2022.07.01.32
Files > Volume 7 > Vol 7 No 1 2022
Recent advances on the development of a universal blood type
1 Yachay Tech University, School of Biological Sciences and engineering, Hda. San José s/n y Proyecto Yachay, 100119, Urcuquí, Ecuador ; [email protected]
2 Yachay Tech University, School of Biological Sciences and engineering, Hda. San José s/n y Proyecto Yachay, 100119, Urcuquí, Ecuador ; [email protected] https://orcid.org/0000-0002-8767-4280
* Correspondence: [email protected]; Tel.: +593984444486
Available from: http://dx.doi.org/10.21931/RB/2022.07.01.32
ABSTRACT
Blood transfusion is the most common form of tissue transplant, and correct blood type matching is decisive for the success of this procedure. However, the availability of different blood types depends on each population, leading to a shortage of blood units from uncommon types. Then, it represents a problem for patients who need a blood transfusion because the supply for their blood type is scarce. Fortunately, researchers have been developing different techniques to engineer universal red blood cells (RBC) that could be transplanted to any human being independent of its blood type. This paper details the main features of blood transfusion and red blood cells maintenance and the two more recent procedures used to produce universal RBCs, the Enzymatically Converted Group O (ECO) and the antigen masking methods.
Keywords. Blood, transfusion, red blood cells, antigen, antibody, universal RBC, immunity
INTRODUCTION
One of the current issues in medicine is blood transfusion, it is the most common form of tissue transplant, and blood type matching is the most challenging point to achieve.1 Besides, an incorrect blood transfusion may induce a severe immune response. If the blood given during a transfusion is incompatible, the donor cells are treated as foreign, and the immune system attacks.2 Nowadays, several patients in hospitals are waiting for the availability of blood units. Therefore, it is essential to develop a new generation of blood transfusion systems to overcome the current issues on blood transplants. The most determinant solution is to develop a universal blood type.
Alloimmunity refers to the immune response against non-self-antigens from members of the same species. It represents an obstacle for blood transfusions because different blood types provoke immune responses in the host. Blood transfusions may induce two reactions, immunosuppression and immune activation.3 To overcome this, scientists worldwide are working to develop a universal blood type to overcome the current problems on immunity. Some researchers, as Yueqi Zhao1 et al., have modified the surface of RBCs, covering them with a hydrogel framework to "hide" their antigens and therefore prevent an immune response.
One of the antigens to produce alloimmunity is the Rhesus D factor (RhD), a protein found on the erythrocyte's surface and considered one of the main immunogenic antigens on red blood cells.1 Moreover, the RhD protein is part of the Rh group, the second most important blood group system, after the ABO blood group. Consequently, the variety of blood groups leads to an unequal supply of RBCs units, which becomes worse for those uncommon blood groups as AB-. Since then, an increasing need for the positive-to-negative transition of the RhD antigen has emerged to make blood transfusions more successful.
Blood: Definition and Components
Blood accounts for about 8 to 10% of each person's body mass.4 Throughout its path, it satisfies several functions such as: mainly transporting oxygen and nutrients, it protects the body due to the leukocytes present in it, keeping the body's water and body temperature in balance, and it also collects the residues to then be deleted.4,5 Blood is composed of parts, mostly the liquid part called plasma, approximately 55% of the total blood, and it is much thicker than water and has a yellowish tone. The remaining percentage is 45%, and it is distributed in different blood cells such as red blood cells, which are the most numerous in the blood. RBCs represent approximately 43%, and the other remaining part is white blood cells and platelets by 2%.6,5
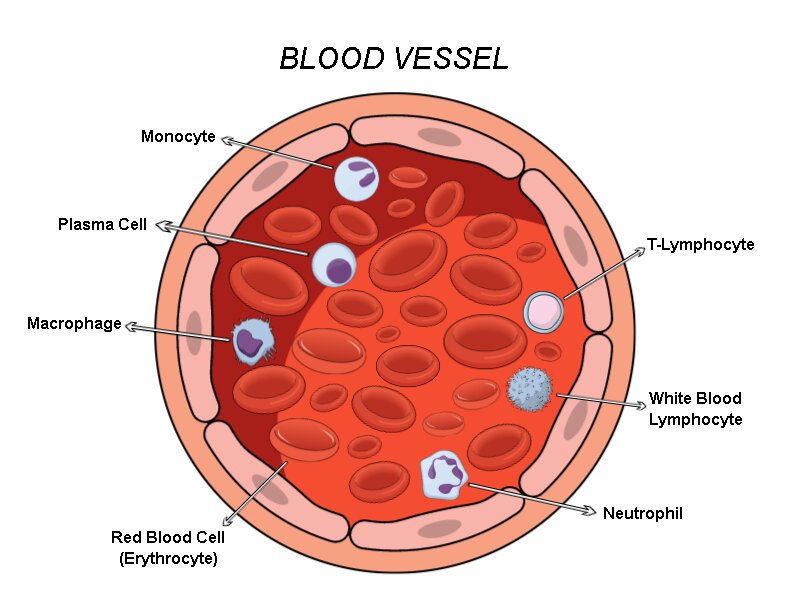
Figure 1. Blood cells composition inside a blood vessel. Inside a blood vessel, there are different types of cells such as the RBCs and immune cells like monocytes, plasma cells, macrophages, neutrophils, white blood lymphocytes, and T-Lymphocytes. In addition to some others, these different types of cells help the circulatory system avoid infections and distribute all the nutrients and molecules the body needs.
