2022.07.04.9
Files > Volume 7 > Vol 7 No 4 2022
Evaluating the serum ferritin levels of COVID-19 patients from Basra
Alaa A. Saleh 1,* and Saad Waheed 2
1 Medical Laboratory Department, Technical Institute, Southern Technical University, Basra, Iraq
2 Al-Basra teaching Hospital, Basra, Iraq
* Correspondence: [email protected]
Available from: http://dx.doi.org/10.21931/RB/2022.07.04.9
ABSTRACT
Coronavirus disease 19, "COVID-19, "is occurred by a coronavirus called (SARS CoV-2), which causes severe infection in many infected persons. Early Identifying risk factors for this disease can significantly help manage critical cases and save patients' lives. This study aimed to assess the predictive value of the ferritin, the erythrocyte sedimentation rate ″ESR″, the C-reactive protein ″CRP ″, and white blood cell ″WBC″. Positive cases of COVID-19 were confirmed by "real-time polymerase chain reaction." From the patient's records were obtained demographic data and laboratory investigations were.
According to clinical syndromes, patients were categorized into two groups, including COVID -19 patients with severe and non-severe diseases. Of 305 COVID-19 patients, they have a mean age of 42.73 ± 16.37 years, 59.01% of patients are female, and 40.99% are male. The levels of ferritin were variable in COVID-19 patients, our results revealed that18.68% had increased serum ferritin in patients, and the ESR, as well as CRP, were high in most patients; it's above the normal range. 4.91% of patients had decreased WBC, and the result showed lymphopenia in 1.96%. Neutrophils were above the normal range in 14.75% of patients, and 2.95% of patients had decreased serum platelets, a significant difference in WBC, Lymphocytes, Neutrophils and Basophils between severe and non-severe COVID-19 patients (p<0.05). A positive correlation was observed between the levels of ferritin and the severity of the disease
Keywords: COVID-19, Ferritin, ESR, CRP, WBC
INTRODUCTION
In 2019, a novel coronavirus was spread in Wuhan, China1; subsequently, this disease spread worldwide, and WHO declared coronavirus 2019 (COVID- 19) a global pandemic in March 2020 2. From asymptomatic infection to the development of interstitial pneumonia, the severity of disease and clinical presentation of novel disease vary, and additionally, failure in respiratory and acute respiratory syndromes3. Pro-inflammatory mediators are released, and the immune system is activated in this disease, which has a complex pathophysiology4. The cytokine storm considers the leading cause of COVID-19 mortality.
Ferritin is a representative of body iron stores, and high levels of it are related to COVID-19. Previous studies observed that increased levels of it were linked to a cytokine storm in severe COVID-19 patients, and an increase in ferritin was associated with the inter and spread of the viral in the human body as well as affected iron metabolism5,6
Many studies confirmed significant changes in several laboratory parameters involving WBC (Lymphocytes, Neutrophils, monocyte, Basophils), ferritin, CRP, and platelets in COVID-19 cases 7-10.
The current study evaluates the correlation of ferritin with many laboratory parameters and the severity of the disease.
MATERIALS AND METHODS
A retrospective study was conducted at Basra Teaching Hospital, which treats COVID-19 patients. Between 1 and 30 April 2020, this center confirmed the cases by nasopharyngeal/throat swab specimens, and RT-PCR was used to diagnose 305 cases, ranging from18 -85 years.
A manual perusal of patients' case sheets was used to review patient medical records. A data collection checklist was used to collect data from electronic medical records on epidemiological, clinical, laboratory, and outcome measures. In addition, patient information, such as past medical history, symptoms, and indicators, as well as laboratory tests, such as CRP, ESR, serum ferritin, WBCs, and platelet count, were collected.
SPSS version 16 (IBM, Chicago, USA) was used to calculate the data. The median and percent were used to represent data and compare variables, and These statistical tests were conducted using″ Mann–Whitney U-tests″ and″ Student's t-tests″.
RESULTS
Three hundred five patients were examined; the mean age of patients was 42.73 ± 16.37 years,182 (59.01%) were females, and 123 (40.99%) were males. 22 (7.23%) patients had severe syndrome, and 281 (92.13%) had non-severe.
