptus leaf extract is inhaled 13. Expel sputum in case of chest infection 14 and treat sore throat and cough 15. It has antispasmodic, anti-stress, and antispasmodic properties and relieves labor pains during pregnancy 16. Eucalyptus is used in arthritis, abscess, burns, and diabetes 17. It can also be used as an anti-insecticide to protect people from parasites like head lice 10 and mosquito bites 18, and represents many analgesics, antiinflammatory 19, anti-oxidant 20, and is effective against cancer 21. Eucalyptus oil is used in various methods, including applying it to the skin's surface, massaging it, and inhaling it. It is rarely taken orally 22. This study aimed to measure the LD50 of Eucalyptus oil in mice and determine the organs will be significantly affected by the toxicity of Eucalyptus oil.
MATERIALS AND METHODS
Plant material collection
The leaves of Eucalyptus camaldulensis were collected during October, November 2020, from the governorate of Misan / awash garden.
Extraction Essential Oil
Freshly collected leaves of Eucalyptus camaldulensis were cleaned with distilled water to remove mud and debris from their surfaces, then air-dried in the shade at room temperature and that slice into smaller pieces 23. and the essential oil extraction was carried out by using the hydrodistillation technique Clevenger-type apparatus, the weight of 100 grams of leaves were separately mixed with 500 ml of distilled water in a round one-liter flask 24 as well as being subjected to hydrodistillation Three hours had been spent co-distilling the essential oil with water. N-hexane was used to separate the volatile oils, then stored in bottles in the refrigerator until needed.
ExperimentalAnimals
Male BALB/c mice are aged 6–8 weeks; their weight was from 25 g, pathogen-free; mice were obtained from the animal house of the department of biology of the College of Sciences University Misan. The study included 50 animals and housed 5 animals per cage with food and tap water accessible ad libitum in a 12/12 h light/dark cycle. The local animal ethics commission accepted all experimental techniques, and they all met German standards.
All experimental procedures were approved by the local animal ethics committee and met German and international guidelines. The study was conducted using 50 adult mice; at least 10 animals were used per group. The handling and use of animals were following the institutional guidelines.
Acute toxicity
The experiment was conducted to determine the average lethal dose, LD50, in five groups in each group of 10 animals (n=10/group) of male mice; the following control group was administered normal saline. The animals were administered eucalyptus oil once each time. If the animal survived from the first dose, the next dose animal would be increased. The next dose is reduced if the animal died from the first dose; the animals fasted for three hours before receiving the eucalyptus oil. One hour after receiving the eucalyptus oil, The four groups were administered by oral gavage using eucalyptus oil at doses of 1200, 1600, 2000, 2400 mg/kg of body weight; the dose was calculated by µl, and the average weight was 25g, animal administered was continued for 14 days. Neurobehavioral and involuntary effects were observed in animals for 24 hours and registration of deaths and survival. The toxicological effect was evaluated according to the mortality rate expressed by LD50, expired animals were counted for the calculation of LD50 (Table 1) . In its arithmetic method, Karber has used the determination of LD50 According to the following equation. 25
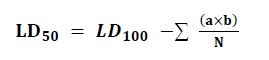
LD100
N = total number of animals in a group
a = the difference between two successive doses administered.
b = the average number of dead animals in two successive doses(Table 2)
Histological study
Euthanasia to mice by placing 1 ml of chloroform (CHCl3) on cotton and then placing it on the animal's nose 26 after animal dissection, the lung, heart, liver, and kidneys were collected from all mice and washed with standard salt solution and then fixation 27 in a neutral buffer formalin (NBF) solution 10% after that fixation process is washed with running water and then dehydration through the concentrations of alcohol. There's the clearing, infiltrating, embedding, and sectioning in 5 µm thick sections, then staining with hematoxylin and eosin, finally examining the tissue sections under a microscope with a force of magnification 400X.