Blood Types.
There are four blood types, and they are classified according to the antigens they possess (AB0 system). But in addition to this classification, blood possesses another critical factor for determining its type, the Rh factor. The antigens are present on the surface of the RBCs, and there are two types, A and B.6,4 Moreover, two types of antibodies react with the proteins present on the RBCs, the Anti-A and Anti-B.6 On the other hand, we have the Rh factor, this hereditary protein is also found on the surface of the RBCs,5 and its classification is general; it is positive if it possesses the protein and negative if it does not have. These two types of classifications are the most common in terms of blood types, and those are the ones we will go aboard in this paper
Type A
This group has the presence of antigen A in its red blood cells. Also, the presence of the Anti-B antibody is observed in its plasma (Figure 2), which means that it cannot get the blood of any other blood type.6 Since your blood contains marker A; it will produce B antibodies. If some problem B markers enter your body (these markers are found in the AB or B blood), your immune system will react against them and attack them. This means you can only get transfusions from people who have type A or O blood, not people with type B or AB blood.6
Type B
This blood group has antigen B in its red blood cells, and the presence of the Anti-A antibody is observed in its plasma (Figure 2).7 If your blood contains marker B, it will produce antibodies A. Therefore, as a person with type B blood, you can only receive transfusions from people who have type B or O blood (universal donor), not from people with type A or AB blood, because if another type of marker enters your body, your immune system will attack and there would be problems.7
Type AB
The AB group has the presence of the two antigens in their red blood cells, both A and B, and it has no antibodies in its plasma (Figure 2). This means that it can receive blood from almost all blood types.8 In the case of blood type AB, things are a bit different. If you have both markers A and B on the surface of your red blood cells, your body won't need to fight the presence of either marker. This implies that people with AB blood can receive transfusions from A, B, AB or O blood. However, it also depended on the Rh factor of each one. 7,8
Type O
It is the universal donor, as its blood type is compatible with almost all other types. It is possible because they do not possess any antigens from the AB0 system. However, Anti-A and Anti-B antibodies are observed in their plasma (Figure 2).6,8 However, if you have O-type blood, your red blood cells do not contain A-markers or B-markers, but your body will react by making A and B antibodies when you are given A, B, or AB blood. This means that a person with blood O can only receive transfusions of the same blood type. 8
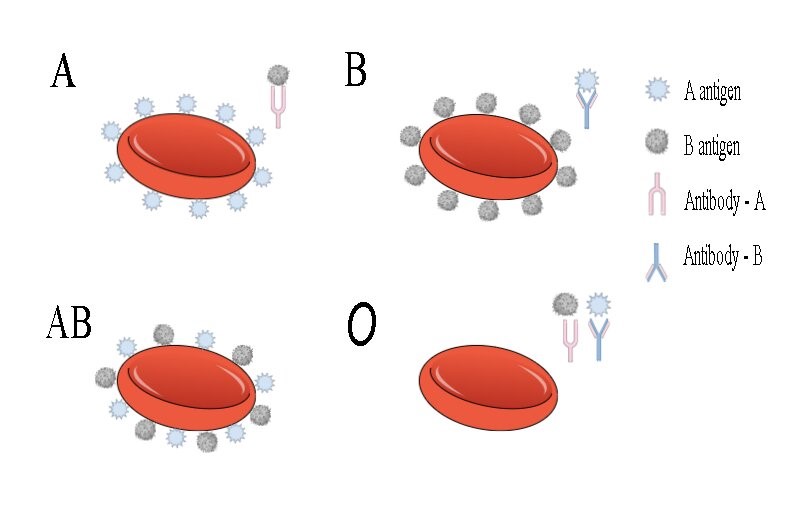
Figure 2. Illustration of the different blood groups of the ABO system. We can see the blood types A, B, AB, and O with their antigens and correspondent antibodies. In the first case, type A has a marker A, and it will produce B antibodies. In the second case, type B has a marker B, it will produce A antibodies- In the third case, type AB has markers A and B, it will not produce antibodies. And the last case, type O has no markers but will produce antigens A and B.
