2022.07.02.56
Files > Volume 7 > Vol 7 No 2 2022
1, Internal Medicine Specialist, Hospital General Ibarra.Ecuador
2, Internal Medicine Specialist, Hospital Básico Antonio Ante. Ecuador
Corresponding author. [email protected]
Available from: http://dx.doi.org/10.21931/RB/2022.07.02.56
If we remember very superficially a little about the complex human immune system, a part of this immune system is made up of leukocytes (especially phagocytes and lymphocytes) that are responsible for detecting invaders and sending them defensive markers that adhere to them (the antibodies) and also to destroy the invaders identified by the system; In addition to the already mentioned leukocytes, our immune system is made up of various other elements such as cells, proteins, tissues and organs, which defend us against germs and microorganisms. 1, 2
Speaking specifically about the immune system and COVID-19, the events mainly involved in the immunopathogenesis of COVID-19 and the dysregulation of the immune response include lymphopenia and increased neutrophil-to-lymphocyte ratio, cytokines, lymphocyte depletion and dysfunction, antibody-dependent enhancement, and monocyte and granulocyte abnormalities. It must be emphasized that lymphopenia is an essential finding in most patients with COVID-19, particularly in those with a severe phenotype.3 It has been shown that multiple viral proteins induce T cells after infection and that T cells that have antiviral signatures related to safety and protection can be achieved by vaccination.4
There are many questions about immunity-based protection still unresolved due to the wide clinical diversity of symptoms, the association between delayed/weak antibody responses and severe outcomes, and the coexistence between coronavirus and its specific IgG; SARS-CoV-2 infection can either weaken or overactivated the immune system, and patients affected by severe forms of COVID-19 can be distinguished by different "immunotypes" associated with poor clinical pathways.5
Despite the significant advances that have been evidenced in terms of knowledge about the virus-host interaction in this pandemic, it has been mentioned that the information on the immunopathology of COVID-19 is still limited, and the current evidence may change soon with the accumulation of new knowledge about the biology of SARS-CoV-2 and host immune responses, so the current hypotheses could be incomplete and may even prove wrong in the near future.6
Every day our understanding of the human body's response to SARS-CoV-2 evolves; however, during this same time, the virus mutates, which leads us to new challenges for our intervention strategies for the patient. Rational drug reuse has primarily focused on drugs with known activity against the cytokine storm (excessive immune response) associated with immune interventions.7, 8
The course of SARS-CoV-2 infection is individual and is influenced by the particularities of immune reactivity. The eradication of the infectious process requires the complete activation of the immune response, innate and adaptive, humoral and cellular, and the synthesis of IFN I, the primary and essential form of antiviral surveillance. The complex interplay between the severity of COVID-19 and the dysregulated host immune response has allowed worldwide evaluation of different immunomodulators currently being investigated for therapeutic effects in COVID-19 patients.9
IgM and IgG seroconversion rate and timing of SARS-CoV-2, SARS-CoV, and MERS-CoV have been comparatively studied. With inconsistent results across studies, in SARS-CoV-2, the majority seroconverted to IgG within 2 weeks and 100% of patients within 4 weeks; IgG was still detectable at the end of follow-up (49-65 days) in all patients; neutralizing antibodies were detected in 92%-100% of patients (up to 53 days), without clarifying whether reinfection with SARS-CoV-2 is possible, with more suggestive studies of intermittent detection of residual RNA. In SARS-CoV, specific IgG was maintained for 1–2 years after infection and then decreased; although one study detected IgG up to 12 years after infection, neutralizing antibodies were detected up to 17 years in another study. Three studies on MERS-CoV reported that IgG could be detected for up to 2 years.10
It is worth mentioning that the proposed course of SARS-CoV-2 infection could start in upper respiratory tract infection (mainly as respiratory droplets), continue in lower respiratory tract infection (mainly as pulmonary infection) , followed by viremia and diffusion to other target organs (including the gastrointestinal tract), the latter being very important as it is associated with the theory that the spread of SARS-CoV-2 in hospitals and nursing homes could occur through fecal contamination, involving health personnel in the infection, who are usually able to protect themselves from droplets, wear masks, but less easily during direct care and hygiene of patients, where people are generally bedridden and not autonomous in the handling of their stools, so it should be reiterated in the proper use of personal protection equipment.11
We wanted to bring this topic up after having treated a 65-year-old female patient, who was referred for consultation to be authorized to be vaccinated against COVID-19 because both her family doctors and several other people had reported her that because she had low defenses, she should not be vaccinated. If a patient has "low defenses," is it not logical to receive medicines that can improve her defenses as part of her treatment? It would be the most rational from our point of view. But what happens when this drug is a vaccine?
