2022.07.03.11
Files > Volume 7 > Vol 7 No 3 2022
Coffee's carbohydrates. A critical review of scientific literature.
Ostilio R. Portillo* 1 and Ana C. Arévalo 2
1 Faculty of Engineering, National Autonomous University of Honduras (UNAH), Tegucigalpa, Honduras.
2 Faculty of Chemistry & Pharmacy, National Autonomous University of Honduras (UNAH), Tegucigalpa, Honduras.
* Correspondence: [email protected]; Tel.: 504-9376-1660
vailable from: http://dx.doi.org/10.21931/RB/2022.07.03.11ABSTRACT
Only two species have gained economic importance in coffee production: Coffea arabica L. (Arabica coffee) & Coffea canephora Pierre ex A. Froehner var. Robusta, with 65 and 35% of world production attributed to C. arabica http://wsx5customurl.comL. & C. canephora P. respectively. In general, it is estimated that 6 mt of fresh and ripe fruits produce approximately 1 mt of raw and dry grains. The grain endosperm is mainly composed of cellulose, hemicelluloses, proteins, minerals and lipids, but starch and tannins are absent. However, the seed's chemical composition of C. arabica and C. canephora, before roasting, differs concerning their primary and secondary metabolites content, which serve as precursors for the synthesis of volatile compounds during the roasting process. For this reason, there are marked organoleptic differences between both species' roasted and ground grain. However, the evidence suggests that such differences can also be attributed to other factors since coffees grown in cool, highland areas generally have better sensory attributes than their counterparts grown in hot, lowland areas. It has been speculated that environmental conditions in cool, highland areas induce the slow accumulation of primary and secondary metabolites during the endosperm development resulting in sensorial differences after roasting. This essay focuses on the study of coffee beans' carbohydrates (primary metabolites) before and after roasting, their influence on cup quality, biosynthesis and differences linked to the involved species, their metabolism, solubility and extraction, as well as a discussion on the analytical techniques used for its determination.
Keywords: sucrose synthase, sucrose phosphate phosphatase, sucrose phosphate synthase, aploplasm, cytoplasm, Manan synthase, Galactosyl transferase.
INTRODUCTION
Carbohydrates (mono, di, oligo, & polysaccharides) constitute the most considerable fraction of the coffee bean composition and are responsible for ~ 50% of its dry weight (Table 1), a concentration calculated once all the other constituents of the raw grain are removed from the total weight (Formula 1).1-4 Additionally, the bean carbohydrates are responsible for approximately 29% of the high molecular weight material in the coffee beverage.5
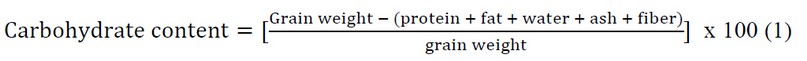
Small amounts of pectins and xyloglucans have been reported in coffee. Also, small amounts of free monosaccharides [eg, arabinose (the only pentose present in the raw grain in significant amounts), fructose, glucose (aka dextrose), galactose, mannose, mannitol, raminose, ribose, & xylose], disaccharides (sucrose), and water-soluble oligosaccharides [eg, raffinose (trisaccharide) & stachyose (tetrasaccharide)].1,3,6-8
However, it should be noted that arabinose, galactose, mannose and glucose are weak acids 9 and in turn, the constitutive monomers of the three main high molecular weight polysaccharides of the coffee bean: galactomannans, arabinogalactans and cellulose, which being reserve carbohydrates, in their refractory form, are part of the cell wall 4,10-19 and together constitute between 47-60% of the raw coffee bean dry weight.8,14,20,21
On the other hand, polysaccharides (soluble and insoluble) are responsible for ~ 43, and 54% of the dry weight of C. arabica and C. canephora, respectively,1,6,22 and these can be identified by studying their constitutive monosaccharides through chemical hydrolysis usually with dilute acids (eg, H2SO4, HCl), 2,9,23 or alkaline treatments (eg, NaOH, KOH).23
Biosynthesis
During fruit development, carbohydrates are synthesized in the leaves and in the fruit pericarp. The type and concentration of soluble sugars present in the endosperm varies during its development. In the early stages, glucose and fructose (reducing sugars) prevail with a higher concentration of the former one 24 and range between 16.5 and 1.6% on a dry matter basis, respectively.14
However, their levels begin to decline as soon as endosperm expansion begins, and by the end of grain development, their concentrations plummet to 0.03 and 0.04% on a dry matter basis, respectively.22,25 However, the sucrose concentration increases by 5-12% 10,24,26 and is used as a precursor for synthesizing lipids and proteins.26
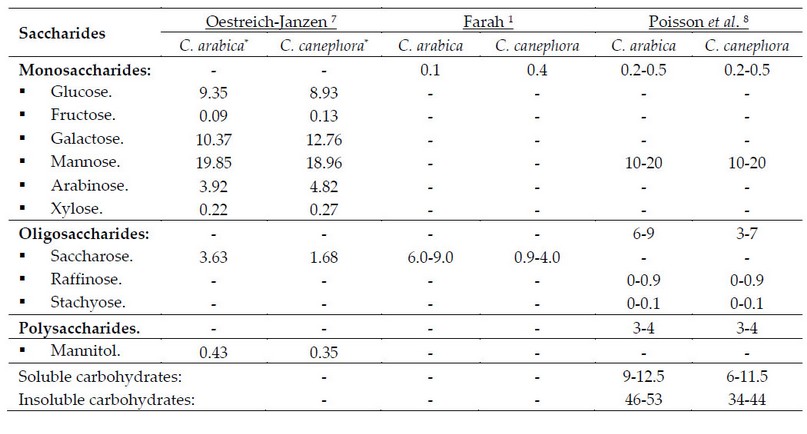
* Percentages calculated based on a dry matter basis.
The observed discrepancies reported by different authors can be attributed to the species involved and the techniques used for the extraction & determination (identification and quantification) of analytes.
Table 1. Raw coffee bean's carbohydrate content (%).
In C. arabica, the biosynthesis and accumulation of sucrose takes place in the grain endosperm and perisperm,25 processes that is carried out in two stages: a) the sucrose degradation by sucrose synthase and invertases & b) the sucrose synthesis by the action of sucrose phosphate phosphatase and sucrose phosphate synthase, the latter being the most important enzyme in the coffee bean's sucrose production (Figure 1).26
A sucrose fraction dissolved in the intercellular fluid manages to cross the cell membrane with the help of specialized transporters. In contrast, another fraction is hydrolyzed into its constitutive hexoses and transported into the cytoplasm (Figure 1).
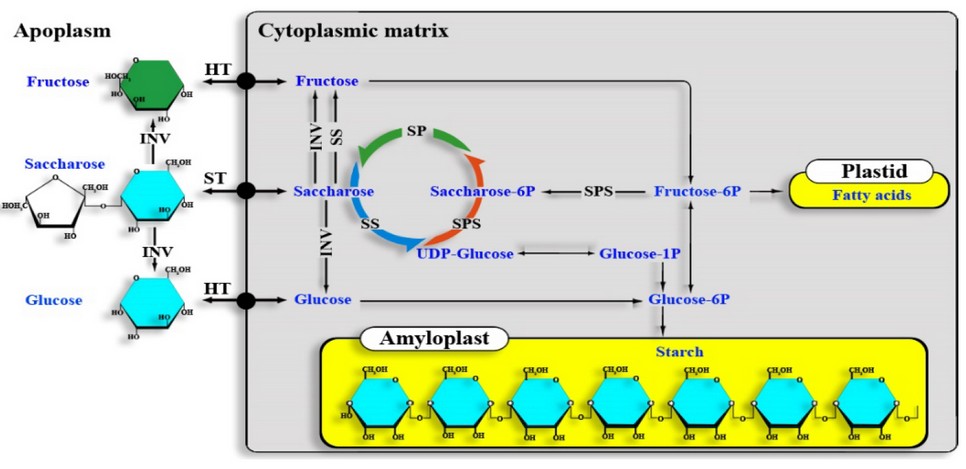
HT: Hexose transporter, ST: Sucrose transporter, INV: Invertase, SS: Sucrose synthase, SP: Sucrose phosphatase, SPS: Sucrose phosphate synthase, Sucrose-6P: Sucrose-6-phosphate, Fructose-6P: Fructose-6-phosphate, Glucose-1P: Glu-cose-1-phosphate & Glucose-6P: Glucose-6-phosphate.