Rh Factor
Type Rh positive
Rhesus factor, commonly known as the RH factor for its acronym, is a protein or antigen inherited from parents and occurs in the red blood cells of certain people. Rh+ are the individuals who present this protein in their erythrocytes. About 85% of people have this protein.4 As shown in Figure 3, the illustration shows that the general population maintains an Rh+; however, a small population maintains an Rh-, so it is important to perform tests for this factor if you plan a pregnancy. 4
Type Rh-negative
The Rh– people do not present the protein in their red blood cells; the figures are very low for this protein. Only about 15% of the world's population possesses this protein (Figure 3).5 However, the lack of this protein is not a disease, and it usually does not affect a person's health.6 RH- is not a disease, but it does affect pregnancy. Pregnancy needs special care if the baby inherits the Rh factor different from that of its mother because it can be inherited from either parent. Rh incompatibility is generated if they are Rh-negative and their baby is Rh-positive, and they need special treatment. 6
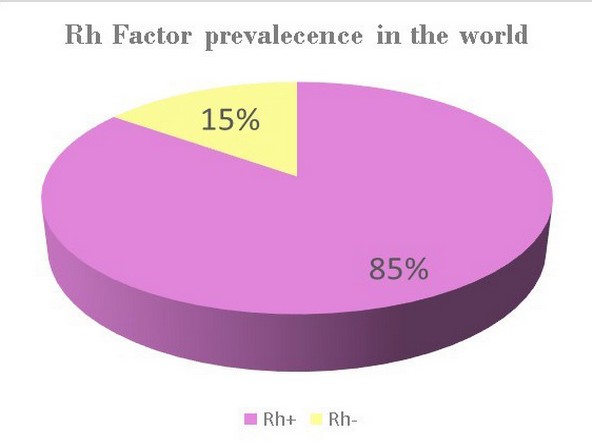
Figure 3. Rh factor, statistics. * Data obtained from OMS.4 and Dean L. 6 In this figure, we see global statistics obtained from several studies about these factors. The illustration shows that Rh+ is the most common factor found in the world population. However, Rh- remains present in a small part of the population, and although it is not a disease, it can generate incompatibility in a pregnancy if the parents have different Rh factors.
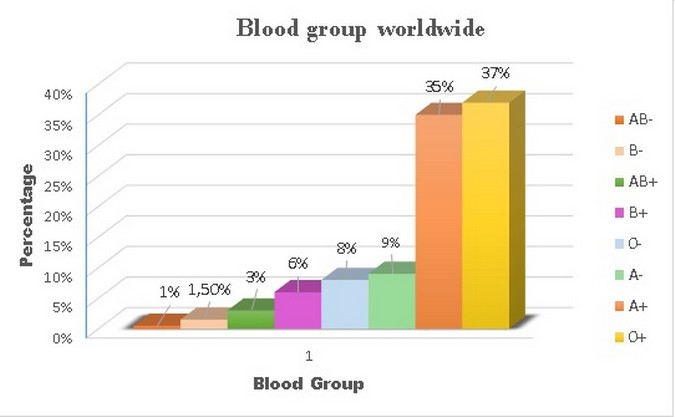
Figure 4. Blood groups statistics. * Data obtained from OMS.4 and Dean L. 6 We can observe that the most common blood group in the world is 0+, followed by A+. Furthermore, it can be observed that the less common blood group is AB-, followed by B-. The predominance of a blood type also varies depending on the countries or continents we are located in.