Serial ferritin data shows a significant difference between both sexes (p<0.05). In most of the variables in table 1, no significant difference was found between male and female participants
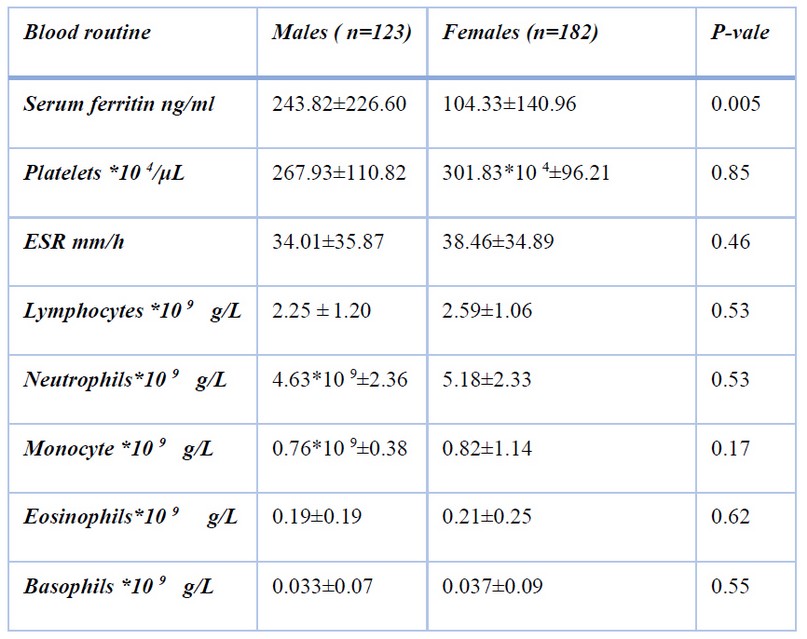
Table 1. Laboratory data between Males and Females COVID-19 patients
The levels of ferritin were variable in COVID-19 patients; our results revealed that 23.6% of patients had decreased serum ferritin and 18.68% had increased serum ferritin, and the levels of ESR and CRP were high than the normal range in most patients.
The blood counts showed 15 (4.91%) patients had decreased white blood cells (< 4*109 g/L), and results were observed lymphopenia in 6 patients (1.96%), neutrophils were high than the normal range in 45 (14.75%) patients as in Table 2
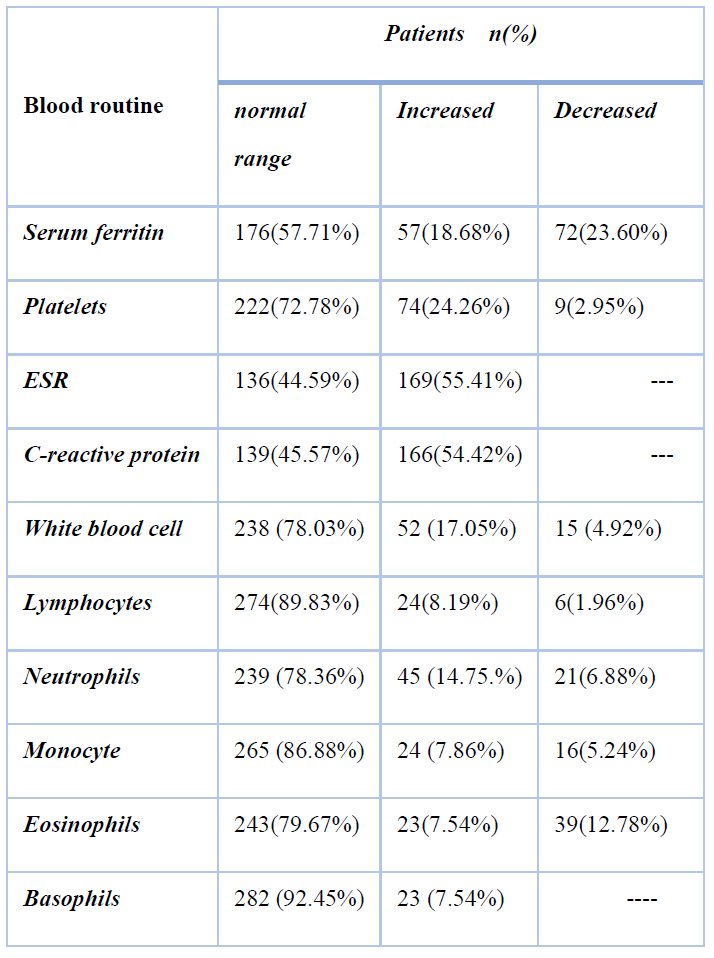
Table 2. Laboratory findings of COVID-19 patients
The results revealed that significant difference in WBC, Lymphocytes, Neutrophils, and Basophils between the disease severe and non-severe (p<0.05), but no significant difference between the two groups in monocyte (p > 0.05) in table 3