Figure 1. Sucrose biosynthesis in coffee. Adapted from Cheng et al. 26.
Furthermore, it can be inferred that sucrose synthesis is the product of the metabolic action of two enzymes operating at different stages of grain development. For example, sucrose phosphate synthase reaches its point of maximum expression (transcription) approximately 60-90 days after anthesis while sucrose synthase reaches its maximum expression approximately 120-150 days after anthesis.26
The continuous flow of sucrose and its monomers from the aploplasm into the cytoplasm concomitantly to the metabolic action of sucrose phosphate synthase and sucrose synthase explains why the sucrose concentration increases significantly at 150 days after anthesis, and after this point, its synthesis decreases but does not stop.
There is a wide sucrose content variation between different species within the genus Coffea (Table 2), which can be used in coffee breeding programs.27 However, sucrose concentrations are always higher in C. arabica than in C. canephora 18,22,26,28; On average, coffee beans of C. arabica and C. canephora display a concentration that varies between 8.2-8.3% and 3.3-4%, respectively (Figure 2)8,29 although other authors have reported 7.3-11.4% 14,25,30,31 and 4-5%, respectively.25,31
In general, Robusta varieties tend to accumulate 30% less sucrose than Arabica varieties. The evidence suggests that the synergistic effect of a higher metabolic activity of sucrose synthase and acid invertase, in the early stages of seed development in Robusta varieties, is the main reason why Arabica varieties have higher sucrose concentration.22,26
On the other hand, during grain filling, the metabolic activity is oriented toward the systematic accumulation of storage polysaccharides; However, the grain polysaccharide constitution also varies as it develops. In the early stages of endosperm development, cellulose and arabinogalactans predominate; while the galactomannans concentration, which at this stage are characterized by a greater degree of lateral branching, is barely 10% of the total polysaccharides.32
In contrast, as the grain reaches maturity, galactomannans prevail, but their number of lateral branches is reduced.2,32
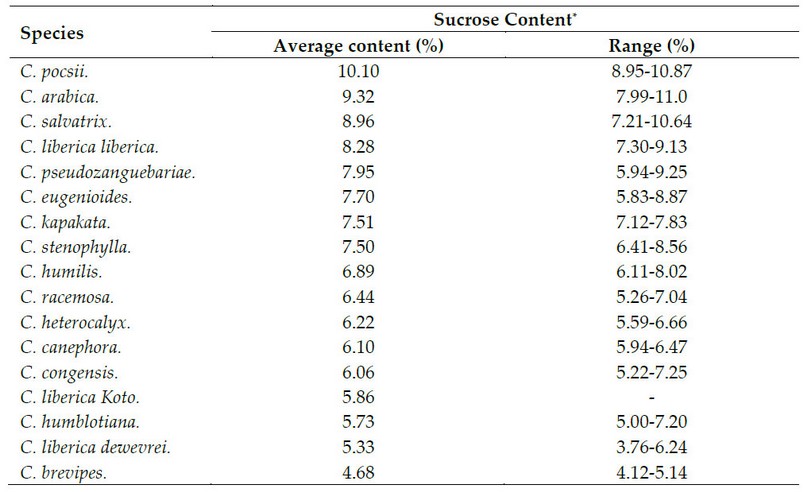
* Contents are calculated based on dry matter. Adapted from Campa et al.27.
Table 2. Sucrose content in several species of the genus Coffea.
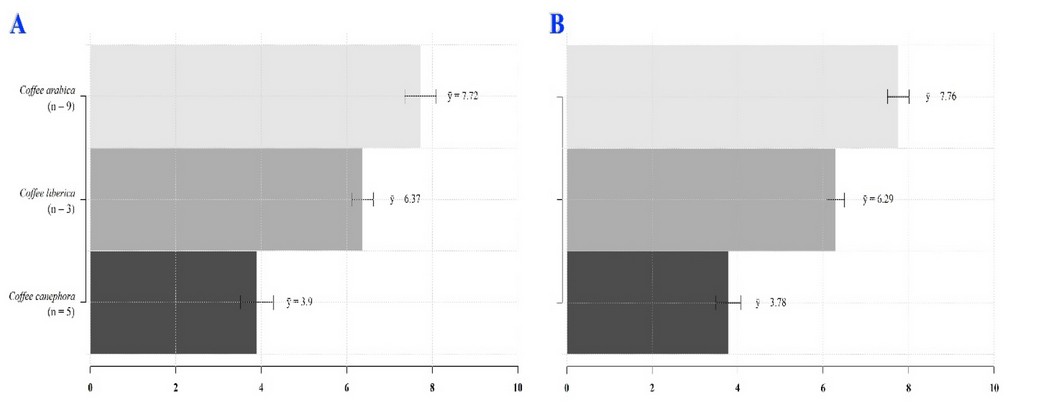
Adapted from Stredansky et al. 33
Figure 2. Raw coffee bean's sucrose content was calculated via HPLC (A) & Biosensors (B).
Regardless of the species, two genes have been proposed as being responsible for galactomannans biosynthesis in coffee beans. During the coffee bean endosperm development, ManS1 and GMGT1 encode two glycosyl transferases: Manan synthase (ManS) and Galactosyl transferase (GMGT), respectively. Both enzymes are located at the Golgi vesicles membranes and fulfill different functions.19 For instance, ManS catalyzes the coupling of D-mannose monomers form-ing the galactomannans backbone while GMGT catalyzes the coupling of D-galactose units at the C-6 position of D-mannose monomers.19,32
In the end, the number of D-galactose units coupled to the main polymer axis is determined by a third enzyme called α-galactosidase,32 but based on the evidence compiled through multiple studies, it has been concluded that the number of D-galactose units incorporated into the mannan backbone in the mature coffee bean is quite low.19
On the other hand, an analysis of the raw coffee bean's polysaccharides content and structure suggests there is a 50% mannans and galactomannans,2,5,19 22% arabinogalactans, and 20% cellulose.2 However, despite the similarities be-tween C. arabica and C. canephora regarding polysaccharide composition, Robusta varieties have approximately 3% more arabinogalactans than Arabica varieties.22
Finally, there is evidence suggesting the presence of enzymes associated with starch biosynthesis during the early stages of seed development,14 indicating a brief starch accumulation followed by its metabolism (catabolism). This would explain the absence of starch in the raw coffee bean,2,16,34 which is why its use in the food industry is limited.
The most important structural polysaccharides of the coffee bean are described below.
