2020.05.03.6
Files > Volume 5 > Vol 5 No 3 2020
INVESTIGATION / RESEARCH
Seasonal abundance and distribution of phytoplankton in Tanintharyi coastal waters, southern Myanmar
Khin Khin Gyi1, Wint Thuzar Nwe2, Zin Zin Zaw3 and Khin Khin San4
Available from: http://dx.doi.org/10.21931/RB/2020.05.03.6
ABSTRACT
The seasonal abundance and distribution of phytoplankton along the Tanintharyi coastal waters were investigated for 24 months from June 2013 to June 2015. A wide fluctuation in cell abundance 72,450-714,396 cells/l at Kawthaung, 47,416-947,501 cells/l at Myeik, 8,930-28,439 cells/l at Kampani, 8,976-17,888 cells/l at Ye and 5,162-16,986 cells/l at Setse were noted during the study period. Amongst, Kawthaung and Myeik stations had the highest phytoplankton abundance, whereas Ye and Setse stations showed remarkably lower abundance. It was noted that Ye and Setse stations were much influenced by freshwater discharge from the Thanlwin River, which deposited huge tons of sediments. The water clarity was lower at these stations compare with others. Therefore, turbidity may affect the occurrence and species abundance of phytoplankton. A clear seasonal trend was found at all five stations with a sharp increase in the pre-monsoon months and a gradual decrease in the monsoon and post-monsoon periods.
Keywords: phytoplankton, Tanintharyi, water clarity, turbidity,
INTRODUCTION
Seasonal replacements of phytoplankton abundance and composition differ in various environments1. The values change in species composition, and a variety of environmental factors can mediate the dominance of phytoplankton. Also, the possible causes of the seasonal phytoplankton bloom correlated with the seasonal increase of nutrient supply that primarily increased the abundance of phytoplankton.2 Other processes, such as meteorological and hydrological events, may drive non-equilibrium dynamics and enhance species diversity. 3
On the other hand, transient forms are superimposed to the spatial measurement in such a way that the recurrence and concentration of a specific occasion may shift concurring to the neighborhood morphometry, profundity, favoring, or not and species coexistence. Agreeing to asset competition hypothesis, species differing qualities are precisely corresponding to the number of assets that are restricting at a given time, though natural variance or unsettling influence improves the levels of diversity 4,10
Species diversity, composition, distributions, and abundance of phytoplankton are the critical factors in ecology, directly linked with the regulation and functioning of the ecosystems that are used to assess the biological integrity of the water body 5. Because the phytoplankton community composition impacts the functioning of the aquatic ecosystems and the global climate, it is essential to understand what factors govern the phytoplankton community assembly and the dynamics.6 The objective of the study is to obtain a better understanding of the seasonal abundance and distribution of phytoplankton along the Tanintharyi coastline.7
MATERIALS AND METHODS
Sampling site
Phytoplankton samples were collected at the five stations along the Tanintharyi coastline, namely, Kawthaung (Lat. 9º 58. 204ʹ N, Long. 98º 33. 701ʹ E), Myeik (Lat. 12º 26. 186ʹ N, Long. 98º 35. 461ʹ E), Kampani (Lat. 14º 05. 288ʹ N, Long. 98º 04. 143ʹ E), Ye (Lat. 15º 11. 585ʹ N, Long. 97º 47. 518ʹ E) and Setse (Lat. 15º 56. 965ʹ N, Long. 97º 36. 330ʹ E) from June 2013 to June 2015 (Fig.1).

Figure 1. Map showing the sample collection sites in Taninthayi coastal waters.
The sampling areas were influenced by the monsoon season. Therefore, a calendar year was divided into three seasons for ecological purposes. The division for three seasons was based on changes in the temperature in the annual cycle of the region. The seasons were recognized as the pre-monsoon period (February to May), the monsoon period (June to September) and the post-monsoon period (October to January).
Sample collection
A small-mesh phytoplankton net of 20 μm is used in the sample collection. The phytoplankton samples of the surface water were collected by a plastic bucket of known volume water, 60L. Then, the water passed through the mesh fixed to the bottom of a plastic cylinder. Care was taken to wash all the cells off the sieve. Samples were preserved immediately in a 1% formaldehyde solution for the species identification and counting.