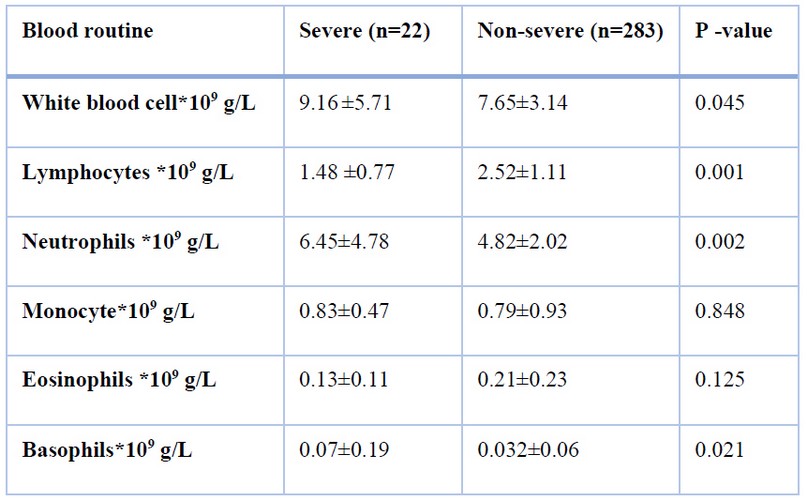
Table 3. Laboratory data between patients with severe and non-severe disease
There was a positive relation R= 0.12 between Ferritin levels and Neutrophils and a negative relation R=-0.3, R=-0.05 between Ferritin levels and Lymphocytes, monocytes respectively, as in figure 1.
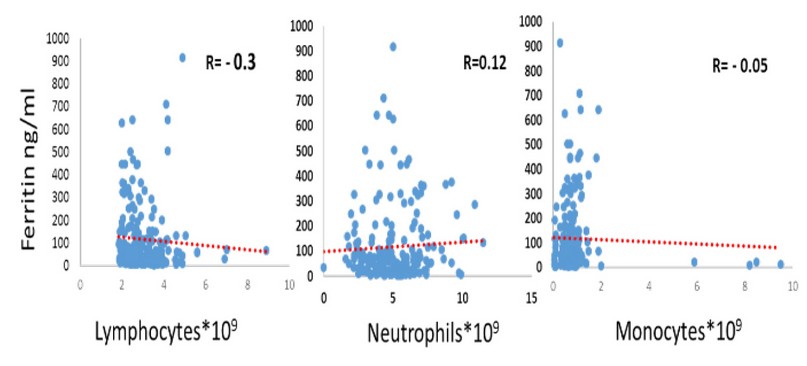
Figure 1. Correlation between ferritin and (Neutrophils, Lymphocytes, and monocyte) levels
A positive relationship was observed between Ferritin levels and the severity of the disease, R= 0.385, as in figure 2
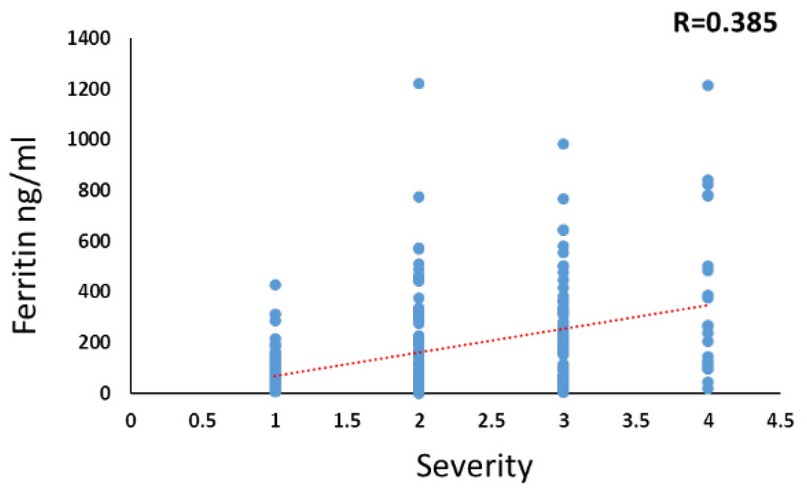
Figure 2. The positive relationship between Ferritin levels and the severity of disease
DISCUSSION
Our results appear that female was more susceptible than male; both sexes are affected by infectious diseases differently. Among the differences are socioeconomic status, gender inequities, occupational exposure and a sex gap in immune responses to sex hormones. According to a study from China, males and females are at risk of contracting the disease11,12.