RESULTS
Clinical signs of acute toxicity
After administering the animals with eucalyptus oil for 10 minutes, mice that were administered a high dose of 2,400 and 2,000 mg/kg of eucalyptus oil developed toxic symptoms in terms of convulsion, dizziness, slow movement, and high breathing (suffocation), as compared with the control group as shown in (Table 2). As for the mice in the lower dose groups of 1200 mg/kg of eucalyptus oil, they did not show toxic symptoms, with good health, and no deaths recorded was, as shown in Table 2, and consequently, the body weight was constantly growing, and food consumed, unlike animals that were given a high dose. The The karber method calculated the value at 1820 mg/kg, as shown in (Table 3). Mortality was recorded only at 1600,2000and 2400mg/kg body weight doses, three mice out of ten animals had died in the group (2) that received 1600mg/kg body weight test dose, was died overnight following the administration, and seven mice out of ten animals in group( 3) died within 24 hours that received 2000mg/kg body weight test does, and nine mice out of ten animals in group (4) died within 24 hours that received 2400mg/kg body weight test dose, was as showed in (Table 2).
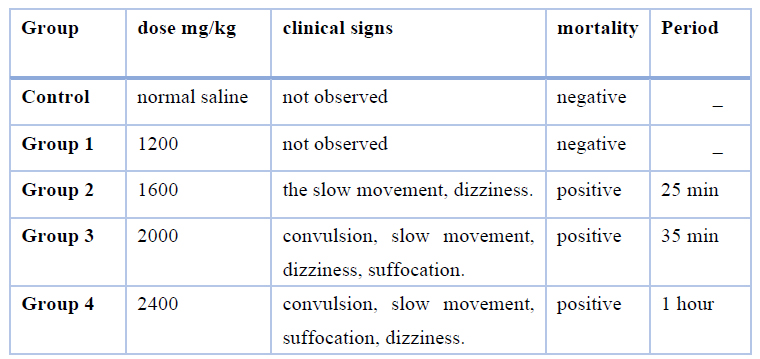
Table 1. Results of observed clinical signs.
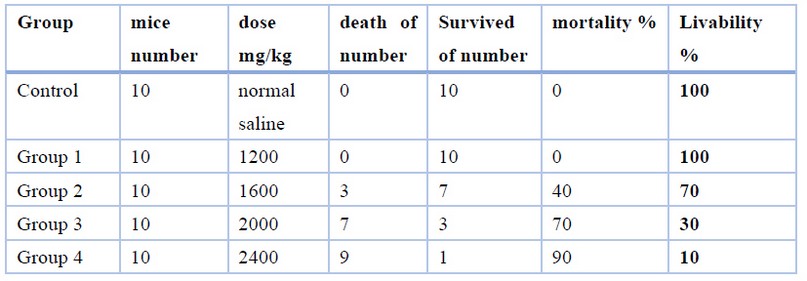
Table 2. The result of the oral acute toxicity test of eucalyptus oil in mice
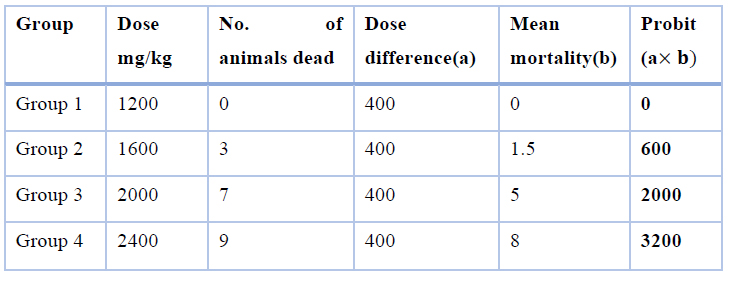
Table 3. Calculation of by the method of karber
Sum of the product = 5800
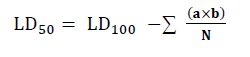
=2400-580
=1820 mg/Kg
Results of Histological changes
The results of this study indicate the occurrence of tissue changes in the organs ( lung, heart, liver, and kidneys) as a result of administering male mice to different doses of eucalyptus oil, and the pathological changes were as follows.