Cellulose
Cellulose is a non-water-soluble polysaccharide 1 that is part of the cell wall and represents ~ 8% of the grain dry weight in C. arabica and C. canephora 7,12,19; Additionally, it also constitutes ~ 20% of the polysaccharide composition of the raw coffee bean; and ~18% of the roasted one.35
Cellulose is a polymer with a rigid and linear structure (that is, without branches) made up of glucose units linked through β-type glycosidic bonds (1→4) (Figure 3), which makes it indigestible for humans. It is considered part of the dietary fiber. In addition, its indigestibility is also attributed to the strong bonds with hydrogen and the intricate network of fibers that block direct contact with acids and digestive enzymes.36
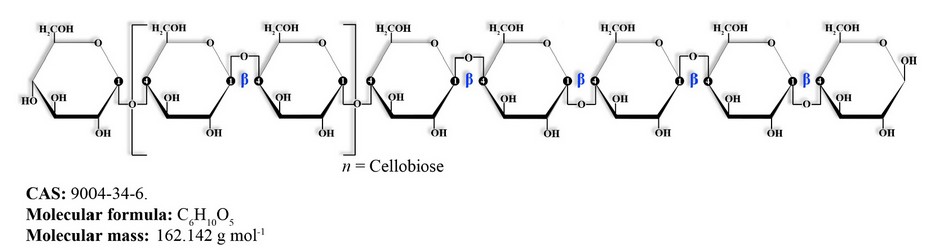
Figure 3. Cellulose molecule diagram.
From a structural point of view, cellulose can also be defined as a polymer of cellobiose, which in turn is defined as glucose dimers linked through β-type glycosidic bonds.37 Cellulose alone has great economic value as it is the main constituent of various industrial products such as cotton and paper.
On the other hand, the cellulose molecular structure presents crystalline (organized) and amorphous (less organized) regions,23,36, which impart rigidity and softness to the molecule, respectively.38 The crystalline and the amorphous regions remain practically unchanged during and after toasting, which is why cellulose is not part of the soluble polysaccharides in the beverage.8,34,39,40
The cellulose resistance to thermal degradation during roasting could be attributed to the fact that the fibers of this polymer are closely associated with the cell wall, forming a dense and complex matrix with galactomannans and arabinogalactans, which isolates and protects from heat.40 Cellulose fibers make up ~ 15% of the high molecular weight polysaccharides in the cell wall.
Galactomannans
The current scientific literature is somewhat ambiguous regarding the name of these polymers, as some authors describe them as mannans while others refer to them as galactomannans. The discrepancy stems from the very low number of D-galactose monomers coupled to the molecular structure. For this reason, it has been suggested to name all D-mannose polymers that have a concentration of D-galactose units ≥ 5% as galactomannans.32
In the coffee bean, the central galactomannans axis is similar to that of cellulose since it is also composed of a chain of monomers (D-mannose) linked through glycosidic bonds type β(1→4), but with substitutions of α-D-galactose or L-arabinose in the sixth position 5,8,11,16,39,41 located in the main axis approximately every 100 monomers.8,17,18 Although substitutions of acetyl groups are also observed in the 2nd, 3rd and 5th position (Figure 4).13,34
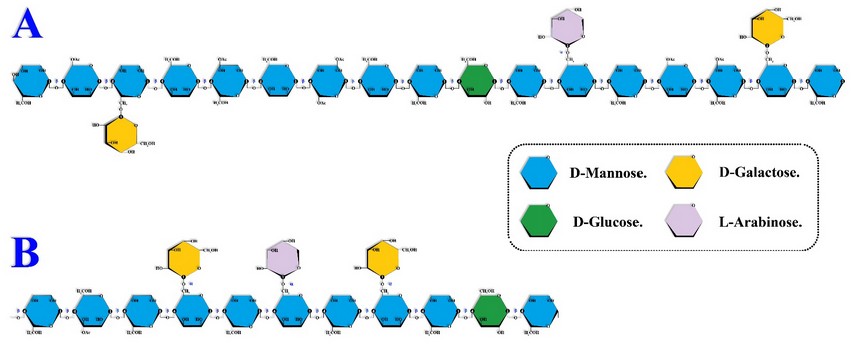
Figure 4. Coffee galactomannans. Adapted from Oliveira Petkowicz 42 (A) & Moreira et al.11 (B).
All organic compounds occur in two isomeric forms. Each isomer contains the same number and type of atoms, but they are arranged in such a way that one is the reverse image of the other. Depending on the direction in which the isomers refract the light when dissolved in an aqueous solution, one is said to be oriented to the right or Dextrorotatory (D) and the other to the left or Levorotatory (L).
In the raw coffee bean, 43% of the mannans extracted with hot water are acetylated in the 2nd (O-2) and 3rd (O-2) positions 5,32,35,41 and to a lesser extent, doubly acetylated and contiguous acetylated monomers.32,41 Likewise, 27% of hot-water extracted mannans from roasted coffee samples are acetylated, doubly acetylated, or have contiguous acetylated monomers.41
However, the degree of acetylation of galactomannans in coffee changes during the roasting. The evidence indicates that the number of acetyl groups incorporated into the polymer structure is reduced when the grain is roasted,32 suggesting a modification in the chemical composition of the polymer.
Likewise, in raw coffee beans it has been possible to verify (through the analysis of glycosidic bonds, enzymatic and chromatographic analyses) the presence of glucose monomers incorporated into the galactomannans main axis through β(1→4) bonds (Figure 4).32 Interestingly, the evidence suggests that in roasted coffee beans, glucose molecules are located only at the reducing end of the polymer,32 which reinforces the hypothesis that changes induced by heat treatment are not limited to galactomannans fragmentation (depolymerization and debranching) but also to structural rearrangements.
On the other hand, galactomannans represent 22-24% of the dry weight of the raw coffee bean for C. arabica and C. canephora,7,12,18,32,43, which in turn represents ~ 46% of the high molecular weight polysaccharides of the raw coffee bean. For this reason, galactomannans and arabinogalactans constitute the most abundant high molecular weight polysaccharides in raw coffee beans, regardless of the species.13,44,45
Likewise, galactomannans are storage carbohydrates that represent ~ 48% of the high molecular weight polysaccharides present in the roasted coffee bean 8,35 and their concentration in the coffee cup is even higher (68-70%) 5,13,41 due to their water solubility.1 It should be noted that water-soluble galactomannans added to type II arabinogalactans, which are also water-soluble, represent the majority of the polysaccharides in the coffee beverage 35 and together constitute between 15-25% of the dry matter present in the infusion.1
The slight percentage increase in galactomannans observed in the roasted grain is the product of a relative increase, that is, its concentration increases due to the denaturation of other polysaccharides more susceptible to thermal degradation. For instance, arabinogalactans are more susceptible to thermal degradation than galactomannans.40
Thermal degradation implies that the polymer's constitutive monomers have been destroyed or chemically modified by high temperatures since the missing carbohydrates (monosaccharides and oligosaccharides) cannot be detected in the roasted coffee. For that reason, we can say that the galactomannans concentration in the roasted grain fluctuates depending on the degree of roasting.5,39
Additionally, the galactomannans concentration in the cup of coffee is also conditioned by the species involved. For instance, according to Simoes et al. 5 "the beverage prepared from Arabica coffee contains a higher concentration of galactomannans (62-80%) than that found in the one prepared from Robusta coffee (44-67%)". Likewise, according to Moreira et al. 32 beverages prepared from Arabica coffee beans contained higher levels of galactomannans than those prepared from Robusta coffee beans, regardless of the degree of roasting.
