Investigation / Researchs
Files > Volume 1 > Vol 1 No 2 2016 > Investigaciones
Genome reprogramming in cells that escape from senescence
Reprogramación del genoma en células que escapan de la senescencia
Available from: http://dx.doi.org/10.21931/RB/2016.01.02.3Xavier Deschênes-Simard 1, Stéphane Roy 2 and Gerardo Ferbeyre 3
Abstract
Many tumorigenic stimuli trigger a protective cellular response known as cellular senescence, a stable cell cycle arrest characterized by an active metabolism, secretion of immunomodulatory factors and activation of tumor suppressor pathways. The senescence state depends on the continuous and aberrant activation of signalling pathways. Senescent cells will remain non-dividing as long as this aberrant signalling is maintained. However, if these signals are attenuated, senescent cells can escape form their dormant condition and dangerously progress into tumor formation. Here we compare the transcriptome of oncogenic Ras–induced senescent cells with that of cells that escaped from senescence after attenuation of the ERK/MAP kinase signaling pathway. We found that cells that escaped from senescence express genes associated to genetic instability and display a highly aberrant karyotype. These cells also express genes commonly altered in the cancer transcriptome of different tissues supporting the idea of a common gene expression program that governs cancer cells. Bioinformatic analyses identified the transcription factors most likely regulating the gene expression programs of senescent cells and cells that escape from senescence. We propose a model of carcinogenesis where cells that escape from senescence provide a heterogeneous population of genetically unstable cells that can progress into malignant tumors.
Keywords: Senescent, tumor suppressor, immunity
Resumen
Muchos estímulos tumorigénicos desencadenan una respuesta celular protectora conocida como senescencia celular, la cual es una detención estable del ciclo celular caracterizada por un metabolismo activo, la secreción de factores inmunomoduladores y la activación de vías de supresión de tumores. El estado de senescencia depende de la activación continua y aberrante de las rutas de señalización. Las células senescentes permanecerán sin dividirse el tiempo que se mantiene esta señalización aberrante. Sin embargo, si se atenúan estas señales, las células senescentes pueden escapar de su condición inactiva y peligrosamente progresar a la formación de tumores. Aquí comparamos el transcriptoma de las células senescentes oncogénicas inducidas por Ras, con el de las células que escaparon de la senescencia después de la atenuación de la vía de señalización de la quinasa / ERK MAP. Encontramos que las células que escaparon de la senescencia expresan los genes asociados a la inestabilidad genética y muestran un cariotipo altamente aberrante. Estas células también expresan genes comúnmente alterados en el transcriptoma de diferentes tejidos cancerígenos que apoyan la idea de un programa de expresión génica común que regula las células cancerosas. Los análisis bioinformáticos identificaron los factores de transcripción más probable que regulan los programas de expresión génica de células senescentes y células que se escapan de la senescencia. Nosotros proponemos un modelo de carcinogénesis donde las células que se escapan de la senescencia proporcionan una población heterogénea de células genéticamente inestables que pueden progresar en tumores malignos.
Palabras clave: senescencia, supresor de tumores, inmunidad.
Introduction
Cellular senescence is a tumor suppressor mechanism with the potential to permanently halt tumor progression or recruit immunity effectors that eliminate cells with the prospective to form malignant tumors.1, 2 Senescent cells are present in premalignant lesions of the prostate 3,4 and the pancreas 5,6 that usually progress to malignant tumors. They are also abundant in benign lesions that rarely become cancerous such as nevi 7,8,9 and benign prostatic hyperplasia. 10,12 The events that allow escaping from senescence in some lesions but not in others remain to be established. Since senescence is identified using a limited set of biomarkers it is plausible that senescent cells that can progress into cancers and those that remain permanently arrested are two distinctive cellular states. High levels of ERK kinases activity can permanently impair cell proliferation and they may define irreversible senescence. 11
Mammary epithelial cells can spontaneously escape from senescence 13 and this has been linked to a downregulation of the ERK pathway. 11 In a model of Ras-induced senescence in human fibroblasts we previously reported that downregulation of ERK allows to bypass senescence and in combination with expression of telomerase leads to malignant transformation. 11 Several studies have concluded that Ras-signaling through the ERK kinases is important for transformation and tumor progression. 14 However, in multiple tissues the most malignant tumors have very low levels of activated ERK, thereby indicating a more complex relationship between these kinases and cancer. 14 To get insights into the process of malignant transformation in cells that escape from senescence we report here a transcriptome analysis of cancer cells obtained after bypassing senescence by reducing ERK activity in comparison with its premalignant senescent precursors that have very high levels of activated ERK.