Histopathological changes in the liver
Sections obtained from the livers of mice for the control group clarified that sinusoids, hepatocytes are large cuboidal or polyhedral epithelial cells with large round central nuclei and eosinophilic cytoplasm, bile ducts, central vein, portal vein, hepatic artery, and immune cells within sinusoid called Kupffer cells (Figure: 1A). The cross-section of liver mice in the group treated with Eucalyptus oil toxic dose shows congestion in the central vein; small vacuoles were shown in hepatocytes, Focal necrosis, pyknosis, narrowing of the sinusoids (Figure:1 B).
Histopathological changes in the heart
A cross-section of heart mice shows the control group's normal appearance: cardiac muscle cells, nuclei of the cardiac muscle cells, intercalated disk, and branched cardiac muscle (Figure: 2 A). The cross-section of heart mice in the group treated with eucalyptus oil showed congestion, cytoplasmic vacuolization of cardiac muscle cells (v), and disarrangement of fibers (Figure:2 B).
Histopathological changes in the kidney
In the kidney mice sections of the control group appeared the renal capsule and renal tubular epithelial cell (RTEC), Bowman's space, Glomerulus, Distal convoluted tubule, Proximal convoluted tubule, Macula dense (Figure: 3 A). The cross-section of kidney mice in the group treated with eucalyptus oil is toxic does show congestion or hyperemia, granular degeneration, widening of the tubular lumen was observed in sections of the kidney, pyknosis of renal tubular epithelial cells, increased the area of bowman's space, desquamation of the epithelial cells of the renal tubules, and perivascular lymphocytic infiltrations were also observed in small areas of the kidney (Figure: 3 B).
Histopathological changes in the lung
The histological structure of mice lungs in the control group appeared to be the alveoli, alveolar sac, bronchioles, the lumen of a bronchus, blood vessels, and the alveolar epithelium (Figure: 4 A). Microscopic examination of the lungs of mice treated with eucalyptus oil showed different tissue changes; the changes represented the occurrence of emphysema, severe congestion in the blood vessels; also the expansion of the blood vessel, as well as the emergence of edema and hyperplasia of the epithelium lining the bronchi, which led to hemorrhage in the interstitial tissue of the lung, the degeneration of the alveoli, destruction of alveolar structures. The epithelial cells in this bronchiole are sloughing into the lumen and have lost their cilia, enlargement of the alveolar airspaces distal to the terminal bronchioles, and loss of elasticity of the lung tissue (Figure:4 B).
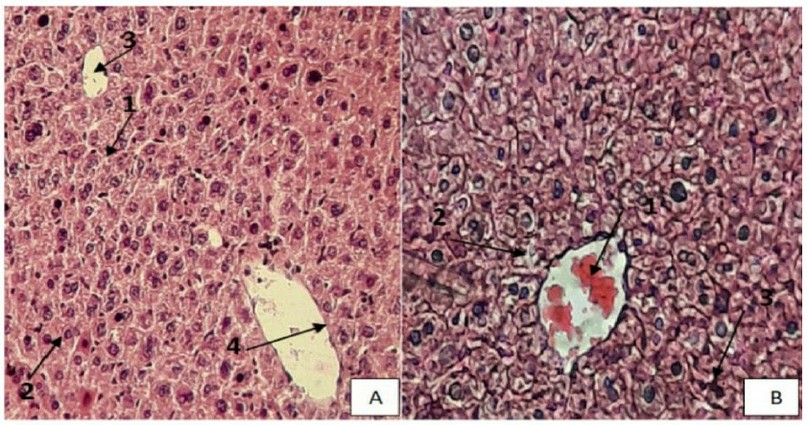
Figure 1. (A). section of the control group of liver mice showing the normal structure of the liver, Sinusoids (1), Hepatocytes (2), Central vein(3), Endothelial cells (4), (H & E stain, 400X). (B) section of liver mice treated by 2400 mg /kg showing, Congestion central venous (1) , degeneration hepatocytes (2) , Focal necrosis(3) , (H & E stain, 400X).