However, it has also been observed that in Robusta coffees, as the degree of roasting increases, the water-soluble galactomannans concentration also increases; Conversely, in Arabica coffees, the water-soluble galactomannans con-centration remains practically unchanged regardless of the degree of roasting.8,32
This phenomenon is attributed to a cell wall with a weaker structure, possibly due to a greater porosity that causes its rapid degradation, which is why when Arabica coffees are roasted they quickly release their hidrosoluble galactomannans.
It has usually been suggested that Robusta coffees' galactomannans have a greater number of lateral branches than those found in Arabica coffees, which is why they are more hidrosoluble. Such side branches are usually linked to the main chain at different intervals through α(1→6) type glycosidic linkages.19
Experiences reported by Fischer et al.16 reported that Robusta coffees (var. ROM) galactomannans present in aqueous solution have a greater number of lateral branches than those found in Arabica coffees (var. Caturra), which in turn would explain their more excellent water solubility, thus facilitating its extraction.
The evidence reinforces the correlation between coffee's galactomannans water solubility and the number of side branches (D-galactose monomers) since galactomannans samples with a low level of side branches have been found in the precipitated fraction of aqueous coffee extracts.32
Finally, thanks to their physical-chemical properties and their innocuousness, galactomannans are used in the textile, cosmetic and pharmaceutical industries, but also in the food industry as stabilizers, in the formulation of dairy products, in confectionery, dietetic products, creams, seasonings, sauces, etc.
Arabinogalactans (AGs)
AGs are heterogeneous polymers of D-galactose and L-arabinose that constitute between 14 and 17% of the dry weight of the coffee bean 7,8,19 of C. arabica and C. canephora, respectively,18 which in turn represents ~ 36% of the high molecular weight polysaccharides of the raw grain; However, this proportion is further reduced during roasting since ~ 34% of them correspond to type II arabinogalactans.35
Generally, arabinogalactans, especially type II arabinogalactans, are found covalently bound to proteins forming AGs-protein complexes 4,11,17,34,45,46 which have an average molecular weight of ~ 600 kDa within a range that varies from 150 to 2000 kDa.12,46
The AGs heterogeneity is manifested by the number of lateral branches attached to their main axis as well as by the degree of polymerization and monosaccharide composition of such branches.34,46
The molecular structure of coffee's AGs was first proposed by Wolfram & Patin in 1965 47 and consists of a backbone composed by a D-galactose monomers chain covalently linked through β(1→3) bonds with lateral branches in the sixth position linked through β(1→6) bonds (Figure 5).4,11,16,39,48 It is in the lateral branches where the monomers of D-galactose or L-arabinose are located.16,39,48,49
The architecture of the side branches is designed in such a way that their main axis is made up of a chain of D-galactose monomers covalently linked through β(1→6) bonds with short side chains of L-arabinose linked to the main axis through α(1→3) bonds.17,32 However, the L-arabinose monomers are linked to each other through α(1→5) bonds (Figure 5).32
On the other hand, the lateral branches' central axis and the short chains of L-arabinose linked to it usually end with monomers of L-rhamnose or glucuronic acid,40 and the latter one can constitute up to 8% of the monomers present in the structure.47
Experiences reported by Fischer et al.16, reveal that the AGs present in Robusta coffees (var. ROM) have a greater number of lateral branches compared to those found in Arabica coffees (var. Caturra), which in turn would explain their greater hydrosolubility and preference, by the industry, in the manufacture of instant coffees.
AGs-protein complexes
The AGs-protein complexes, also known as proteoglycans, are amphipathic molecules 47 which represent 90% of the coffee bean's AGs and are characterized by their low level of extraction since approximately 92% of them are part of the cell wall 12 in addition to displaying hydrocolloid properties, that is, they increase the viscosity of the medium in which they are found.34
AGs-protein complexes are responsible for ~ 25% of the cell wall structural constitution 46 and also represent ~ 15% of the dry weight of the raw grain.34,47,50
They have a tripartite composition: the AGs (the majority and carbohydrate fraction); the protein fraction that constitutes 0.5-2.0% 4,7,13 and the glucuronic acids incorporated as terminal monomers linked to the side chains of the AGs 4,13 which represent the remaining 6-10%.4,12,46
Likewise, AGs-protein complexes have a heterogeneous molecular structure. Such heterogeneity is conditioned by the number of lateral branches which are usually spaced, on the main axis, at intervals of three or four D-galactose monomers.4,50 Likewise, its lack of uniformity is conditioned by the chemical composition of the lateral branches.4 The lateral branches usually present, in their molecular structure, a Galactose/Arabinose ratio of 3:1 or 9:1, that is, for each arabinose monomer there are three to nine galactose monomers.50
Its water solubility is increased by its high number of lateral branches as well as by the degradation (depolymerization) of the main axis and its branches during grain roasting.50 Finally, coffee's AGs-protein complexes are attributed with a series of pharmacological properties, such as the ability to lower blood cholesterol levels and immunoregulatory activity.50
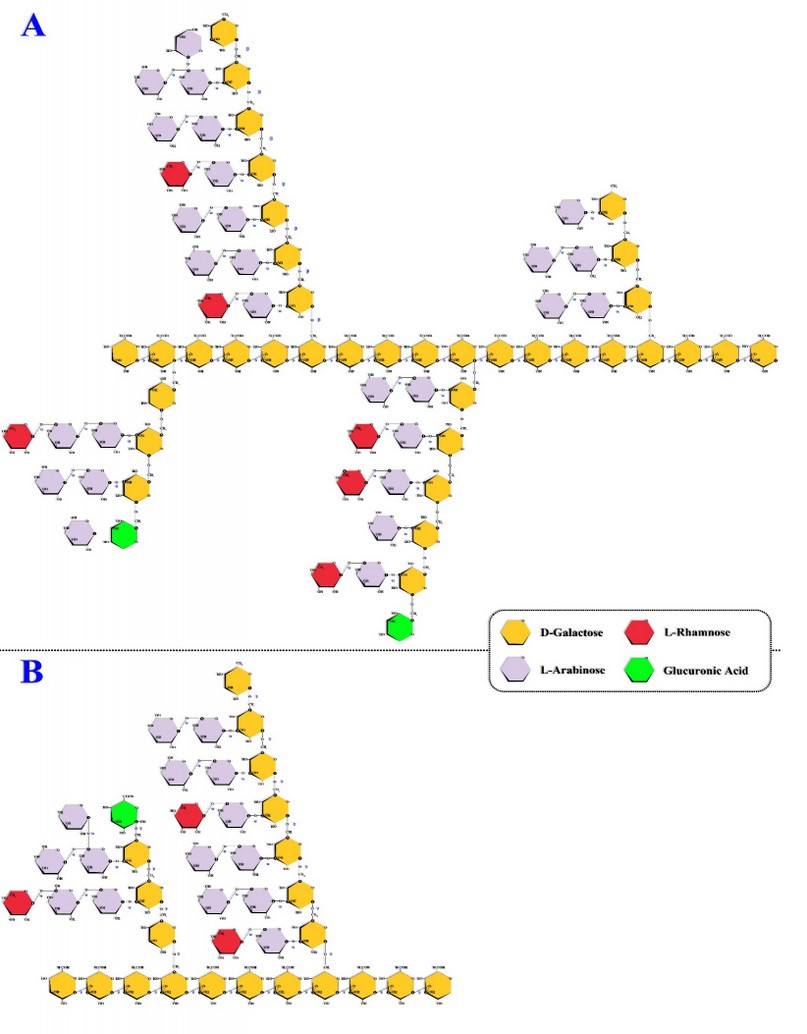
Figure 5. Coffee's arabinogalactans. Adapted from (A) Oliveira Petkowicz42 & (B) Moreira et al.11
Solubility and extraction of coffee polysaccharides
The polysaccharide's insolubility is the result of their high level of polymerization. The larger the molecule, the lower its hidrosolubility; polysaccharides are linked to each other through hydrogen bonds, further decreasing their hidrosolubility. However, according to Simoes et al.5 "the polysaccharides water solubility increases with the number of side branches and the polymer's degree of acetylation". That is, the more lateral branches and acetyl groups incorporated into the molecule structure, the greater its water solubility.