In this study, ferritin level was higher in males than females, maybe due to the presence of Hepcidin; Hepcidin regulates ferritin levels and is related to sex hormones involving estrogens and testosterone13. As well as high serum ferritin levels as is associated with some diseases, including insulin resistance, cardiovascular disease, and COVID-1914-16
In the current study, the counts of WBC were within the normal range at 78.03% of patients, increased at 17.05% of patients, and decreased at 4.92% of patients; leukopenia has been changed according to the severity of the disease17. The count of WBC was less elevated among COVID-19 patients who needed intensive care unit admission or died compared with other patients18. Early studies did not observe changes in eosinophils because of a relatively small number among WBCs19; however, our results appear that the counts of eosinophils in severe COVID-19 patients were low but not significant compared to patients in another group. This indicates the critical role of eosinophils in COVID-19 infection.
Non-severe COVID-19 infection was observed to be related to decreased counts of neutrophils, monocytes, and basophils compared to severe disease; inflammatory cell depletion occurred in patients during the recovery phase but may worsen as the disease progress. Previous studies showed the decreased count of peripheral inflammatory cells throughout the movement of it from peripheral blood towards the lungs; these cells have a role in tissue repair20 and immune defense against infection of virus21.On the other hand, many studies reported that the differences in the count either increased or decreased in monocyte22 and neutrophil23 in COVID-19; levels of neutrophil were increased on the first days of infection; in contrast, the levels were slightly reduced after the treatment24, other researchers indicated that the count of neutrophils, could help predict severe cases of COVID-1925.
In our study, the ferritin level was highly significant in COVID-19 patients with severe disease, and the relationship between levels of ferritin and disease severity was positive. So, the current study supports the correlation between ferritin levels and severe COVID‐19. Ferritin is an iron storage protein and an inflammatory biomarker in acute and chronic inflammation. Many factors can cause an increase in the level of ferritins, such as some cytokines that stimulate ferritin synthesis and cellular damage in inflammation that leads to leakage of intracellular ferritin26. In this study, ferritin levels were 365 ng/mL in severe COVID-19 patients, which was above the normal ferritin range. Another study found that ferritin levels in infection with severe disease of COVID-19 were generally above the normal range16.
CONCLUSION
Ferritin is correlated with the severity of the disease. Therefore, ferritin can be used for the assessment of the state of disease and. treatment of COVID-19 patients.
REFERENCES
1. Zhu N.; Zhang D.; Wang W.; Li X.; Yang B.; Song J.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med, 2020; 382:727–33.
2. Zhou P.; Yang XL.; Wang XG, Hu B.; Zhang L.; Zhang W.; et al. Discovery of a novel coronavirus associated with the recent pneumonia outbreak in humans and its potential bat origin. bioRxiv , 2020.https://doi.org/10.1101/2020.01.22.914952.
3. Zhang YZ.; Holmes EC.; A genomic perspective on the origin and emergence of SARS-CoV-2. Cell ,2020;181(2):223–7.
4. Chan JF.; Zhang AJ.; Yuan S.; et al. Simulation of the clinical and pathological manifestations of Coronavirus Disease 2019 (COVID-19) in golden Syrian hamster model: implications for disease pathogenesis and transmissibility. Clin Infect Dis, 2020;71(9):2428-46. https://doi.org/ 10.1093/cid/ciaa325
5. Kernan KF., Carcillo JA.; Hyperferritinemia and inflammation. Int Immunol, 2017, 29,pp.401-409. 10.1093/intimm/dxx031
6. Wessling-Resnick M: Crossing the iron gate: why and how transferrin receptors mediate viral entry. Annu Rev Nutr. 2018, 38:431-458. 10.1146/annurev-nutr-082117-051749
7. Rodriguez-Morales AJ.; Cardona-Ospina JA.; Gutiérrez-Ocampo E.; Villamizar-Peña R.; Holguin-Rivera Y.; Escalera-Antezana JP.; et al. Clinical, laboratory and imaging features of COVID-19: a systematic review and meta-analysis. Travel Med Infect Dis, 2020; 34:101623.
8. Huang Y.; Liu A.; Liang L.; Jiang J.; Luo H.; Deng W.; et al. Diagnostic value of blood parameters for community-acquired pneumonia. Int Immunopharmacol.2018; 64:10–5.