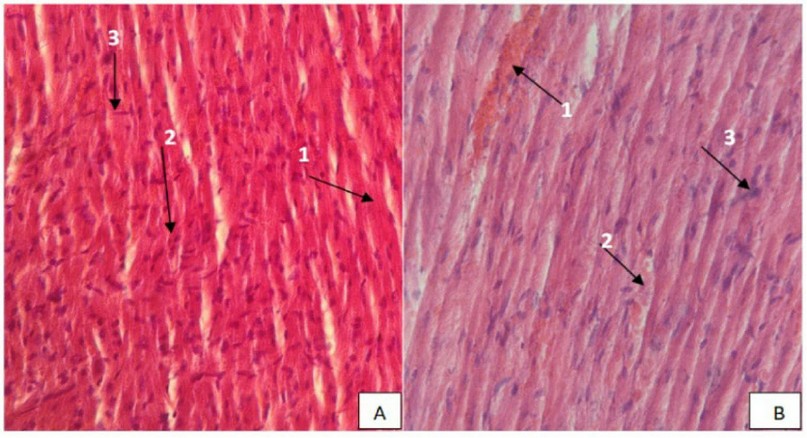
Figure 2. A Section of the control group of Heart mice showing the typical structure of the Heart, cardiac muscle cells(1), nuclei of the cardiac muscle cells(2), intercalated disk(3), (H & E stain, 400X). (B). Section of Heart mice treated by 2400 mg /kg showing hyperemia(1), coagulation necrosis(2), formation of a dense collagenous scar(3), (H & E stain, 400X).
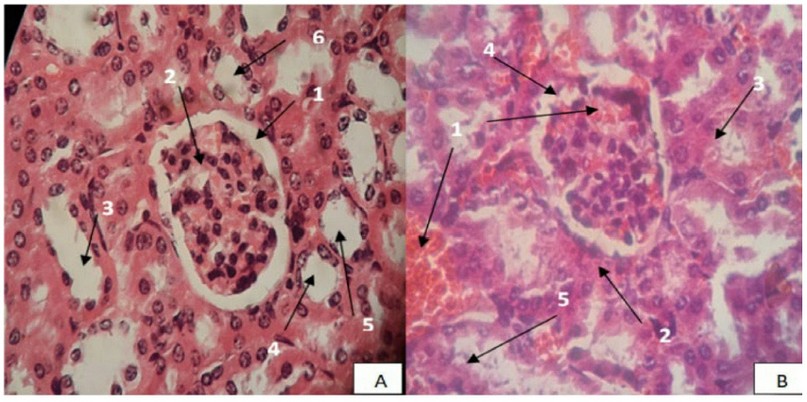
Figure 3. (A). Section of the control group of Kidney mice showing the typical structure of Kidney, Bowman's space (1), Glomerulus (2), Collection duct(3), Distal convoluted tubule(4), Proximal convoluted tubule(5), Macula dense(6), (H & E stain, 400X). (B) section of Kidney mice treated by 2400 mg /kg showing hyperemia (1), granular degeneration (2), pyknosis of renal tubular epithelial cells (3), lobulated renal capsules (4), desquamation epithelial cells(5) (H & E stain, 400X).
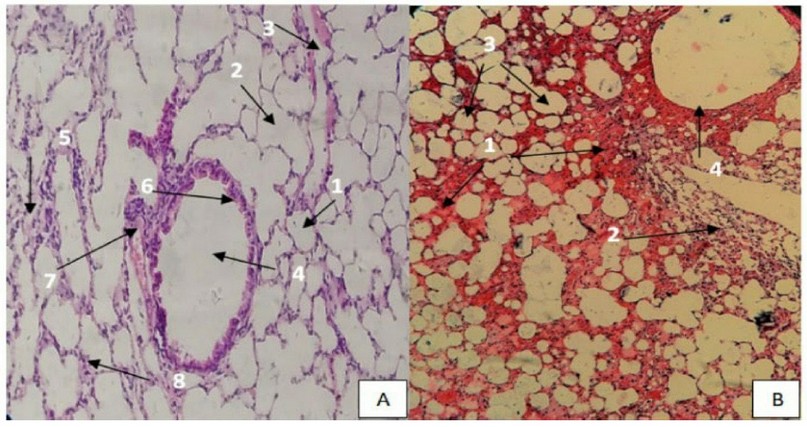
Figure 4. (A). Section of the control group of Lung mice showing the standard structure of Lung, Alveoli(1), alveolar sac(2), alveolar duct(3), bronchiole(4), smooth muscle(5), club cells(6), blood vessels(7), the alveolar epithelium(8) (H & E stain, 400X). (B). section of Lung mice treated by 2400 mg /kg showing hyperemia(1), destruction of alveolar structures (2), expansion alveoli(3), epithelial cell bronchiole sloughing (4), (H & E stain, 400X).