Materials and methods
Cells, experimental cell biology, protein analysis and cytogenetics.
Normal human diploid fibroblasts IMR90 were obtained from American Type Culture Collection (ATCC, Manassas, VA). Cells were cultured in Dulbecco’s modified Eagle medium (DMEM; Wisent, Montréal, QC) supplemented with 10% fetal bovine serum (FBS; Wisent) and 1% penicillin G/streptomycin sulfate (Wisent). Retroviral gene transfer, senescence biomarkers, immunoblotting and RNA purification were done as described previously. 11 Karyotypic analysis were performed by the cytogenetics platform of the Quebec Leukemia Cell Bank (http://bclq.org/).
Microarray and bioinformatic analysis
Microarray analysis was performed on total RNA purified from three independent samples of senescent cells or cells that escaped from senescence due to the expression of shERK2. We used the service of Genome Québec Innovation Centre. Microarray data files from triplicates of both conditions were analysed with the FlexArray 1.6.3 software (http://genomequebec.mcgill.ca/FlexArray) 15 using Affymetrix default analysis settings. Raw data was deposited in NCBI’s Gene Expression Omnibus (GEO) and are accessible through the accession number GSE33613. Biological functions were analysed using the platform DAVID (https://david.ncifcrf.gov/) 16 or Gene Set Enrichment Analysis (GSEA) software. 17 GSEA computes a Normalized Enrichment Score (NES) that reflects the degree to which a test gene set is overrepresented in upregulated or downregulated genes of another gene set. The significance of NES is mainly affected by the False Discovery Rate (FDR; q-value). 17 We considered significant gene sets with a p-value ≤ 0.05 and a q-value ≤ 0.25. To predict transcription factors regulating a particular set of genes we used the Distant Regulatory Elements of Co-regulated genes (DIRE) web-based application. 18
Results
Global analysis of transcriptome changes in cells that escape from Ras-induced senescence
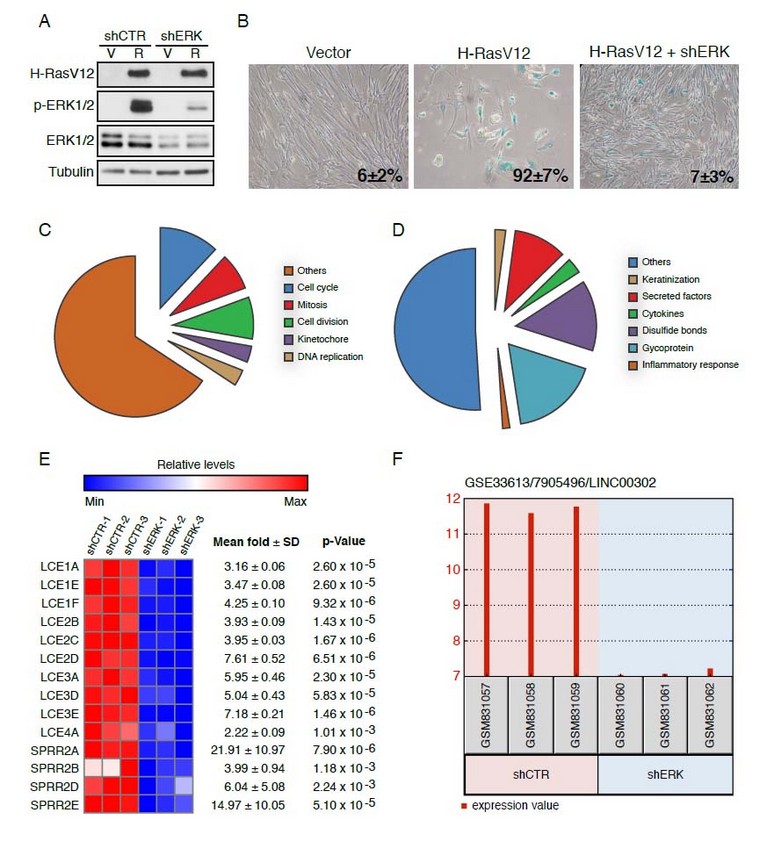
To compare the senescent cells transcriptome with cells that escape from senescence becoming malignant we first expressed the human telomerase gene hTERT in normal diploid human fibroblasts IMR90. Then, we introduced oncogenic Ras in these cells and either a shRNA control or a shRNA against ERK2 (Figure 1A). Ras induced a strong senescence phenotype in cells with shRNA control but cells where ERK2 was downregulated massively escaped from senescence and displayed a transformed morphology (Figure 1B). We then prepared total RNA from these two cell populations and obtained their transcriptome profile by microarray analysis using the service of Genome Québec (http://gqinnovationcenter.com/index.aspx?l=e). A total of 1048 transcripts with a fold change higher or equal to 2 and a p < 0.05 according to a two-sample Student’s t-test were used for a bioinformatics analysis with the DAVID platform (https://david.ncifcrf.gov/) (16). As expected, cells that escaped from senescence express high levels of genes annotated for functions such as the cell cycle (p=5.0E-60), chromosome organization (p=9.0E-38), DNA replication (p=5.5E-27), mitosis (p=1.4E-54), and DNA metabolism (p=4.3E-22) (Figure 1C). Since most of these genes are regulated by RB/E2F complexes, their increase in cells that escape from senescence suggest that they can efficiently disable the RB protein, thereby releasing its inhibitory activity over the E2F transcription factors. On the other hand, senescent cells expressed many genes coding for proinflammatory (p= 3.6E-7) and secreted factors (p=6.1E-13) (Figure 1D), which is a characteristic that has been well documented in many other studies of the transcriptome of senescent cells. 19
Unexpectedly, senescent cells expressed a series of keratinization genes clustered in chromosome 1q21 (Figure 1D). They include multiple late cornified envelope genes and several small proline-rich proteins (Figure 1E). These proteins form a cornified envelope under the plasma membrane of differentiated keratinocytes and contribute to the barrier function of the skin. 20 Their role in senescence remains to be investigated but the fact that p53 regulates their expression suggest that they may be part of the overall tumor suppression functions of senescence.21 Intriguingly, other genes located at 1q21 were also upregulated in senescent cells and they include CTSS, TUFT1, SMCP, S100A8, IVL, HEPHL1, CRCT1, C1orf46 and the long non-coding RNA LINC00302. The differential expression of these genes between senescent cells and cells that escape senescence can be easily visualized using the GEO2R program available at the GEO site containing our microarray data. Our GEO accession number is GSE33613 and one example for differential gene expression found with GEO2R is shown for LINC00302 in figure 1F.
Figure 1. Changes in global gene expression patterns in human fibroblasts that escape from senescence. (A) Immunoblots for the indicated proteins in IMR90 cells expressing oncogenic Ras (R) or a vector control (V) and either an shRNA control (shCTR) or an shRNA targeting ERK2 (shERK). (B) Senescence-associated b-galactosidase of IMR90 cells expressing the indicated vectors. The percentage and standard deviation of SA-b-Gal positive cells is indicated in each panel. (C and D) Bioinformatics analysis of gene expression data with the platform DAVID. (C) Most significant annotation clusters for genes upregulated in cells that escape from senescence. (D) Most significant annotation clusters for genes downregulated in cells that escape from senescence. (E) Senescent cells express high levels of keratinization genes that cluster at the chromosome locus 1q21. (F) GEO2R allows retrieving microarray data for selected genes. Here the long non-coding RNA LINC00302 is shown as an example.