Sample analysis
Sedgwick-Rafter counting chamber was used to count the phytoplankton following the method described by LeGresley and McDermott 20107. Before analysis, the content in the sample bottle was turned gently for making the samples homogeneous. Three replicates of 1mL subsample were taken from a well-mixed sample by using a pipette, and the sample aliquot is then dispensed into the counting cell, and then placed under the light microscope at 40x and 10x magnification. The counting unit of all phytoplankton species is expressed in Cells/L.
RESULTS
Kawthaung Station: The abundance of phytoplankton ranged from 72,450-714,396 Cells/L, was observed during the study period from June 2013 to June 2015. The maximum phytoplankton abundance, 714,396 Cells/L was noticed in the pre-monsoon period, especially in April (2015) which was followed by 624,999 Cells/L in April (2014), 493,976 Cells/L in May (2014) and 448,321 Cells/L in May (2015). In contrast, the minimum cell abundance 72,450 Cells/L, 79,000 Cells/L, and 94,862 Cells/L were observed in November (2013), December (2013), and (2014) of the post-monsoon period (Fig. 2).

Figure 2. Monthly variations of phytoplankton abundance (Cells/L) at Kawthaung station from June 2013 to June 2015.
During the study period, Guinardia striata (155,000 Cells/L), Thalassionema nitzschioides (147,583 Cells/L), Hemiaulus sinensis (135,000 Cells/L), Lauderia annulata (44,445 Cells/L), Nitzschia seriata (38,500 Cells/L), Rhizosolenia setigera (35,439 Cells/L), Guinardia flaccida (33,333 Cells/L), Eucampia cornuta (28,333 Cells/L), Thalassiosira subtilis (27,833 Cells/L), Rhizosolenia imbricata (23,333 Cells/L), Bellerochea horologicalis (21,667 Cells/L), Odontella sinensis (20,333 Cells/L), Ditylum sol (17,667 Cells/L), Chaetoceros curvisetus (17,444 Cells/L), Thalassionema frauenfeldii (15,500 Cells/L), Melosira nummuloides (13,333 Cells/L), Chaetoceros subtilis (10,833 Cells/L), Rhizosolenia calcar-avis (10,000 Cells/L) and Ceratium furca (5,334 Cells/L) were observed as dominant species in Kawthaung coastal waters. Hyalodiscus subtilis, Planktoniella sol, Asteromphalus flabellatus, Biddulphia rhombus, Triceratium reticulum, Streptotheca indica, Syringidium americanum, Bacteriastrum hyalium var. princeps, Striatella unipunctata, Lyrella lyra, Campyloneis grevillei, and Metadinophysis sinensis were rarely observed, 50-333 Cells/L.
Myeik Station: Phytoplankton abundance varied from 47,416 to 947,501 Cells/L. The maximum values 947,501 and 793,887 Cells/L were seen in April (2014) and (2015), which were followed by 543,892 and 426,379 Cells/L in May (2015 and 2014). In the following months of March (2014, 2015), June (2013, 2014, 2015), and July (2013, 2014), the cell numbers were recorded in the range between 312,248-398,491 Cells/L. Then, the numbers were continually dropped until post-monsoon months and reached the least value of 47,416 Cells/L in December (2013). During the study period, the abundance of phytoplankton was high in the pre-monsoon season, and the peak values were noticed in April, and then in February and May (Fig. 3).

Figure 3. Monthly variations of phytoplankton abundance (Cells/L) at Myeik station from June 2013 to June 2015.
Thalassionema nitzschioides (263,667 Cells/L), Ditylum sol (142,167 Cells/L), Melosira borreri (120,833 Cells/L), Bellerochea horologicalis (98,333 Cells/L), Odontella sinensis (61,000 Cells/L), Thalassiosira subtilis (58,333 Cells/L), Rhizosolenia setigera (53,583 Cells/L), Nitzschia seriata (41,333 Cells/L), Lauderia annulata (28,500 Cells/L), Thalassionema frauenfeldii (25,833), Odontella mobiliensis (19,667 Cells/L), Hemialus sinensis (18,167 Cells/L), Cylindrotheca closterium (13,667 Cells/L), Ceratium furca (13,333 Cells/L), Nitzschia sigma (10,917 Cells/L) and N. longissimia (10,167 Cells/L) were plentifully collected during the study period. Planktoniella sol, Eunotogramma laevis, Biddulphia biddulphiana, B.rhombus, Triceratium dubium, Streptotheca indica, Syringidium americanum, Centronella reicheltii, Lyrella lyra, Oestrupia musca, Amphora cymbifera, A. lineolata, Metadinophysis sinensis, and Cladopyxis hemibranchiata were rarely observed.