For this reason, chemically induced acetylation is an avenue to increase the solubility of coffee beans' high molecular weight polysaccharides. Interestingly, artificially acetylated polysaccharides differ in their structural conformation from naturally acetylated ones.
According to Moreira et al.32, in naturally acetylated galactomannans the acetyl groups are directly attached to the main axis of the molecular structure; usually at the O-2 position 35; While the artificially acetylated show the new acetyl groups incorporated into the side branches of the polymer.
Additionally, the evidence reported by Simões et al.35 suggests that chemically induced acetylation causes preferential coupling of new acetyl groups to L-arabinose monomers which in turn are laterally and covalently linked to the galactomannan backbone.
On the other hand, it has been speculated that as a result of the chemically induced acetylation the D-galactose monomers, laterally attached to the main polymer axis, should also be acetylated; However, since they have the same molecular weight as the D-mannose monomers, it is difficult to differentiate both sugars once they have been acetylated.35
However, depending on the degree of roasting and polysaccharides resistance to thermal degradation, between 12 and 40% of the high molecular weight polysaccharides are degraded (depolymerization and debranching) during grain roasting,5,8,32,34 thus reducing their molecular weight as a result of their depolymerization, fragmenting them into smaller molecules, which increases their water solubility 3,20,21,32 and ability to interact with free amino acids, peptides and pro-teins during the non-enzymatic Maillard reaction and in the process forming compounds that affect the beverage's color and aroma.
The non-enzimatic Maillard reaction is named after the French chemist Louis Camille Maillard (1878-1936), who in 1912 was the first to report how free amino acids derived from peptides and proteins interact with reducing sugars to form heterocyclic nitrogen compounds and melanoidins. It is considered as the most important reaction in the field of food chemistry since, through it the sensory characteristics are produced (eg, color, aroma and flavor) as well as a series of compounds at the expense of a significant reduction in nutritional value due to the essential amino acids degradation.
According to Moreira et al.32 the degree of resistance to thermal degradation varies depending on the type of high molecular weight polysaccharides. For example, when these are well roasted, when black coffee is produced, the galactomannans and arabinogalactans are thermally fragmented up to 36 and 60%, respectively.32
The galactomannans' higher degree of resistance to thermal degradation compared to arabinogalactans is attributed to the thermal resistance of their constituent monomers, which is described by the following gradient: D-Mannose > D-Galactose > L -Arabinose.32
According to Moreira et al.32 , as a consequence of grain roasting, losses of D-mannose monomers of 6 and 36% are recorded in lightly and well roasted grains (black coffees), respectively, thus evidencing the thermal degradation of galactomannans.
On the other hand, coffee polysaccharides (galactomannans, & arabinogalactans) are partially extracted by hot water (~ 6-12%) (Figure 6) 32 and although higher proportions can be extracted through more rigorous conditions, it would not be representative of what happens when the customer makes a cup of coffee.
Although arabinogalactans constitute the larger fraction extracted, through hot water, from raw coffee beans, galactomannans are the most abundant polysaccharides in the coffee beverage, thus increasing the infusion sedimentation 15,32 and viscosity,5,13,15,17 which is perceived in the mouth as a creamy sensation.5,21 This characteristic is known as the body of the drink.17,21

Figure 6. Coffee bean fractions are based on their degree of solubility. Adapted from Wang et al.51.
However, approximately 70% of the polysaccharides present in roast and ground coffee are retained in the remains recovered after the extraction of soluble compounds.15,17 The polysaccharide's insolubility is a consequence of their close association with the cell wall, which is a matrix with a very dense architecture.
One of the disadvantages of the reduced solubility of coffee polysaccharides is their limited availability for the study of their structural conformation, which is why many molecular structures published in the scientific literature are based on studies of the soluble fraction, which are not representative of most of the structural polysaccharides associated with the cell wall.
In summary, in coffee, the hidrosolubility of high molecular weight polysaccharides associated with the cell wall is the product of the synergic effect of: the number of lateral branches, the number of acetyl groups incorporated into its molecular structure, its thermal degradation (depolymerization and debranching) during roasting, the destabilization of the cell wall during roasting which causes its dilation and increases in the diameter of its pores and, the particle size to which the roasted bean is reduced by grinding. In general, the smaller the particle, the greater its exposure surface, which facilitates the removal of water-soluble compounds.
Quantification and structural characterization of polysaccharides
Carbohydrate determination analysis can be carried out by quantifying total carbohydrates in aqueous solutions and/or their structural characterization through chromatographic, spectroscopic, and spectrophotometric techniques, among others:
Quantification
There is a diversity of colorimetric methods for quantifying total carbohydrates in a biological sample, which include reagents such as anthrone, phenol, orcinol or resorcinol; However, the most widely used colorimetric method for determining carbohydrate concentration is the phenol-sulfuric acid method with its different variations.
In this method, adding sulfuric acid to aqueous carbohydrate solutions causes dehydration, favoring dehydration to furfural in the case of pentoses and 5-hydroxymethylfurfural (HMF) in hexoses. Furthermore, the added phenol interacts with the HMF facilitating the formation of complexes that allow the solution's coloration, enabling the quantification of carbohydrates through spectrophotometry.
This quantification method has advantages such as: easily applicable, sensitivity, speed of results and is appropriate for quantifying monosaccharides, oligosaccharides and polysaccharides.16,52
Structural characterization
The general steps for carbohydrate characterization involve:
a. The degreasing. The degreasing of plant material using apolar solvents such as n-hexane via the Soxhlet technique,39,53 allows the removal of the lipid from the heterogeneous matrix, which otherwise acts as interfering compounds in the analysis.
b. The removal. The degreased sample is subjected to extraction to recover the coffee beans' polysaccharide fraction. Extraction with hot water, ethylenediaminetetraacetic acid (EDTA), EDTA/NaOH, and NaOH at concentrations of 0.05, 1 and 4 M, among others, have been reported.35,39,49
c. The purification. The recovered extracts are subsequently subjected to processes of centrifugation, filtration, concentration, and purification by ultrafiltration, dialysis and lyophilization to retrieve fractions rich in purified polysaccharides.16,35,50,53
d. The structural characterization. Once the purified extracts have been isolated, it is necessary to know the monomers' identity that makes it up, so basic, enzymatic 41 or acid hydrolyses are performed, 50 which reveals the identity of the neutral sugars present in the purified fractions.