DISCUSSION
Little is known regarding the toxic quality of eucalyptus oil; however, they have been separated and labeled as GRAS by USEPA 28; due to the potential health risk, eucalyptus oil requires a quality assessment for its effectiveness and safety due to its growing demand for drugs.
Clinical signs of eucalyptus oil toxicity developed when administered at 2400 and 2000 mg/kg, such as convulsions, choking, slow motion, dizziness, and not consuming food and water (Table 1), and this is consistent with report 29, which talks about the appearance of toxic effects such as insomnia, followed by fatigue and reduction of food and water consumption, as well as the similar result with the report 30, which speak of the appearance of toxic symptoms in rats, such as slow movements and convulsions. The cause of death is respiratory failure.
The study's objective was to assess the toxicity and safety of eucalyptus oil using oral toxicity in mice. In this study, the acute toxicity was shown where the value was 1820 mg / kg (Table 3). The value calculated during this study corresponds to report 31, which addresses the acute toxicity of eucalyptus oil in mice because the value is 1824.01 mg/kg. The output also disagreed with report 30, where the LD50 eucalyptus oil emulsion derived from water and oil was 3500 mg/kg.
The liver is a significant site for plasma protein synthesis, and any strain attached to the liver produces high levels of AST and ALT 32. This is the first sign of damage to liver cells and the presence of tiredness, necrosis, toxicosis, or biliary liver disease 33
Histopathologic changes have occurred mainly in the liver, heart, lungs, and kidneys. The toxic dose has been shown with eucalyptus oil in focal hepatic necrosis, pyknosis, vacuolar degeneration, and distorted general architecture (Figure 1). Pyknoses occur when a cell receives an apoptosis signal 34. The presence of pyknosis and protoplasmic changes such as vacuolation in swollen hepatocytes may indicate possible morphological signs of necrosis or degeneration, The distorted general hepatic architecture in the eucalyptus oil treatment experimental group was probably due to abnormal changes in hepatocytes. Abnormal changes in hepatocytic structure also alter the overall microscopic architecture of the liver, including the eradication of specific sinusoidal spaces 35, caused by eucalyptus oil the congestion of blood vessels into the area of the door associated with inflammatory infiltration. Also, small vacuolar were found in hepatocytes. Our outcomes are consistent with those achieved by 36. This study identified an increase in the number of Kupffer cells in the liver.
In terms of histopathological examination of mice kidney tissue shown in (Figure 3) showed dilated congested renal vein surrounded by infiltrated renal glomeruli and renal tubules and aggregation of inflammatory cells; the results of the present study on the mice kidney showed hyperemia in the Glomerulus, the epithelial cells lining the renal tubules, this result is similar to the report 5, these tubular-related changes could contribute to the increased concentration of potentially toxic essential oil components in the urine filtrate 37, The lobulated renal corpuscles and desquamation of the epithelial cells of the renal tubules were seen in the kidneys of Eucalyptus oil-treated mice (Figure 3). In a similar trend, the data was obtained by 36 peritubular and peri corpuscular mononuclear cellular infiltration into the kidneys. Previously, eucalyptus oil therapy was suggested to enhance prostaglandin synthesis, which is a critical factor in inflammation, apoptosis, and cell growth processes 38.