Gene set enrichment analysis of gene expression changes that accompany senescence escape
A pathway controlling a particular cellular function is regulated by multiple genes. Small changes in many of those genes can have a strong effect on the final output but are ignored by only considering genes with substantial changes in expression. To circumvent this problem, gene sets has been defined for multiple biological categories. DAVID, the bioinformatics program used above, uses gene sets as defined by the Gene Ontology project. This method ignores genes that may play a role not yet discovered in a biological process and is therefore biased. Gene set enrichment analysis (GSEA) compares microarray data to gene sets defined by previous microarrays studies. It thus determines whether members of a gene set are enriched at the top or the bottom of the gene expression data. 17 GSEA considers all genes in the microarray data without arbitrary cut-off based on fold change or significance.
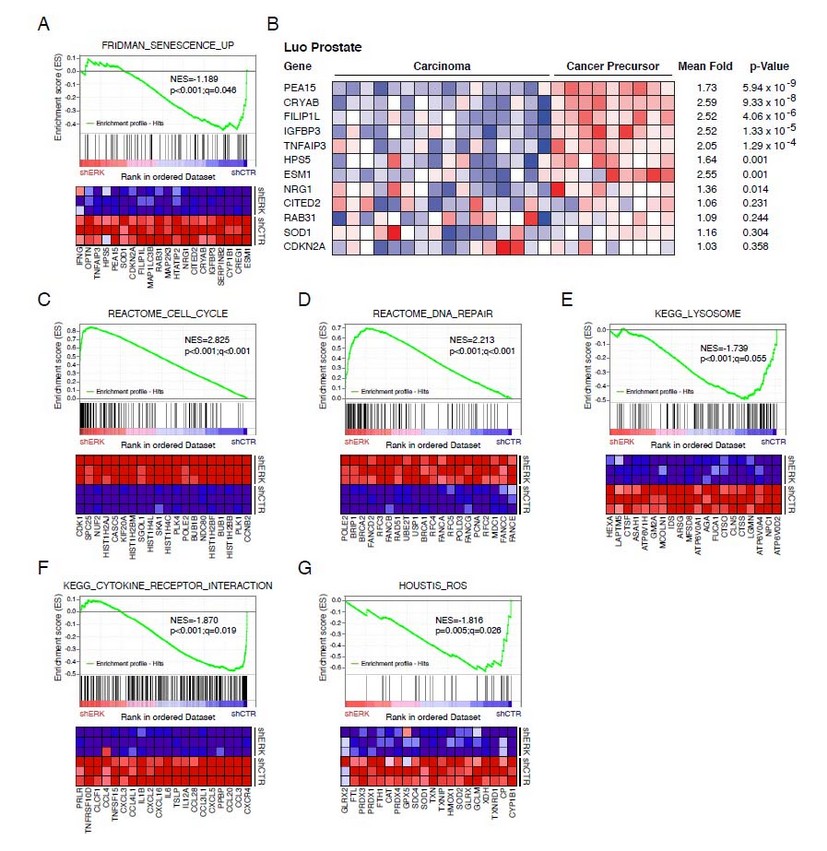
We used GSEA to compare the transcriptome data from both senescent cells induced by oncogenic Ras and cells that escaped from this senescence after inhibition of ERK by and shRNA. We found that several gene sets were significantly enriched in Ras-induced senescent cells compared to cells that bypassed senescence (Figure 2). First, there was an overlap with the senescent gene set defined by Fridman and Tainsky (77 genes) from multiple microarray studies (22) (Figure 2A). Of note, previous data on the transcriptome of senescent cells compared control cells with senescent cells that were not treated to induce senescence. 22 We rather compared senescent cells with cells expressing the same stressor (oncogenic Ras) but which escaped from senescence, thereby refining the senescence gene signature to 20 genes. This list includes PEA-15, a gene that controls the localization of the ERK kinases in senescent cells. 