Kampani Station: Phytoplankton abundance extended in the range between 8,930 and 28,439 Cells/L. The maximum abundance 24,358 Cells/L, 27,904 Cells/L, 21,978 Cells/L and 25,483 Cells/L, 28,439 Cells/L, 22,438 Cells/L were observed in pre-monsoon months, especially in March, April, and May of 2014 and 2015. During the monsoon period, the abundance fluctuated between 11,004 and 19,597 Cells/L. In contrast, the lowest number of 8,930 Cells/L was found in October 2013 of the post-monsoon period (Fig. 4).

Figure 4. Monthly variations of phytoplankton abundance (Cells/L) at Kampani station from June 2013 to June 2015.
During the study period, Fragilaria capucina (5,792 Cells/L), Bacteriastrum delicatulum (5,089 Cells/L), Paralia sulcata (4,700 Cells/L), Bacteriastrum hyalinum (4,644 Cells/L), Thalassionema nitzschioides (3,471 Cells/L), Cylindrotheca closterium (3,108 Cells/L), Tabellaria fenestrata (3,050 Cells/L), Climacosphenia moniligera (2,844 Cells/L), Rhizosolenia imbricata (2,833 Cells/L) Ceratium furca (2,778 Cells/L), Prorocentrum micans (2,461 Cells/L), Cyclotella striata (1,739 Cells/L), Dictyocha finbula (1,613 Cells/L), Chaetoceros diversus (1,250 Cells/L) and Ditylum sol (1,125 Cells/L) were abundantly collected. In contrast, Druridgea compressa, Actinoptychus senarius, Streptotheca indica, Centronella reicheltii, Cymbella lanceolata, Oestrupia musca, Diploneis splendida, Amphora cymbifera, Plagiodiscus nervatus, Campylodiscus noricus, Metadinophysis sinensis, Cladopyxis hemibranchiata, Oxytoxum sceptrum, Podolampas palmipes, and Protoperidinium compressum were rarely observed.
Ye Station: Phytoplankton abundance ranged between 8,976-17,888 Cells/L. The maximum number 17,888 Cells/L was found in April (2014) followed by 17,439 Cells/L in April (2015) and 16,967 Cells/L in May (2014) whereas the minimum number 8,976 was observed in October 2013, followed by 9,877 Cells/L in October 2014, respectively. A seasonal trend of phytoplankton abundance with decreased values was noted during the monsoon and post-monsoon periods. However, the numbers of phytoplankton were progressively increased and stretched to maximum abundance in April of the pre-monsoon period (Fig. 5).
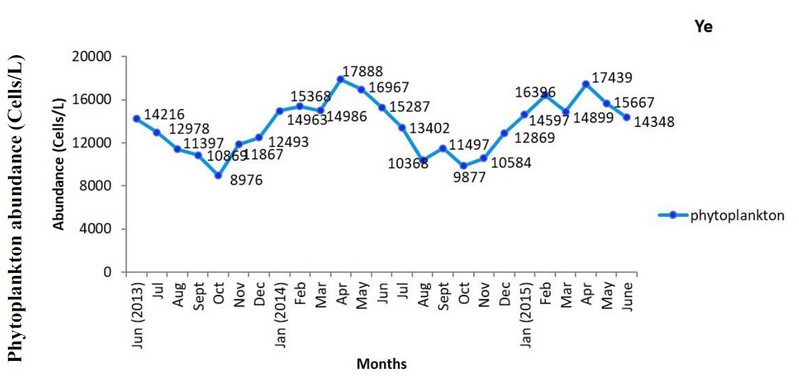
Figure 5. Monthly variations of phytoplankton abundance (Cells/L) at Ye station from June 2013 to June 2015.