9. Abdalbakee, H. N. & Mohammed, Th. T. Effect of using different levels of azolla as a substitute for soybean meal in the production performance of fish carp. Plant Archives. 2019, 19(1): 573-577. Doi: 10.13140/RG.2.2.23568.15367
10. Pantzaris N-D.; Platanaki C.; Pierrako C.; Karamouzos V.; Velissaris D.; Neutrophil- to-lymphocyte ratio relation to sepsis severity scores and inflammatorybiomarkers in patients with community-acquired pneumonia: a case series. J Transl Intern Med. 2018; 6(1):43–6
11. Li Q.; Guan X.; Wu .; Wang X.; Zhou L.; Tong Y.; et al. Early Transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020, 382:1199–207. doi: 10.1056/NEJMoa2001316
12. Jin, J.-M.; Bai, P., He, W., Wu, F., Liu, X.-F., Han, D.-M., et al. Gender Differences in Patients With Covid-19: Focus on Severity and Mortality. Front. Public Health ,2020,8, 152. doi: 10.3389/fpubh.2020.00152
13. Guo W.; Bachman E.; Li M.; Roy CN.; Blusztajn J.; Wong S.; et al. Testosterone administration inhibits hepcidin transcription and is associated with increased iron incorporation into red blood cells. Aging Cell ,2013,12, pp. 280–291. doi: 10.1111/acel.12052
14. Li J.; Cao Y.; Serum ferritin as a biomarker for diabetes and insulin resistance: a further study. Acta Diabetol.,2015, 52;pp. 417–419. doi: 10.1007/s00592-015-0719
15. Cho YS.; Kang JH.; Kim SA.; Shim KY.; Lee HS. ; Association of serum ferritin and abdominal obesity and insulin resistance. J Korean Soc Study Obes.,2005, 14, pp. 76–81
16. G'omez-Pastora J.; M. Weigand M.; Kim J.; et al., Hyperferritinemia in critically ill COVID-19 patients - is ferritin the product of inflammation or a pathogenic mediator? Clin. Chim. Acta ,2020, PP.249–251, https://doi.org/10.1172/
17. Li X.; Xu S.; Yu M.; et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J Allergy Clin Immunol 2020;146(1):110-8. https://doi.org/10.1016/ j. jaci.2020.04.006
18. Wang D.; Hu B.; Hu C.; Zhu F.; Liu X.; Zhang J.; et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA, 2020; 323, PP. 1061–1069.
19. Bingdi Yan.; Junling Yang.; Yan Xie.; Xiaolei Tang.; Relationship between blood eosinophil levels and COVID-19 mortality. World Allergy Organization Journal, 2021,14,3,
https://doi.org/10.1016/j.waojou.2021.100521
https://doi.org/10.1016/j.waojou.2021.100521
20. Furuta G.T.; F.D. Atkins F.D.; N.A. Lee N.A.; J.J. Lee J.J. Changing roles of eosinophils in health and disease. Ann Allergy Asthma Immunol, 2014,113, pp. 3-8
21. Mogensen SC.; Role of macrophages in natural resistance to virus infections. Microbiol Rev., 1979;43(1):1-26. doi:10.1128/mr.43.1.1-26.1979.
22. Curtolo A.; Oliva A.; Volpicelli L.; Ceccarelli G.; D'Ettorre G.; Borrazzo C.; Mastroianni CM, Venditti M. Monocyte absolute count as a preliminary tool to distinguish between SARS-CoV-2 and influenza A/B infections in patients requiring hospitalization. Infez Med. 2020, 1;28(4), PP.534-538. PMID: 33257627.
23. Hazeldine J.; Lord JM.; Neutrophils and COVID-19: Active Participants and Rational Therapeutic Targets. Front Immunol. 2021, 2; 12:680134. doi: 10.3389/fimmu.2021.680134. PMID: 34149717; PMCID: PMC8206563.
24. Thunström Salzer, A., Niemiec, M. J.; Hosseinzadeh, A.; Stylianou, M.; Åström, F., Röhm, M., Ahlm, C., Wahlin, A., Ermert, D., & Urban, C. F. Assessment of Neutrophil Chemotaxis Upon G-CSF Treatment of Healthy Stem Cell Donors and in Allogeneic Transplant Recipients. Frontiers in immunology. 2018, 9, 1968. https://doi.org/10.3389/fimmu.2018.01968
25. Reusch N.; Domenico E.; Lorenzo Bonaguro L.; Jonas Schulte-Schrepping J.; Baßler K., Schultze J. L., and Aschenbrenn A.C., Neutrophils in COVID-19, Front. Immunol., 2021 | y
26. Kernan, K. F.; & Carcillo, J. A., Hyperferritinemia and inflammation. International immunology, 2017,29(9),401–409. https://doi.org/10.1093/intimm/dxx0
Received: 20 July 2022 / Accepted: 15 October 2022 / Published:15 November 2022
Citation: Saleh A A, Waheed S. Evaluating the serum ferritin levels of COVID-19 patients from Basra. Revis Bionatura 2022;7(4) 9. http://dx.doi.org/10.21931/RB/2022.07.04.9