Reviewing the medical file, the patient had a documented history of almost 20 years with leukopenia, which, due to its mild nature, had not been given much importance; however, in the current scenario of the COVID-19 pandemic, for the patient, it became a very relevant situation from one moment to another since the "low defenses" did not allow her to be vaccinated creating personal conflicts by not being able to go to public or private places where the vaccination card is required and with her family since they were avoiding her for being the only one not to be vaccinated, causing all this feeling of guilt, anxiety and depression.
Decisions about the best method to achieve control of the pandemic should not be left only to doctors and medical authorities. However, they should be shared between vaccine experts, regulators, financial and political leaders, and public representatives to establish the best acceptable safety margins and efficacy thresholds for vaccines, which can increase confidence in the decision process and its results.12
The condition of this patient does not constitute a contraindication for vaccination; moreover, vaccination is currently encouraged in patients with immune compromise since immunosuppressed people are especially vulnerable to COVID-19; however, there is widespread misinformation about the adverse effects and contraindications of the different vaccines, including in health personnel, which often leads us to not offer adequate advice to our patients regarding vaccination.13
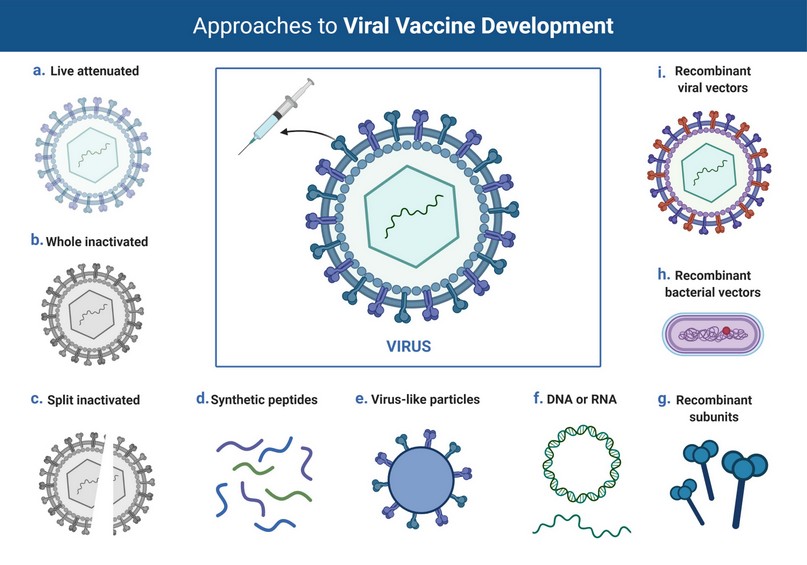
Figure 1. Overview of different platforms for viral vaccine development. Various strategies include live attenuated, whole inactivated, split inactivated, synthetic peptides, and recombinant approaches (Biorender). 15,16
CONCLUSION
There is a large sector of the world population that, despite all the scientific information available, persists with uncertainty and/or disbelief about the importance of vaccines against COVID-19; however, this is not the first time that vaccines have been rapidly manufactured and tested to deal with a public health crisis and even though accelerated Phase I-II-III clinical programs have been applied, they have followed the traditional development route to ensure the efficacy and safety of vaccines.14 We hope, for her good, that this patient will decide to get vaccinated. The enemy is the virus, not the vaccine
REFERENCES
1. The history of vaccines [Internet]. The College of Physicians of Philadelphia; 2022 [cited 15 jan 2022]. The Human Immune System and Infectious Disease; [aprox. 5 p.] Available at: https://www.historyofvaccines.org/index.php/content/articles/human-immune-system-and-infectious-disease
2. Palomo I, Pereira J, Koenig C. Células y órganos del sistema inmune. En: Palomo I, Ferreira A, Sepúlveda C, et al. Fundamentos de inmunología básica y clínica. Talca: EDitorial Universidad de Talca; 2002. p. 53-84. Available at: http://editorial.utalca.cl/docs/ebook/inmunologia.pdf
3. Lotfi R, Kalmarzi RN, Roghani SA. A review on the immune responses against novel emerging coronavirus (SARS-CoV-2). Immunol Res. 2021; 69(3), 213–24. DOI: https://doi.org/10.1007/s12026-021-09198-0
4. DiPiazza AT, Graham BS, Ruckwardt TJ. T cell immunity to SARS-CoV-2 following natural infection and vaccination. Biochemical and Biophysical Research Communications. 2021; 538, 211-7. DOI: https://doi.org/10.1016/j.bbrc.2020.10.060.