The hydrolysis of neutral sugars is carried out with H2SO4 or trifluoroacetic acid (TFA), using specific times and temperatures according to the analysis protocols. Hydrolysis allows the monomers to be released and subsequently transformed into their corresponding alditol acetate derivatives.41,49,50,53,54
Techniques such as high-performance liquid chromatography (HPLC) or gas chromatography (GC) are used to identify the released and derivatized monosaccharides.
e. Chain sequencing analysis. To know the positions of the bonds that the monomers present, the methylation technique is used, this allows the conversion of all the free hydroxyl groups into methoxyl groups, followed by a hydrolytic rupture of the glycosidic bonds, with subsequent conversion of the monomers into derivatives partially methylated acetylated, which are identified by gas chromatography coupled to a mass detector (GC-MS), high-performance ion exchange chromatography 39,41 and subsequent GC quantification.16,35,49,50
f. The distribution of carbohydrates molecular weights in the extract is performed by high-performance size exclusion chromatography monitored by a refractive index detector, which allows distinguishing molecule populations based on their molecular weight.16,53,54
Another powerful technique for characterizing carbohydrate polymers is one-dimensional and two-dimensional Nuclear Magnetic Resonance (NMR); This has many advantages over the abovementioned techniques.50,53 It is a non-destructive technique used to identify the carbon and anomeric proton sugar signals, allowing the monomers to be assigned their anomeric configurations. Correlations and chemical shifts obtained by NMR can be used as fingerprints to identify monomers and binding sequences.50
Most of the consulted literature reference is made to the study published by Oosterveld et al.55, which is used as a basis to carry out carbohydrate's structural characterization, while other authors have made process modifications to improve the efficiency of the analysis.
Metabolism and bioactive capacity
Human digestive enzymes do not metabolize most polysaccharides present in the coffee drink, so they become part of the dietary fiber,6,15,30,32 which reaches concentrations ranging between 0.14-0.65 g 100 ml-1 or 0.47-0.75 g 100 ml-1 of infusion depending on the species (C. arabica vs. C. canephora), the degree of roasting, the particle size, the beverage preparation technique and the employed determination method.6,32
On the other hand, by not being absorbed through the intestinal wall (duodenum), dietary fiber manages to reach the colon, where mono-mannans and oligo-mannans are converted into short-chain fatty acids 32,34; In addition, dietary fiber also contributes to the maintenance of beneficial microorganisms in the colon, such as bifidobacteria.15,32,56
However, the digestion of type II arabinogalactans is less efficient. This is especially observed in the L-arabinose monomers in the polymer's side branches, which are covalently linked to each other through α(1→5) bonds.32 This could be attributed to the presence of L-rhamnose units located at the terminal positions of the side branches and which are linked to the L-arabinose monomers through the same type of glycosidic bond (Figure 5).
Carbohydrates and drink quality
Although the polysaccharides present in the coffee drink do not affect its flavor, monosaccharides (glucose and fructose) and disaccharides such as sucrose (glucose monomer + fructose monomer), are associated with the infusion flavor.
Consequently, the higher the sucrose content in the coffee beans, the more intense their flavor will be.57 Interestingly, when sucrose is thermally degraded during grain roasting, it releases its constituent monomers, of which fructose is almost twice as sweet as sucrose per se.
The best cup quality conferred to C. arabica is attributed, in part, to its higher sucrose content in the dry and raw bean,33,57, which is responsible for more than 90% of the oligosaccharides present in it.3 However, according to Privat et al. 25 although a good part of the sucrose is degraded during roasting, a fraction of it remains in the roasted bean (0.4-2.8% on a dry matter basis), contributing to the beverage flavor.
On the other hand, even when sucrose is largely degraded during the early stages of grain roasting, the volatile and non-volatile compounds generated from it, such as aliphatic acids (eg, acetic, formic and lactic acids), among others (eg, furans, aldehydes, etc.) product of their interaction with other compounds during the non-enzymatic Maillard reaction (Figure 7) also contribute to the infusion flavor.21,26,28
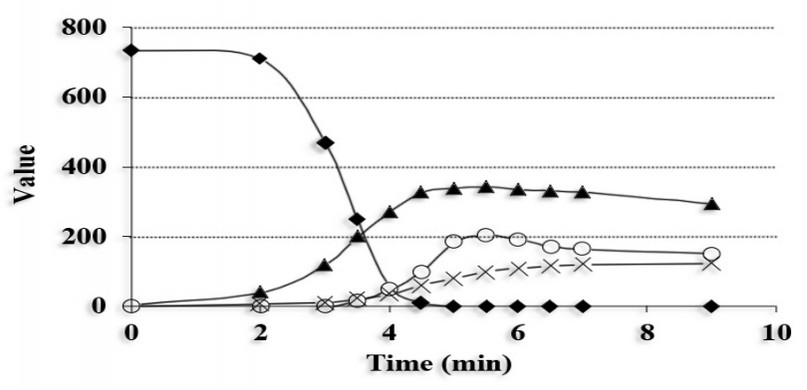
Figure 7. Sucrose degradation and accumulation of derived compounds during grain roasting.
♦= Sucrose, ▲= acetic acid, O: formic acid, X: lactic acid. Adapted from Wei et al.21
CONCLUSIONS
Once roasted, the coffee grain is ground to fine particle size and exposed to hot water, thus inducing the particle's humidification and solubilization of water-soluble solutes, concomitant with the hydrolysis of some insoluble compounds due to the high temperature. Subsequently, the aqueous extract is filtered to separate the insoluble materials (bagasse), thus recovering an aqueous solution rich in water-soluble compounds, which imparts the typical coffee beverage taste and smell.
Hydrosoluble carbohydrates constitute a major fraction of the water-soluble compounds in the coffee infusion. In general, Robusta coffees produce 10% more soluble solutes than Arabica coffees, which causes an increase in their demand since they are preferred in the production of instant coffees; However, they are usually blended with Arabica coffees to reduce production costs as well as improve the quality of the final product.
Additionally, the arabinogalactans and galactomannans extracted in aqueous solution from Robusta coffees have a greater number of lateral branches than those found in Arabica coffees, which in turn would explain their greater water solubility, thus facilitating its extraction.
The synergic effect of polysaccharides' greater number of lateral branches added to a greater degree of water solubility would explain the greater percentage of extraction of soluble solutes in Robusta varieties; even when they have a lower content of galactomannans (19-22%) and arabinogalactans (6-8%) compared to Arabica varieties (25-30% & 9-13% respectively).
On the other hand, during the soluble coffees manufacture, after the extraction of the water-soluble compounds, 70% of the roasted and ground bean's polysaccharides remain in the bagasse. In addition, the undigestible bagasse' polysaccharides are part of the total fiber, of which 16-35% is soluble, and has an antioxidant capacity similar to that observed in other products such as red wine and peaches, which is why it can be considered as dietary fiber.
The bagasse is a low-density residue that remains after the extraction of soluble materials during the coffee beverage preparation. One ton of raw coffee seed is estimated to produce ~ 650 kg of bagasse. Approximately 45.3% of the bagasse's chemical composition corresponds to neutral detergent fiber composed of hemicellulose, cellulose and lignin, and 29.8% of acid detergent fiber composed of cellulose and lignin; However, the bagasse also contains other high molecular weight polysaccharides such as galactomannans and arabinogalactans.
The world coffee bagasse production is estimated to be ~ 6 000 000 mt/year and since it is not edible, it does not have a commercial value; thus, it constitutes a serious environmental problem since it is usually discarded in large piles which, due to its humidity, start the fermentation process that can lead to spontaneous combustion.
In conclusion, despite the coffee grain's rich carbohydrate composition, it is not water-soluble and resistant to human gastrointestinal digestive acids and enzymes limiting its use in the food industry.
Funding: This research received no external funding.
Acknowledgments
The authors thank the Office for the Scientific, Humanistic and Technological Research (DICIHT) of the National Autonomous University of Honduras for their sponsorship during the publication of this article.
Conflicts of Interest: The authors declare no conflict of interest.