Eucalyptus oil in the lung of mice led to a state of alveolar emphysema, which is known as obstruction of the small respiratory tracts, which causes pressure on the alveolar walls and their rupture. Pathological alterations are congestion of the pulmonary vessels (Figure 4), that the results of this study are compatible with what he found 39, where has arisen congestion of blood vessels in the lungs of mice, loss of normal lung architecture with partial obliteration of bronchiole light by excretion of epithelial cells or occasionally causing complete bronchial deformation 40, that the inflow of inflammatory cells into the interstitial and broncho-alveolar space is considered an essential role in the progression of lung injury 41.
Our histopathological analysis revealed that this structural change induced by eucalyptus oil was characterized by lymphoid infiltration around blood vessels, perinuclear vacuolation of cardiac muscle cells (Figure 2), and interfibrillar hemorrhaging, and misarrangement of myocardial beams; this result was similar to report 42.
The organs affected by eucalyptus oil toxicity include the liver, kidneys, lungs, and heart, where the organs most affected by eucalyptus oil are the lungs, and the least affected is the heart. This result is inconsistent with report 29, which talks about the lack of side effects of eucalyptus oil emulsion on the cardiovascular and respiratory systems.
CONCLUSIONS
It can be concluded from the results of the current study that the value of eucalyptus oil for Eucalyptus camaldulensis was 1820 mg/kg, and it produces a severe toxic effect after oral administration in doses of up to 2400 mg/kg, and the high dose causes clinical signs such as slow movement, convulsion, suffocation, and target organs are lung, liver, kidney, and heart. It depends on the dose and the results of this study. Eucalyptus oil is a safe drug for external use.
Author Contributions: Conceptualization, AKA and ZMJ; methodology, AKA; software, ZMJ; validation, ZMJ and AKA; formal analysis, ZMJ; investigation, AKA; resources, ZMJ; data curation, AKA; writing—original draft preparation, ZMJ; writing review and editing, AKA; visualization, ZMJ; supervision, AKA; project administration, AKA. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding
Institutional Review Board Statement: All experimental procedures were approved by the local animal ethics committee in the department of biology of the College of Sciences University Misan and met German and international guidelines (30/8/2020). The study follows the rules of scientific research at Misan University, Iraq.
Conflicts of Interest: The authors declare no conflicts of interest.
REFERENCES
1.Aglal A, Nwara A, Ali A. Toxicity of aqueous extract of Eucalyptus in swiss albino. Suez Canal Veterinary Medical Journal. 2010; 1: 199-214.
2. Hur J, Ahn S, Koh Y, and Lee C. Antimicrobial properties of cold-tolerant Eucalyptus species against phytopathogenic fungi and food-borne bacterial pathogens. The Plant Pathology Journal. 2000; 16.5: 286-289.
3. Radulović N, Randjelović P, Stojanović N, Blagojević P, Stojanović-Radić Z, Ilić I et al. Toxic essential oils. Part II: Chemical, toxicological, pharmacological, and microbiological profiles of Artemisia annua L. volatiles. Food and Chemical Toxicology. 2013; 58:37-49.
4. Vostinaru O, Heghes S, and Filip, L. Safety profile of essential oils. In: Essential Oils-Bioactive Compounds, New Perspectives, and Applications. London, UK: IntechOpen. 2020; p. 1-13.
5. Stojanović N, Randjelović P, Mladenović M, Ilić I, Petrović V, Stojiljković N et al. Toxic essential oils, part VI: Acute oral toxicity of lemon balm (Melissa officinalis L.) essential oil in BALB/c mice. Food and Chemical Toxicology. 2019;133:110794.
6. Ali B, Al-Wabel N, Shams S, Ahamad A, Khan S, Anwar F. Essential oils used in aromatherapy: A systemic review. Asian Pacific Journal of Tropical Biomedicine. 2015;5(8):601-611.
7. Hu Z, Feng R, Xiang F, Song X, Yin Z, Zhang C, Shi F et al. Acute and subchronic toxicity as well as evaluation of safety pharmacology of eucalyptus oil-water emulsions. International Journal of Clinical and Experimental Medicine. 2014; 7.12: 4835.