5. Napoli C, Benincasa G, Criscuolo C, Faenza M, Liberato C, Rusciano M. Immune reactivity during COVID-19: Implications for treatment.
Immunology Letters. 2021; 231: 28-34. DOI: https://doi.org/10.1016/j.imlet.2021.01.001.
6. Boechat JL, Chora I, Morais A, Delgado L. The immune response to SARS-CoV-2 and COVID-19 immunopathology - Current perspectives. Pulmonology 2021; 27: 423-37. DOI: https://doi.org/10.1016/j.pulmoe.2021.03.008
7. Talmadge JE, Marceau F. Covid-19 challenges to immune investigations and therapies. International Immunopharmacology. 2021; 95: 107543. DOI: https://doi.org/10.1016/j.intimp.2021.107543.
8. Chen Z, Feng Q, Zhang T, Wang X. Identification of COVID-19 subtypes based on immunogenomic profiling. International Immunopharmacology. 2021; 96:107615. DOI: https://doi.org/10.1016/j.intimp.2021.107615.
9. Mihaescu G, Chifiriuc MC, Vrancianu CO, Constantin M, Filip R, Popescu MR, et al. Antiviral immunity in SARS-CoV-2 infection: From protective to deleterious responses. Microorganisms [Internet]. 2021; 9(12): 2578. DOI: http://dx.doi.org/10.3390/microorganisms9122578
10, O Murchu E, Byrne P, Walsh KA, Carty PG, Connolly M, De Gascun C, et al. Immune response following infection with SARS-CoV-2 and other coronaviruses: A rapid review. Rev Med Virol.2021;31:e2162. DOI: https://doi.org/10.1002/rmv.2162
11. Signorini L, Dolci M, Castelnuovo N, Crespi L, Incorvaia B, Bagnoli P, et al. Longitudinal, virological, and serological assessment of hospitalized COVID-19 patients. J. Neurovirol. 2022. DO: Ihttps://doi.org/10.1007/s13365-021-01029-0
12. Tau N, Yahav D, Shepshelovich D. Vaccine safety – is the SARS-CoV-2 vaccine any different?. Human Vaccines & Immunotherapeutics. 2021: 17(5); 1322-5. DOI: 10.1080/21645515.2020.1829414
13. COVID-19 [Internet]. Centers for Disease Control and Prevention; 2022 [cited 15 jan 2022]. COVID-19 Vaccines for moderately or severely immunocompromised people; [aprox. 4 p.] Available at: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/recommendations/immuno.html
14. Patterson S, Fu B, Meng Y, Bailleux F, Chen J. Statistical observations on vaccine clinical development for pandemic diseases. Statistics in Biopharmaceutical Research. 2021. DOI: 10.1080/19466315.2021.1919197
15. Afrough B, Dowall S, Hewson R. Emerging viruses and current strategies for vaccine intervention. Clin Exp Immunol. 2019;196(2):157-166. doi:10.1111/cei.13295
16. Graham BS. Advances in antiviral vaccine development. Immunol Rev. 2013;255(1):230-242. doi:10.1111/imr.12098
Received: 24 January 2022 / Accepted: 21 February 2022 / Published:15 May 2022
Citation.Valarezo-Sevilla D, Sarzosa-Terán V. The enemy is the virus, not the vaccine.
Revis Bionatura 2022;7(2) 56. http://dx.doi.org/10.21931/RB/2022.07.02.56