Obtaining Blood Sample
The process starts with the blood collection directly from the bloodstream to go into a well-labeled container with a pre-measured anticoagulant. Moreover, the addition of an anticoagulant avoids the agglutination of the blood and prevents the RBCs from undergoing hemolysis. There are different preservative solutions for maintaining RBC units: acid citrate dextrose (ACD), citrate phosphate dextrose (CPD), or CPD-adenine. Different solutions such as anticoagulants and nutrients help RBCs store and transport, enabling modern blood banking.9 Also, between the most common anticoagulant solutions, we found Citrate–phosphate–dextrose (CPD), which is a key for blood preservation.10 However, many others novel storage solutions allow the conservation of RBCs for much more time, such as SAG-Mannitol or AS-1.
The influence of ATP over the RBCs also has to be considered to keep them in efficient conditions. Since these cells do not have mitochondria, they rely on ATP from the medium as an energy source. Significantly, when ATP has been suppressed, some characteristics of the RBCs are compromised. According to research, during RBCs storage, adenosine triphosphate (ATP) decreases, modifying membrane lipid content and cell rigidity that results in shape changes.11 The pH also changes during the RBCs storage, and the production of lactic acid mainly causes this due to ATP metabolism. So then, proper RBC storage relies on the correct control of the ATP levels.
RBCs Preservation
The storage and maintenance of RBCs units require precise control for some factors such as temperature, osmotic pressure, and pH. When RBCs are stored, they deteriorate, compromising their metabolic, morphologic, and membranous characteristics. The elements that are mainly altered are the hemoglobin, potassium concentration, supernatant, and a loss of discoid shape and lipid membrane vesicles.12 Moreover, RBCs units can be stored for up to 42 days. However, its ability to deliver oxygen decreases on time due to decreased 2,3-diphsophoglycerate levels.13 Temperature, for instance, has been demonstrated to modify the live time of RBCs during storage. The best-used technique for their preservation is cryogenics. Nevertheless, even low temperatures of 4°C ± 2°C help to the viability of RBCs and protect them from bacterial growth.3
The osmotic pressure is another crucial factor for the maintenance of RBCs. If modified, RBCs undergo hemolysis by breaking their lipid membrane because of the different gradient concentrations. To avoid that, cells have to be under isotonic conditions (0.9% NaCl) in which the outside and the inner osmotic pressure are the same. One of the leading isotonic preservative solutions used for RBCs storage is EDTA. However, using this solution for blood unit preservation produces changes in erythrocyte morphology and osmotic fragility.14 Fortunately, AS-3 is another preservative solution that works better. Recent research showed that using AS-3 as the storage solution for 6 weeks allowed the recovery of 78–84% of the RBCs, and only 0.4% of hemolysis were detected.9
RBCs maintenance also depends on the pH concentration. Cells keep producing metabolites such as Lactate and CO2 during blood storage, acidic molecules. The accumulation of those acidic compounds during storage results in a decrease of pH levels, and when it drops below 6.4, the deterioration of RBCs occurs faster. Moreover, the main consequences of a low pH are the cessation of glycolysis and the change in ATP and 2,3-DPG levels.12 While according to the literature, the optimum pH for glycolysis is high (7,8-8,5) when the temperature is between 15°C and 35°C.15 Therefore, changes in pH during prolonged storage time can result in RBCs damage. Fortunately, when RBCs are stored at low temperatures, their metabolism slows down, allowing the pH to stay at optimum concentrations for a longer time.
Blood Transfusion
Blood transfusion is a medical procedure in which a patient or receptor obtains blood or any of its components from the donor's blood.4 Blood transfusions provide the patient with the components of the blood they need, and RBCs are the most commonly used.7 A person may need a blood transfusion for several reasons, such as surgeries, injuries, replenishing blood volume, correct serum protein levels, and improved hemoglobin and oxygen-carrying capacity.16
To start a blood transfusion, it is essential to know the type of blood the patient has (AB0 system) and whether the Rh factor is positive or negative. This is because the donor's blood has to be compatible with the patient's to avoid an immune response. After checking all the data, the temperature of the blood bag is checked, and if the blood is icy, it is necessary to wait a few minutes for it to be at room temperature.6 Transfusion in high volumes when blood is cold can cause cardiac arrest.16 In most cases, it is not necessary to heat the blood to be transferred. However, suppose blood needs to be heated. In that case, it can be heated to temperatures of approximately 35 to 38 ° C and being careful not to exceed over 42 ° C.17 This is mainly because of the viability of the blood unit when blood is exposed to temperatures above 42 ° C hemolysis can occur.10 Also, blood cannot be returned to the blood bank for reuse after some warming process. Once the temperature of the blood bag is checked, it is perforated with a blood filter, purged, and deposited in a bath. Furthermore, finally, blood is placed through the intravenous.18 It is necessary to monitor the patient for a few minutes to find any problems or reactions.