8. Esposito E, Bystrek M, and Klein J. An elective course in aromatherapy science. American Journal of Pharmaceutical Education. 2014; 78.4.
9. Buchbauer G.The detailed analysis of essential oils leads to the understanding of their properties. Chemical Weekly-Bombay-. 2000; 45.49: 163-165.
10. Yang Y, Choi H, Choi W, Clark J, Ahn Y. Ovicidal and Adulticidal Activity of Eucalyptus globulus Leaf Oil Terpenoids against Pediculus humanus capitis (Anoplura: Pediculidae). Journal of Agricultural and Food Chemistry. 2004;52(9):2507-2511.
11. Sartorelli P, Marquioreto A, Amaral-Baroli A, Lima M, Moreno P. Chemical composition and antimicrobial activity of the essential oils from two species of eucalyptus. Phytotherapy Research. 2007;21(3):231-233.
12. Brooker M, and Kleinig D. Field Guide to Eucalyptus. Third edition. Melbourne, South-eastern Australia: Bloomings. 2006; vol.1.
13. Yohannes, et al. phytochemical screening, Anthelmintic activity of eucalyptus globules essential oils: world journal of pharmacy and pharmaceutical sciences. 2014; 4, 52-60.
14. Maghsoodlou M, Kazemipoor N, Valizadeh J, Seifi M, and Rahneshan N. Essential oil composition of Eucalyptus microtheca and Eucalyptus viminalis. Avicenna journal of Phytomedicine. 2015; 5.6: 540.
15. Kheder D, Al-Habib O, Gilardoni G, Vidari G. Components of Volatile Fractions from Eucalyptus camaldulensis Leaves from Iraqi–Kurdistan and Their Potent Spasmolytic Effects. Molecules. 2020;25(4):804.
16. Perry, Nicolette; Perry, Elaine. Aromatherapy in the management of psychiatric disorders. CNS drugs. 2006;20.4: 257-280.
17. Oyesina T, Salihu M, and Adedayo D. Effect of essential oil of the leaves of Eucalyptus globulus on heamatological parameters of wistar rats. African Journal of Biochemistry Research. 2012;6(4).
18. Kuehn B. new repellents for West Nile fight. JAMA. 2005; 293.21: 2583-2583.
19. Li H, Huang L, Zhou A, Li X, and Sun J. Study on antiinflammatory effect of different chemotype of Cinnamomum camphora on rat arthritis model induced by Freund's adjuvant. Zhongguo Zhong yao za zhi= Zhongguo Zhongyao Zazhi= China Journal of Chinese Materia Medica. 2009;34.24: 3251-3254.
20. Bayala B, Bassole I, Gnoula C, Nebie R, Yonli A, Morel L et al. Chemical Composition, Antioxidant, Antiinflammatory and Anti-Proliferative Activities of Essential Oils of Plants from Burkina Faso. PLoS ONE. 2014;9(3):e92122.
21. Su Y, Li Q, Yao C, Lu Y, and Hong J. Antitumor action of ethanolic extractives from camphor leaves. Chemical Industry and Engineering Progress. 2006; 25.2: 200.
22. Dunning T. Aromatherapy: overview, safety and quality issues. OA Altern Med. 2013; 1.1: 6.
23. Abed K, and Naife T. Extraction of Essential Oil from Iraqi Eucalyptus camadulensis leaves by water distillation methods. In: IOP Conference Series: Materials Science and Engineering. IOP Publishing. 2018; p. 012163.
24. Shalaby S, El-Din M, Abo-Donia S, Mettwally M, and Attia Z. Toxicological affects of essential oils from Eucalyptus Eucalyptus globules and clove Eugenia caryophyllus on albino rats. Pol. J. Environ. Stud. 2011; 20.2: 429-434.
25. Karber G. Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche. Naunyn-Schmiedebergs Archiv für experimentelle pathologie und pharmakologie. 1931; 162.4: 480-483.