The phytoplankton such as Rhizosolenia setigera (5,872 Cells/L), Asterionellopsis glacialis (4,078 Cells/L), Skeletonema costatum (3,583 Cells/L), Fragilaria crotonensis (2,894 Cells/L), Ditylum sol (2,106 Cells/L), Prorocentrum micans (1,892 Cells/L), Cylindrotheca closterium (1,672 Cells/L), Thalassionema nitzschioides (1,511 Cells/L), Proboscia alata (1,328 Cells/L), Chaetoceros subtilis (1,149 Cells/L), Thalassiosira rotula (1,083 Cells/L) and Ceratium furca (856 Cells/L) were plentifully observed during the study period. Conversely, Coscinodiscus asteromphalus, Triceratium favus, Syringidium americanum, Lithodesmium undulatum, Centronella reicheltii, Synedra unla, Cymbella lanceolata, Diploneis smithii, Pleurosigma pelagicum, and Gonyaulax spinifera were rarely collected.
Setse Station: Phytoplankton abundance varied from 5,162 to 16,986 Cells/L. The maximum number 16,986 Cells/L was recorded in April (2015), followed by 16,935 Cells/L in April (2014) and 16,739 Cells/L in March (2015) of pre-monsoon months. On the other hand, the minimum abundance of 5,162 and 7,426 Cells/L were seen in the heavy rainfall months, especially in August (2013 and 2014). The number of phytoplankton again increased to 9,633-11,452 Cells/L in the post-monsoon period of October, November, and December 2013 and 2014 (Fig. 6).

Figure 6. Monthly variations of phytoplankton abundance (Cells/L) at Setse stations from June 2013 to June 2015.
Fragilaria crotonensis (5,944 Cells/L), Ceratium furca (4,442 Cells/L), Thalassionema nitzschioides (3,983 Cells/L), Lauderia annulata (3,542 Cells/L), Dinophysis caudata (2,146 Cells/L), Prorocentrum micans (2,100 Cells/L), Cylindrotheca closterium (1,694 Cells/L), Nitzschia seriata (1,675 Cells/L), Chaetoceros densus (1,529 Cells/L), Ditylum sol (1,217 Cells/L), Thalassiosira subtilis (1,083 Cells/L), Thalassionema frauenfeldii (1,033 Cells/L), Odontella mobiliensis (1,028 Cells/L), Proboscia alata (1,025 Cells/L), Coscinodiscus lineatus (838 Cells/L), Chaetoceros lorenziauns (838 Cells/L) and Paralia sulcata (769 Cells/L) were abundantly seen during sampling periods. In contrast, Asteromphalus cleveanus, Rhizosolenia setigera, Bacteriastrum varians, Climacosphenia moniligera, Grammatophora marina, Surirella ovalis, and Ceratium tripos were hardly seen during the study period.
DISCUSSION
The present study was mainly emphasized on the seasonal variations of phytoplankton abundance at the five stations of Tanintharyi coastal waters, namely, Kawthaung, Myeik, Kampani, Ye and Setse. A wide fluctuation in cell abundance was noted at all stations such as 72,450-714,396 Cells/L at Kawthaung, 47,416-947,501 Cells/L at Myeik, 8,930-28,439 Cells/L at Kampani, 8,976-17,888 Cells/L at Ye and 5,162-16,986 Cells/L at Setse, respectively. The highest cell abundance was observed at Kawthaung and Myeik whereas the least abundance was detected in Ye and Setse stations. It was noted that a clear seasonal trend in phytoplankton abundance was seen at all five stations with a sharp peak in the pre-monsoon period, especially in April, after that a gradual decrease in monsoon and post-monsoon periods.