Transfusions can last approximately 1 to 3 hours,18 but should not last more than 4 hours because there are dangers of bacterial proliferation,10 when blood increases its temperature by staying at room temperature. According to McCullough (2017): "For routine transfusion to stable patients' rates of 2–3 mL/mn for red cells and 7–10 mL/mn for platelets and plasma are recommended" .10
Blood Transfusion Issues
Blood transfusions usually do not have adverse effects but sometimes have different effects depending on the patient (transfusion reactions). Several reactions may be due to storage, transportation, or administration of blood units. These reactions are very varied and can be fatal or straightforward. The most common are mentioned below.8 One transfusion reaction can be incompatible blood: When the donor's blood is not compatible with the patient's. The patient's cells treat the donor's blood as foreign invaders, and the immune system attacks accordingly.6 Another can be allergic reactions: These can cause by fever, itching, and hives.19
The most common problems in transfusions are mainly in massive transfusions. Since these transfusions replace the total volume of the patient's blood, these transfusions involve problems such as cardiac arrest,10 coagulation, hypothermia.19 On the other hand, another frequent problem is Transfusion-related acute lung injury (TRALI), an acute respiratory distress syndrome.8
Transfusion-related infections: Many protocols are now followed to prevent these infections; however, few occur. These infections can be bacterial or viral. In the case of bacterial contamination, these are often rare. For example, this can occur if an asymptomatic donor has bacterial contamination at the time of blood donation or venous puncture.17 In contrast, the viral infection. They are unusual; nevertheless, if they contract, they can provoke: Hepatitis A, hepatitis B, which occurs in approximately 1 in 100 000 cases. Hepatitis C occurs in less than 1 in 1 000 000 cases.16 And HIV occurs in less than 1 in 4 000 000 cases.17 There are many other adverse effects, but those mentioned above are the most common.
Recent RBCs Modification Methods
The fatalities related to Blood transfusion, discarding those due to disease transmission, have been estimated to happen once in 100,000 patients. The most common reason is hemolytic reactions due to incorrect blood type matching.10 As there are many immunogenic antigens on RBCs and blood transfusions that rely on the removal of these to succeed, new methods have emerged to avoid an immune response when blood transfusion is performed. The objective of these novel methods is to produce non-immunogenic RBC; therefore, these blood units can be used as a source of universal RBCs for blood transfusions. In this way, the primary goal of modified RBCs is to elicit neither an immediate nor a secondary immune response when a transfusion is accomplished.20 Furthermore, in clinical application, these techniques would establish an entirely new way to treat many chronic diseases with a need for a rare blood type supply. So the entire blood banking system dedicated to blood collection, distribution, and transfusion, which is focused on the ABO group, can be entirely revolutionized by the success of any RBCs modification techniques that we detail next.
Besides, it has been calculated that around 100 million units of blood are donated each year; however, the indications for RBC transfusion and the suitable time of storage before transfusion are still uncertain.13 Blood banks have to collect and store all blood types, even the most common ones, which spend more time saved than the uncommon ones. This can result in non-viable RBCs. However, the two main general procedures can be used to create a universal RBC; these techniques can potentially overcome the previously mentioned issues. The first way is by permanently removing the RBC antigens anchored to their surface, and the other way is by hiding their antigens so that the immune system cannot detect them.21 These two methods have been explored over the years, and we detail these two recent and promising RBC surface modification techniques.
6.1. Enzymatically Converted Group O (Eco)
The first modification technique is then enzymatically converted group O (ECO), which removes the antigens from the RBCs surface. The main feature of this method is the use of an α-galactosidase enzyme, capable of removing the immunodominant part from the A and B antigens, which are the main characteristics of the A and B blood type respectively. For blood type A, its immunodominant monosaccharide is a terminal a1-3 linked N-acetylgalactosamine (GalNAc), and for type B, its monosaccharide is a1-3 linked galactose (Gal).22 As the blood group O does not have these monosaccharides on their oligosaccharide terminal chains. Therefore, the ECO technique enables A and B blood groups to be converted into O types. Although this method can produce the universal blood type donor O, it has faced some obstacles to succeed in clinical practice because of the low efficacy of the enzyme.