26. Blackshaw J, Fenwick D, Beattie A, Allan D. The behaviour of chickens, mice and rats during euthanasia with chloroform, carbon dioxide and ether. Laboratory Animals. 1988;22(1):67-75.
27. LUNA L. Manual of histologic staining methods of the Armed Forces Institute of Pathology. 1968.
28. Batish D, Singh H, Kohli R, Kaur S. Eucalyptus essential oil as a natural pesticide. Forest Ecology and Management. 2008;256(12):2166-2174.
29. Xu J, Hu Z, Wang C, Yin Z, Wei Q, Zhou L, and Yin L. Acute and subacute toxicity study of 1, 8-cineole in mice. International journal of clinical and experimental pathology. 2014; 7.4: 1495.
30. Gebremickael A, Gebru G, Debella A, Addis G. Acute and Sub-chronic Oral Toxicity Evaluation of Eucalyptus globulus Essential Oil-Water Emulsion in Mice. Journal of Cytology and Histology. 2017;08(02).
31. Yu Y, Wang C, Feng G et al. Acute toxicity and genetic toxicity of eucalyptus oil. J Toxicol. 2010; 24: 501-503.
32. Arise R, Malomo S, Adebayo J, and Igunnu A. Effects of aqueous extract of Eucalyptus globulus on lipid peroxidation and selected enzymes of rat liver. Journal of Medicinal Plants Research. 2009; 3.2: 077-081.
33. Hilaly J, Israili Z, Lyoussi B. Acute and chronic toxicological studies of Ajuga iva in experimental animals. Journal of Ethnopharmacology. 2004;91(1):43-50.
34. Berghe T, Vanlangenakker N, Parthoens E, Deckers W, Devos M, Festjens N et al. Necroptosis, necrosis and secondary necrosis converge on similar cellular disintegration features. Cell Death and Differentiation. 2009;17(6):922-930.
35. Ebaid H, Dkhil M, Danfour M, Tohamy A, Gabry M. Piroxicam-induced hepatic and renal histopathological changes in mice. Libyan Journal of Medicine. 2007;2(2):82-89.
36. Abdel-Gawad A, EL-Mahrouky F and Khidr F. Histological effects of camphor leave ethanol extract in some organs of the house sparrow and palm dove. Journal of Agricultural Science, Mansoura University, Egypt. 2001;26, (10), 6451.
37. McQueen C. Comprehensive Toxicology, 2nd ed. Elsevier Science and Technology, Oxford, UK.2010; pp 6448.
38. Martínez B, Pérez-Castillo A, Santos A. The mitochondrial respiratory complex I is a target for 15-deoxy-Δ12,14-prostaglandin J2 action. Journal of Lipid Research. 2005;46(4):736-743.
39. Gupta N, Su X, Popov B, Lee J, Serikov V, Matthay M. Intrapulmonary Delivery of Bone Marrow-Derived Mesenchymal Stem Cells Improves Survival and Attenuates Endotoxin-Induced Acute Lung Injury in Mice. The Journal of Immunology. 2007;179(3):1855-1863.
40. Barker A, Bergeron A, Rom W, Hertz M. Obliterative Bronchiolitis. New England Journal of Medicine. 2014;370(19):1820-1828.
41. Grommes J, Soehnletn O. Contribution of neutrophils to acute lung injury. Molecular medicine. 2011; 17.3: 293-307.
42. Amara I, Timoumi R, Annabi E, Neffati F, Najjar M, Bouaziz C et al. Di (2-ethylhexyl) phthalate induces cardiac disorders in BALB/c mice. Environmental Science and Pollution Research. 2019;26(8):7540-7549.
Received: 29 January 2022 / Accepted: 17 March 2022 / Published:15 May 2022
Citation. Zainab Muhsan A, Ali Khalaf A. Histological Study for Median lethal Dose (LD50) of Eucalyptus Oil Administrated Orally in ( Mice mus musculus). Revis Bionatura 2022;7(2) 49. http://dx.doi.org/10.21931/RB/2022.07.02.49