The 42 common phytoplankton species observed at Tanintharyi coastal waters were Melosira nummuloides, M. borreri, Thalassiosira subtilis, T. rotula, Skeletonema costatum, Paralia sulcata, Cyclotella striata, Lauderia annulata, Coscinodiscus lineatus, C. radiatus, Odontella sinensis, O. mobiliensis, Eucampia cornuta, Hemiaulus sinensis, Bellerochea horologicalis, Ditylum sol, Rhizosolenia imbricata, R.setigera, Proboscia alata, Guinardia striata, G. flaccida, Bacteriastrum delicatulum, B. hyalinum, Chaetoceros curvisetus, C. diversus, C. densus, C. lorenzianus, C. subtilis, Asterionellopsis glacialis, Fragilaria crotonensis, F. capucina, Tabellaria fenestrata, Climacosphenia moniligera, Thalassionema nitzschioides, T. frauenfeldii, Nitzschia sigma, N. longissimia, N. seriata, Cylindrotheca closterium, Prorocentrum micans, Dinophysis caudata, and Ceratium furca. Taylor 19758 reported Guinardia flaccida was noted as the predominant diatom species in Thailand which was in agreement with the present result found at Kawthaung station. Thalassionema nitzschioides and T. frauenfeldii were abundantly seen at Tanintharyi waters throughout the present study period agrees well with the finding of Kamba and Yuki 19805. Aquino et al. 20151 reported Odontella mobiliensis, Chaetoceros subtilis, Thalassionema nitzschioides, Gyrosigma balticum, Bacillaria paxillifera, Cylindrotheca closterium, and Nitzschia lorenziana were the widespread species in the Passos River estuary, Brazil. That result coincided with the present investigation. Moreover, our findings of dominant phytoplankton species in Tanintharyi coastal waters were similar to the results reported by Khin Yu Nwe 20116, Yin Yin Htay 201411, and 201912. Zar Ni Ko Ko 201413 reported Coscinodiscus, Hemidiscus, Rhizosolenia, Proboscia, Guinardia, Eucampia, Ditylum, Odontella, Thalassionema, Nitzschia, Dinophysis, and Protoperidinium were abundantly found in Elphinstone Island waters, Myeik Archipelago. His finding was in agreement with the present study. In his study, phytoplankton abundance was high in November and low in August. However, in the present study, the seasonal trend indicated the highest abundance in April and the lowest in December at Myeik area.
Zaw Moe Aung 201114 described Coscinodiscus, Odontella, Ditylum, Nitzschia, and Ceratium species were very common in Setse waters. Moreover, Aung Myo Hsan 20132 reported Coscinodiscus marginatus, C. radiatus, Minidiscus trioculatus, Cyclotella striata, Skeletonema costatum, Leptocylindrus danicus, Syringidium americanum, Thalassionema nitzschioides, and T. frauenfeldii were frequently observed in Thanlwin River mouth to Setse at Mon coastal waters and high phytoplankton abundance was recorded in the pre-monsoon months. The present result agrees well with their statements. Besides, Thida Nyunt 20139 reported phytoplankton abundance was high from November to May (post-monsoon to pre-monsoon) in Mon coastal waters. Her observation was much similar to the present study.
Phytoplankton communities at the five stations along Tanintharyi coastline, viz., Kawthaung, Myeik, Kampani, Ye, and Setse showed noticeable variations in cell abundance. The maximum cell abundance of phytoplankton was found in pre-monsoon months at all stations which were noted that a mesotrophic environment favor species richness and density.
CONCLUSIONS
Phytoplankton community in Tanintharyi coastal waters showed noticeable variations in species occurrence and abundance, space, and time. The present study observed the highest phytoplankton abundance in the pre-monsoon season at all five stations. An extensive variation in phytoplankton abundance was detected in the monsoon through earlier months of post-monsoon periods due to significant variations in environmental parameters that were strongly affected by monsoon season in the Indian Ocean. Phytoplankton abundance at Kawthaung and Myeik recorded as maximum among five stations because of more diverse kinds and frequently occurring phytoplankton species at these water columns. The existence of mangrove forests and the creation of tide shelter by a chain of numerous islands also support high species diversity of phytoplankton at Kawthaung, Myeik, and Kampani stations compare with Ye and Setse stations where less diverse phytoplankton species were found.
Acknowledgments
The authors deeply indebted to Dr. Aung Myat Kyaw Sein, Rector of Mawlamyine University, and Dr. San San Aye, Pro-Rector of Mawlamyine University, for their permission to undertake this research. We wish to thank Dr. Khin Maung Cho, Pro-Rector (Retd.), Mawlamyine University, for his kind suggestions in preparing the manuscript. Special thanks are to the Department of Marine Science, Mawlamyine University for providing lab facilities.