Nevertheless, new research shows that more efficient glycosidase enzymes present in bacteria. The first approach for the conversion of type B RBCs to O was by using an 𝛼-galactosidase enzyme, which can be extracted from coffee beans.22 For this enzyme, many studies have been carried out. However, it was hard to move into the clinical application, mainly because it showed low kinetic properties. Fortunately, entirely new families of bacterial exo-glycosidases have been found, and they show better kinetic properties to remove A and B antigens from RBCs.23 Moreover, new clinical trials are now being performed to prove the efficacy of this new family of enzymes for further developing a universal blood type.
The most relevant benefit of ECO technology on transfusion medicine is eliminating hemolytic reactions due to ABO-incompatible transfusion. The technique reduces the RBC units' typing costs, avoids the unnecessary storage of common blood types, increases the availability of rare blood types units in blood banks, creates a universally compatible RBC, and saves hospital money for an inventory of blood units.23
RBC Antigen Masking
Unlike ECO which converts different blood groups into O type, the second method attempts to camouflage or mask the antigens from the RBC surface. RBCs undergo a surface modification procedure, where these cells are treated with the polyether polymer PEG or other substances such as hydrogels and polymers. These compounds are chemically anchored to RBC surface proteins through various liking procedures. Also, this method results in stealth RBCs, which allows them to pass unseen from the immune system, disabling antigen-antibody reactions and alloimmunization to additional antigens.21 Therefore, one of the main goals of this modification technique is to obtain healthy RBCs capable of typically performing their metabolic processes as oxygen transfer and nutrient transport.
PEG in transfusion medicine has become a key tool to avoid immune responses, and PEGylation is the process by which PEG is linked to other molecules. This procedure improves the pharmacokinetic behavior of many substances because it avoids recognition by the cells of the immune system, allowing the introduction of several agents into the human body.24 Between the most used substances for the PEG-RBC linking, we found: cyanuric chloride (CN), ethylene glycol (CH2OH)2, propionic acid (SPA), and benzyltriazobyl carbonate (BTC).25 Moreover, the research of PEGylated RBCs becomes more reliable since 4 groups of research showed that if PEG is covalently attached to the RBC surface, these cells are not capable of reacting with antigens ABO group.25 As a result, PEG-RBC linking is one of the most valuable procedures to stealth RBCs towards developing a universal blood type.
Nevertheless, this method may carry some complications and cell changes. For example, some biomimetic molecules such as polydopamine (PDA) can shield RBCs. However, this treatment produces noticeable changes in membrane fluidity, which leads to increased cell fragility.1 RBCs depend on their normal membrane stability to freely circulate through the bloodstream without fragmentation. Consequently, a decreased stability may lead to a breakdown even under everyday circulatory stresses.26 To solve the fragility problem, new methods have emerged to overcome this problem, such as polysialic acid (PSA)–tyramine hydrogel, which creates an enhanced flexible framework around RBCs that ensures their surface stability. This article's clinical trials explain the details of an experiment done with (PSA)–tyramine and rabbits.
Universal RBCs Transfusion
Different criteria must be carefully examined when modified RBCs are transfused. Some of these are similar to the analysis made on regular RBCs transfusions. However, it is more critical for the modified cells to see possible immunogenic reactions. In addition, the ECO technology is the only one that has passed into human clinical trials and with relevant success.23 This technology showed no significant changes in the RBCs morphology and functions, so it was allowed to be used in phase 1 of clinical trials. Indeed, research have shown that during the in vitro transfusion tests, there was no noticeable increase in anti-B for the samples of type A or O. Actually, their sera remained with no indications of agglutination or hemolysis after seven weeks of the transfusion.25 Even though this method does not present relevant problems to the host, it is essential to take immuno-tests periodically to evaluate that the procedure has done well.
Pre-Clinical Trials
There are no clinical trials of transfusions with modified universal red blood cells yet. Nevertheless, researchers used New Zealand white rabbits in a pre-clinical study performed in vivo to evaluate immunogenicity. These rabbits were randomly divided into four groups (Table 1). Table 1 below shows the distribution of each group of rabbits, which injections they received, and the immune response for each one.