REFERENCES
1. Farah A. Coffee constituents. In: Coffee. 1 ed. Chu Y-F, editor. Wiley-Blackwell: Oxford, UK; 2012. p. 21-58. doi: 10.1002/9781119949893.ch2.
2. Flament I.; Bessière-Thomas Y. Coffee flavor chemistry. 1 ed. John Wiley & Sons: West Sussex, ΡΟ19 1UD, England; 2001 November. doi: NO_DOI.
3. Esquivel P.; Jiménez V.M. Functional properties of coffee and coffee by-products. Food Res Int. 2012;46(2):488-95. doi: 10.1016/j.foodres.2011.05.028.
4. Matulová M.; Capek P.; Kaneko S.; Navarini L.; Liverani F.S. Structure of arabinogalactan oligosaccharides derived from arabinogalactan-protein of Coffea arabica instant coffee powder. Carbohydr Res. 2011;346(8):1029-36. doi: 10.1016/j.carres.2011.03.016.
5. Simoes J.; Madureira P.; Nunes F.M.; Domingues Mdo R.; Vilanova M.; Coimbra M.A. Immunostimulatory properties of coffee mannans. Mol Nutr Food Res. 2009;53(8):1036-43. doi: 10.1002/mnfr.200800385.
6. Ludwig I.A.; Clifford M.N.; Lean M.E.; Ashihara H.; Crozier A. Coffee: biochemistry and potential impact on health. Food Funct. 2014;5(8):1695-717. doi: 10.1039/c4fo00042k.
7. Oestreich-Janzen S. Chemistry of coffee. In: Comprehensive natural products II. Elsevier: Oxford; 2010. p. 1085-117. doi: 10.1016/b978-008045382-8.00708-5.
8. Poisson L.; Blank I.; Dunkel A.; Hofmann T. The chemistry of roasting - Decoding flavor formation. In: The craft and science of coffee. Folmer B, editor. Academic Press: 2017. p. 273-309. doi: 10.1016/b978-0-12-803520-7.00012-8.
9. Daniel D.; Lopes F.S.; Santos V.B.D.; do Lago C.L. Detection of coffee adulteration with soybean and corn by capillary electrophoresis-tandem mass spectrometry. Food Chem. 2018; 243:305-10. doi: 10.1016/j.foodchem.2017.09.140.
10. Redgwell R.; Fischer M. Coffee carbohydrates. Braz J Plant Physiol. 2006;18(1):165-74. doi: 10.1590/s1677-04202006000100012.
11. Moreira A.S.; Nunes F.M.; Domingues M.R.; Coimbra M.A. Coffee melanoidins: structures, mechanisms of formation and potential health impacts. Food Funct. 2012;3(9):903-15. doi: 10.1039/c2fo30048f.
12.Velez Muriel G.A. Extraction and characterization of arabinogalactan-proteins (AGP) from green coffee beans. Wageningen University: The Netherlands; 2013. doi: NO_DOI.
13. Tarzia A.; Dos Santos Scholz M.B.; De Oliveira Petkowicz C.L. Influence of the postharvest processing method on polysaccharides and coffee beverages. Int J Food Sci Technol. 2010;45(10):2167-75. doi: 10.1111/j.1365-2621.2010.02388. x.
14. Joet T.; Laffargue A.; Salmona J.; Doulbeau S.; Descroix F.; Bertrand B., et al. Metabolic pathways in tropical dicotyledonous albuminous seeds: Coffea arabica as a case study. New Phytol. 2009;182(1):146-62. doi: 10.1111/j.1469-8137.2008.02742.x.
15. Ballesteros L.F.; Cerqueira M.A.; Teixeira J.A.; Mussatto S.I. Characterization of polysaccharides extracted from spent coffee grounds by alkali pretreatment. Carbohydr Polym. 2015; 127:347-54. doi: 10.1016/j.carbpol.2015.03.047.
16. Fischer M.; Reimann S.; Trovato V.; Redgwell R.J. Polysaccharides of green Arabica and Robusta coffee beans. Carbohydr Res. 2001;330(1):93-101. doi: 10.1016/s0008-6215(00)00272-x.
17. Campos-Vega R.; Loarca-Piña G.; Vergara-Castañeda H.A.; Oomah B.D. Spent coffee grounds: a review on current research and future prospects. Trends Food Sci Technol. 2015;45(1):24-36. doi: 10.1016/j.tifs.2015.04.012.
18. Wang X.; Lim L.-T. Physicochemical characteristics of roasted coffee. In: Coffee in health and disease prevention. Academic Press: San Diego; 2015. p. 247-54. doi: 10.1016/b978-0-12-409517-5.00027-9.
19. Pre M.; Caillet V.; Sobilo J.; McCarthy J. Characterization and expression analysis of genes directing galactomannan synthesis in coffee. Ann Bot. 2008;102(2):207-20. doi: 10.1093/aob/mcn076.
20. Hwang C.-F.; Chen C.-C.; Ho C.-T. Contribution of coffee proteins to roasted coffee volatiles in a model system. Int J Food Sci Technol. 2012;47(10):2117-26. doi: 10.1111/j.1365-2621.2012.03078.x.
21. Wei F.; Tanokura M. Chemical changes in the components of coffee beans during roasting. In: Coffee in health and disease prevention. Academic Press: San Diego; 2015. p. 83-91. doi: 10.1016/b978-0-12-409517-5.00010-3.
22. Wei F.; Tanokura M. Organic compounds in green coffee beans. In: Coffee in health and disease prevention. Academic Press: San Diego; 2015. p. 149-62. doi: 10.1016/b978-0-12-409517-5.00017-6.
23. Ballesteros L.F.; Teixeira J.A.; Mussatto S.I. Extraction of polysaccharides by autohydrolysis of spent coffee grounds and evaluation of their antioxidant activity. Carbohydr Polym. 2017; 157:258-66. doi: 10.1016/j.carbpol.2016.09.054.
24. Sunarharum W.B.; Williams D.J.; Smyth H.E. Complexity of coffee flavor: a compositional and sensory perspective. Food Res Int. 2014; 62:315-25. doi: 10.1016/j.foodres.2014.02.030.
25. Privat I.; Foucrier S.; Prins A.; Epalle T.; Eychenne M.; Kandalaft L., et al. Differential regulation of grain sucrose accumulation and metabolism in Coffea arabica (Arabica) and Coffea canephora (Robusta) revealed through gene expression and enzyme activity analysis. New Phytol. 2008;178(4):781-97. doi: 10.1111/j.1469-8137.2008.02425.x.
26. Cheng B.; Furtado A.; Smyth H.E.; Henry R.J. Influence of genotype and environment on coffee quality. Trends Food Sci Technol. 2016;57:20-30. doi: 10.1016/j.tifs.2016.09.003.
27. Campa C.; Ballester J.F.; Doulbeau S.; Dussert S.; Hamon S.; Noirot M. Trigonelline and sucrose diversity in wild Coffea species. Food Chem. 2004;88(1):39-43. doi: 10.1016/j.foodchem.2004.01.020.
28. Perrone D.; Donangelo C.M.; Farah A. Fast simultaneous analysis of caffeine, trigonelline, nicotinic acid and sucrose in coffee by liquid chromatography-mass spectrometry. Food Chem. 2008;110(4):1030-35. doi: 10.1016/j.foodchem.2008.03.012.
29. Mishra M.K.; Slater A. Recent advances in the genetic transformation of coffee. Biotechnol Res Int. 2012; 2012:580857. doi: 10.1155/2012/580857.