Funding
None.
Conflicts of interest
The authors declare that there is no conflict of interest.
REFERENCES
- Aquino, E.P., Figueiredo, L.G.P., Borges, G.C.P., Ferreira, L.C., Passavante, J.Z.D.O., Gloria, M.D. and Silva-Cunha, G.D. Seasonal and spatial variation in phytoplankton community structure of an estuary in Northeastern Brazil. Tropical Ecology. 2015. 56(1): 125-131.
- Aung Myo Hsan. A study on the occurrence and abundance of phytoplankton along the Thanlwin river mouth. MRes Thesis. Department of Marine Science, Mawlamyine University. 2013. 166 pp.
- Dussaram, K. Variation in the phytoplankton abundance, biomass and diversity of first-order streams having different upstream habitats. Department of Limnology, Institute of Ecology and Evolution, Uppsala University. 2007.46 pp.
- Hutchinson, G.E. The paradox of the plankton. The American Naturalist. 1961. 95(882):137-145.
5. Kamba, M. and Yuki, K. Plankton of Burmese coasts. Institute of Oceanic Research and Development. Tokai University. 1980. 2: 89-142.
- Khin Yu Nwe. Study on the species identification, composition, distribution and abundance of phytoplankton from Myeik adjacent waters. Unpublished MRes Thesis. Department of Marine Science, Myeik University. 2011. 108 pp.
7. LeGresley, M. and McDermott, G. Counting chamber methods for quantitative phytoplankton analysis: hemocytometer, Palmer-Maloney cell and Sedgewick-Rafter cell. In: Karlson, B., Cusack, C. and Bresnan, E. (Eds.) Intergovernmental Oceanographic Commission of ©UNESCO. Microscopic and molecular methods for quantitative phytoplankton analysis. IOC Manuals and Guides no.55. 2010. 25-30.
- Taylor, F.J.R. The phytoplankton of water adjacent to a tropical Asian mangrove area. A report to UNESCO. 1975. 32 pp.
- Thida Nyunt. Phytoplankton communities in Mon coastal waters. PhD Thesis. Department of Marine Science, Mawlamyine University. 2013. 282 pp.
- Tilman, D. Resource competition and community structure. Monographs in Population Biology. Princeton University Press, Princeton, New Jersey. 1982. 17: 1-296.
- Yin Yin Htay. Ecology of phytoplankton communities in Myeik coastal waters. PhD Thesis, Department of Marine Science, Mawlamyine University. 2014. 501 pp.
- Yin Yin Htay, Tin Tin Kyu and Moe Lwin Lwin. Species composition and distribution of some phytoplankton in Myeik Archipelago, southern Myanmar. 2019. Journal of Aquaculture & Marine Biology. 8(5): 163-169.
13. Zar Ni Ko Ko. Study on the phytoplankton common in the Elphinstone Island waters area, Myeik Archipelago. MRes Thesis. Department of Marine Science, Myeik University. 2014. 121 pp.
- Zaw Moe Aung. Studies on the primary productivity of marine phytoplankton in Setse waters. MRes Thesis, Department of Marine Science, Mawlamyine University. 2011. 98 pp.
Received: 24 April 2020
Accepted: 15 June 2020
Khin Khin Gyi1, Wint Thuzar Nwe2, Zin Zin Zaw3 and Khin Khin San4
1: Lecturer, Department of Marine Science, Mawlamyine University, Mon State, Myanmar https://orcid.org/0000-0003-2489-2480
2: Assistant Lecturer, Department of Marine Science, Mawlamyine University, Mon State, Myanmar https://orcid.org/0000-0003-3456-8625
3: Lecturer, Department of Marine Science, Pathein University, Ayeyarwady Division, Myanmar https://orcid.org/0000-0002-6170-5640
4: Lecturer, Department of Marine Science, Sittwe University, Rakhine State, Myanmar https://orcid.org/0000-0002-5316-9892
*Corresponding author:
Khin Khin Gyi
Lecturer
Department of Marine Science, Mawlamyine University
Mawlamyine City, Mon State, Myanmar
Tel: (+95) 9 889 640 400
Email: [email protected]