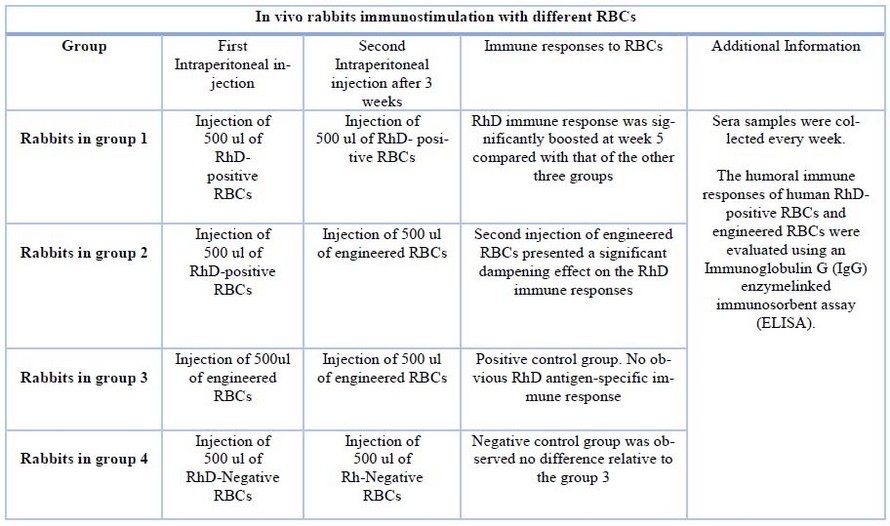
Table 1. Process in vivo rabbits immunostimulation with different RBCs. *Data obtained from Zhao Y. et al. 1
The study continues with ELISAs, according to Zhao Y. et al. (2020), as follows:
ELISA was performed to analyze the binding of both the framework and the non-framework to RhD-positive red blood cells and evaluate RhD antibody titers in vivo. Recombinant human RhD proteins were used to coat ELISA plates. At night they were incubated at 4 ° C, then the plates were washed three times with PBS, and after this, for two hours, they were blocked with a blocking buffer at 37 ° C. Next, the plates were washed three times, and the sera samples (100 l) were added.1 We proceeded to incubate at 37ºC for one hour, then the plates were washed three times, and 100 μl of secondary antibody conjugated with HRP were added. Next, we proceeded to incubate at 37ºC for 45 minutes, and afterward, the plates were washed three times, and 100 l of TMB chromogenic substrate solution was added. After 10 minutes, 50 l of H2SO4 1 M were added to stop the reaction of the endpoint titers. Using a microplate reader, absorbance was measured at 450 nm. 1
It was seen that anti-RhD immunity in surface modifications could be avoided, and favorable biocompatibility was also promoted. Also, there was no recognition between antigens and antibodies. Therefore, the trial worked properly as it avoided immune responses altogether, thus ensuring a successful RhD-stealth blood transfusion.1
CONCLUSIONS
Blood units are used worldwide, and blood type matching is one of the biggest challenges in the medical field. Because there are different blood groups, and most of them have specific antigens, blood transfusion has to overcome different typing processes before being transplanted. To maintain RBCs on their normal conditions, factors such as coagulation, temperature, pH, and storage time must be controlled to prevent changes in RBCs' morphology and oxygen transport capacity. Additionally, the variety of blood types results in an unequal supply for hospitals and clinical procedures. However, there are two techniques to engineer universal red blood cells. The first is the ECO technique, which uses an α-galactosidase enzyme that removes the surface antigens from RBCs to transform them into O type. The second is antigen masking, here a framework usually made of hydrogels, or PEGs, binds to the surface of red blood cells. Hence, the host's immune system cannot recognize them after transfusion. These two methods are promising and have been extensively studied to help supply blood banks, especially for people with uncommon blood types.
Author Contributions: Conceptualization, methodology, resources and writing—original draft preparation, Esteban Guamba. Writing—review and editing, visualization, supervision, Alejandra Cevallos.
Funding: "This research received no external funding"
Acknowledgments: Thanks to Ph.D. Santiago Vispo, who encouraged and tutored us to write this article
Conflicts of Interest: The authors declare no conflict of interest..
REFERENCES
1. Zhao Y, Fan M, Chen Y, Liu Z, Shao C, Jin B, et al. Surface-anchored framework for generating RhD-epitope stealth red blood cells. Sci Adv. 2020;6(12):1–12.
2. García-Roa M, Del Carmen Vicente-Ayuso M, Bobes AM, Pedraza AC, GonzálezFernández A, Martín MP, et al. Red blood cell storage time & transfusion: Current practice, concerns & future perspectives. Blood Transfus. 2017;15(3):222–31.