30. Turnbull A.J. Coffee. Nutr Bull. 1981;6(3):153-65. doi: 10.1111/j.1467-3010. 1981.tb00494. x.
31. Stadler R.H.; Theurillat V. Acrylamide in coffee. In: Coffee. Chu Y-F, editor. Wiley-Blackwell: 2012. p. 259-73. doi: 10.1002/9781119949893.ch15.
32. Moreira A.S.P.; Nunes F.M.; Domingues M.R.M.; Coimbra M.A. Galactomannans in coffee. In: Coffee in health and disease prevention. Academic Press: San Diego; 2015. p. 173-82. doi: 10.1016/b978-0-12-409517-5.00019-x.
33. Stredansky M.; Redivo L.; Magdolen P.; Stredansky A.; Navarini L. Rapid sucrose monitoring in green coffee samples using multienzymatic biosensor. Food Chem. 2018; 254:8-12. doi: 10.1016/j.foodchem.2018.01.171.
34. van Dam J.E.G.; Harmsen P. Coffee residues utilization. Report. Wageningen UR Food & Biobased Research, VLAG, Technology FBBNF; 2010. Disponible en: NO_DOI.
35. Simões J.; Nunes F.M.; Domingues M.d.R.M.; Coimbra M.A. Structural features of partially acetylated coffee galactomannans presenting immunostimulatory activity. Carbohydr Polym. 2010;79(2):397-402. doi: 10.1016/j.carbpol.2009.08.020.
36. Ballesteros L.F.; Teixeira J.A.; Mussatto S.I. Chemical, functional, and structural properties of spent coffee grounds and coffee silverskin. Food Bioproc Tech. 2014;7(12):3493-503. doi: 10.1007/s11947-014-1349-z.
37. Mussatto S.I.; Carneiro L.M.; Silva J.P.A.; Roberto I.C.; Teixeira J.A. A study on chemical constituents and sugars extraction from spent coffee grounds. Carbohydr Polym. 2011;83(2):368-74. doi: 10.1016/j.carbpol.2010.07.063.
38. Kovalcik A.; Obruca S.; Marova I. Valorization of spent coffee grounds: a review. Food Bioprod Process. 2018; 110:104-19. doi: 10.1016/j.fbp.2018.05.002.
39. Oosterveld A.; Voragen A.G.J.; Schols H.A. Effect of roasting on the carbohydrate composition of Coffea arabica beans. Carbohydr Polym. 2003;54(2):183-92. doi: 10.1016/s0144-8617(03)00164-4.
40. Redgwell R.J.; Trovato V.; Curti D.; Fischer M. Effect of roasting on degradation and structural features of polysaccharides in Arabica coffee beans. Carbohydr Res. 2002;337(5):421-31. doi: 10.1016/s0008-6215(02)00010-1.
41. Nunes F.M.; Domingues M.R.; Coimbra M.A. Arabinosyl and glucosyl residues as structural features of acetylated galactomannans from green and roasted coffee infusions. Carbohydr Res. 2005;340(10):1689-98. doi: 10.1016/j.carres.2005.05.002.
42. Oliveira Petkowicz C.L.d. Polysaccharides in coffee and their relationship to health. In: Coffee in health and disease prevention. Preedy VR, editor. Academic Press: San Diego; 2015. p. 163-72. doi: 10.1016/b978-0-12-409517-5.00018-8.
43. Navarini L.; Rivetti D. Water quality for Espresso coffee. Food Chem. 2010;122(2):424-28. doi: 10.1016/j.foodchem.2009.04.019.
44. Reichardt N.; Gniechwitz D.; Steinhart H.; Bunzel M.; Blaut M. Characterization of high molecular weight coffee fractions and their fermentation by human intestinal microbiota. Mol Nutr Food Res. 2009;53(2):287-99. doi: 10.1002/mnfr.200700509.
45. Rufián-Henares J.A.; Pastoriza S. Melanoidins in coffee. In: Coffee in health and disease prevention. Academic Press: San Diego; 2015. p. 183-88. doi: 10.1016/b978-0-12-409517-5.00020-6.
46. Redgwell R.J.; Curti D.; Fischer M.; Nicolas P.; Fay L.B. Coffee bean arabinogalactans: acidic polymers covalently linked to protein. Carbohydr Res. 2002;337(3):239-53. doi: 10.1016/s0008-6215(01)00316-0.
47. Redgwell R.J.; Schmitt C.; Beaulieu M.; Curti D. Hydrocolloids from coffee: physicochemical and functional properties of an arabinogalactan–protein fraction from green beans. Food Hydrocoll. 2005;19(6):1005-15. doi: 10.1016/j.foodhyd.2004.12.010.
48. De Maria C.A.B.; Trugo L.C.; Neto F.R.A.; Moreira R.F.A.; Alviano C.S. Composition of green coffee water-soluble fractions and identification of volatiles formed during roasting. Food Chem. 1996;55(3):203-07. doi: 10.1016/0308-8146(95)00104-2.
49. Oosterveld A.; Harmsen J.S.; Voragen A.G.J.; Schols H.A. Extraction and characterization of polysaccharides from green and roasted Coffea arabica beans. Carbohydr Polym. 2003;52(3):285-96. doi: 10.1016/s0144-8617(02)00296-5.
50. Capek P.; Matulová M.; Navarini L.; Suggi-Liverani F. Structural features of an arabinogalactan-protein isolated from instant coffee powder of Coffea arabica beans. Carbohydr Polym. 2010;80(1):180-85. doi: 10.1016/j.carbpol.2009.11.016.
51.Wang X.; Lim L.T. Investigation of CO2 precursors in roasted coffee. Food Chem. 2017; 219:185-92. doi: 10.1016/j.foodchem.2016.09.095.
52. López-Legarda X.; Taramuel-Gallardo A.; Arboleda-Echavarría C.; Segura-Sánchez F.; Restrepo-Betancur L.F. Comparación de métodos que utilizan ácido sulfúrico para la determinación de azúcares totales. Revista Cubana de Química. 2017;29(2):180-98.
53. Navarini L. Polysaccharides from hot water extracts of roasted Coffea arabica beans: isolation and characterization. Carbohydr Polym. 1999;40(1):71-81. doi: 10.1016/s0144-8617(99)00032-6.
54. Oosterveld A.; Beldman G.; Schols H.A.; Voragen A.G.J. Arabinose and ferulic acid rich pectic polysaccharides extracted from sugar beet pulp. Carbohydr Res. 1996; 288:143-53. doi: 10.1016/s0008-6215(96)90791-0.
55. Oosterveld A.; Beldman G.; Schols H.A.; Voragen A.G. Arabinose and ferulic acid rich pectic polysaccharides extracted from sugar beet pulp. Carbohydrate Research. 1996;288:143-53.
56.Vitaglione P.; Fogliano V.; Pellegrini N. Coffee, colon function and colorectal cancer. Food Funct. 2012;3(9):916-22. doi: 10.1039/c2fo30037k.
57. Somporn C.; Kamtuo A.; Theerakulpisut P.; Siriamornpun S. Effect of shading on yield, sugar content, phenolic acids and antioxidant property of coffee beans (Coffea arabica L. cv. Catimor) harvested from north-eastern Thailand. J Sci Food Agric. 2012;92(9):1956-63. doi: 10.1002/jsfa.5568.
Received: 20 March 2022 / Accepted: 25 july 2022 / Published:15 Agoust 2022
Citation: Portillo, O.R..; Arévalo, A.C. Coffee's carbohydrates. A critical review of scientific literature. Revis Bionatura 2022;7(3) 11. http://dx.doi.org/10.21931/RB/2022.07.03.11