3. Brunskill S, Thomas S, Whitmore E, McDonald CP, Dorée C, Hopewell S, et al. What Is the Maximum Time That a Unit of Red Blood Cells Can Be Safely Left Out of Controlled Temperature Storage? Transfus Med Rev [Internet]. 2012;26(3):209-223.e3. Available from: http://dx.doi.org/10.1016/j.tmrv.2011.09.005
4. OMS. Organización Mundial de la Salud [Internet]. [cited 2020 Apr 16]. Available from: https://www.who.int/es
5. Cruz Roja Española. Grupos sanguineos - Centro de Donación de Sangre de Cruz Roja [Internet]. [cited 2020 Apr 16]. Available from: https://www.donarsangre.org/grupossanguineos/
6. Dean L. Blood Groups and Red Cell Antigens. ABO blood Gr [Internet]. 2005;(Md):Chapter 5. Available from: http://www.ncbi.nlm.nih.gov/books/NBK2267
7. OMS. La Base de Datos Global de la OMS para la Seguridad Sanguínea. 1999. Organ Mund la Salud. 2001;8.
8. Goodnough LT, Brecher ME, Kante M, AuBuchon J. Transfusion Medicine. Br J Educ Stud. 1965;13(2):208–11.
9. Hess JR. An update on solutions for red cell storage. Vox Sang. 2006;91(1):13–9.
10. Mccullough J. 13 Techniques of Blood Transfusion. 2012;362–77.
11. Klein HG, Spahn DR, Carson JL. Series Transfusion Medicine 1 Red blood cell transfusion in clinical practice. Prevention. 2007;415–26.
12. Holme S. Current issues related to the quality of stored RBCs. Transfus Apher Sci. 2005;33(1):55–61.
13. Carson JL, Guyatt G, Heddle NM, Grossman BJ, Cohn CS, Fung MK, et al. Clinical practice guidelines from the AABB: Red blood cell transfusion thresholds and storage. JAMA - J Am Med Assoc. 2016;316(19):2025–35.
14. Antwi-Baffour S. Prolong Storage of Blood in EDTA Has an Effect on the Morphology and Osmotic Fragility of Erythrocytes. Int J Biomed Sci Eng. 2013;1(2):20.
15. Guppy M, Attwood P V., Hansen IA, Sabaratnam R, Frisina J, Whisson ME. pH, Temperature and Lactate Production in Human Red Blood Cells: Implications for Blood Storage and Glycolytic Control. Vox Sang. 1992;62(2):70–5.
16. Goodnough LT. Risks of blood transfusion. Crit Care Med. 2003;31(12 SUPPL.).
17. Maxwell MJ, Wilson MJA. Complications of blood transfusion. Contin Educ Anaesthesia, Crit Care Pain. 2006;6(6):225–9.
18. Patil V, Shetmahajan M. Massive transfusion and massive transfusion protocol. Indian J Anaesth. 2014;58(5):590–5.
19. Eder AF, Chambers LA. Noninfectious complications of blood transfusion. Arch Pathol Lab Med. 2007;131(5):708–18.
20. Bradley AJ, Test ST, Murad KL, Mitsuyoshi J, Scott MD. Interactions of IgM ABO antibodies and complement with methoxy-PEG-modified human RBCs. Transfusion. 2001;41(10):1225–33.
21. Lublin DM. Universal RBCs. Transfusion. 2000;40(11):1285–9.
22. Olsson ML, Hill CA, De La Vega H, Liu QP, Stroud MR, Valdinocci J, et al. Universal red blood cells - Enzymatic conversion of blood group A and B antigens. Transfus Clin Biol. 2004;11(1):33–9.
23. Olsson ML, Clausen H. Modifying the red cell surface: Towards an ABO-universal blood supply. Br J Haematol. 2008;140(1):3–12.
24. Shimizu T, Abu Lila AS, Awata M, Kubo Y, Mima Y, Hashimoto Y, et al. A Cell Assay for Detecting Anti-PEG Immune Response against PEG-Modified Therapeutics. Pharm Res. 2018;35(11).
25. Garratty G. Progress in modulating the RBC membrane to produce transfusable universal/stealth donor RBCs. Transfus Med Rev. 2004;18(4):245–56.
26. Chasis JA, Mohandas N. Erythrocyte Membrane Deformability and Stability : J Cell Biol. 1986;103(August):343–50.
Received: 25 October 2021 / Accepted: 25 November 2021 / Published: date. 15 February 2022
Citation: Guamba E, Cevallos A. Recent advances on the development of a universal blood type. Revis Bionatura 2022;7(1). 32. http://dx.doi.org/10.21931/RB/2022.07.01.32