23 A set of these genes is highly expressed in preneoplastic lesions of the prostate (Figure 2B), which is consistent with the concept that senescent cells constitute a barrier for tumor progression and form the bulk of preneoplastic lesions. 3, 5, 12, 24-26 Cell cycle and DNA repair genes were found downregulated in senescent cells (Figure 2C and 2D) in agreement with previous studies showing that the retinoblastoma tumor suppressor pathway repress the E2F transcription factors required for the expression of these genes. 12, 27 Autophagy-dependent protein degradation was found to be highly active in senescent cells 28 explaining the high expression of lysosomal genes observed in our study (Figure 2E). We also found significant overlap with a gene set of cytokines (Figure 2F) and a gene set of genes regulated by reactive oxygen species (Figure 2G). Senescent cells are characterized by mitochondrial dysfunction and production of high levels of reactive oxygen species explaining the induction of ROS-responsive genes.29, 30
Figure 2. Gene set enrichment analysis (GSEA) of genes highly expressed in senescent cells in comparison with cells that escaped from senescence. (A and C-G) In each panel, the enrichment score (ES) along the data set is shown on the x axis of the graphic. Each vertical bar represents a gene, and genes enriched in either condition are at the right (shERK) or left (shCTR) parts of the graph. The normalized enrichment score (NES), the p-value and the false discovery rate (q-value) are indicated in the insert. At the bottom, each panel shows a heat map with the top 20 genes that are highly expressed (red) in either the shERK (escape from senescence) condition or the shCTR (senescent) condition. (B) Heat maps obtained from Oncomine showing the expression of several genes of the Fridman senescent signature (panel A) in samples from benign and malignant prostate tumors.
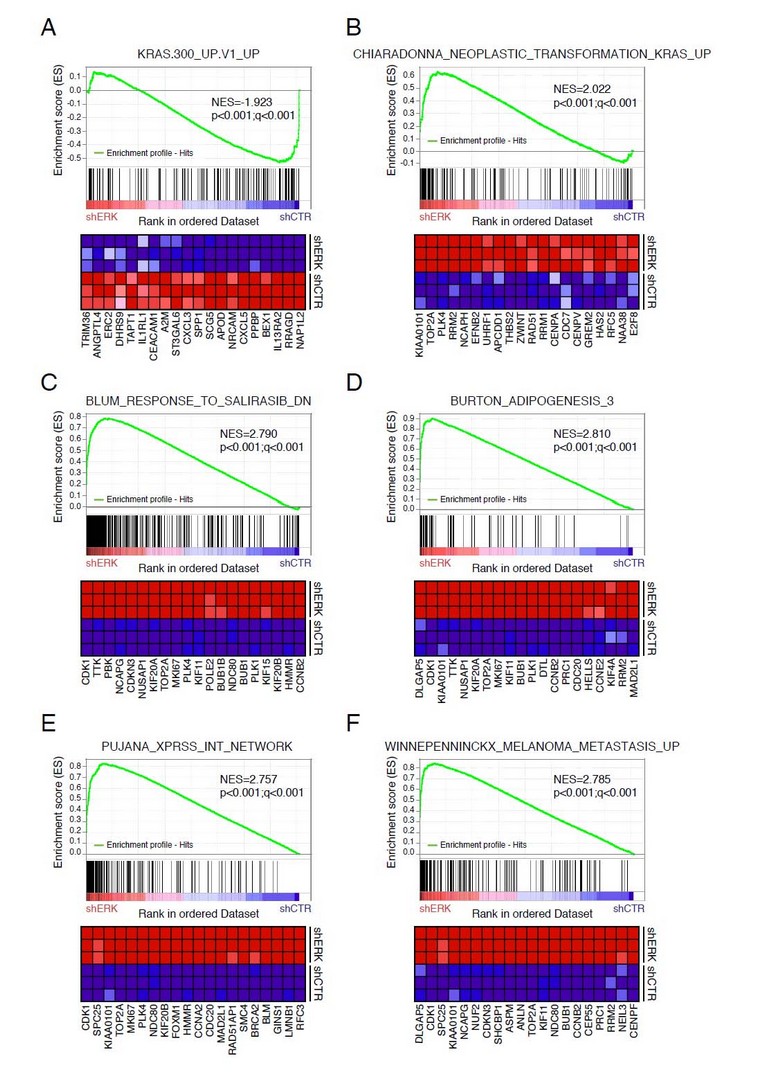
Cells that escape from senescence due to ERK downregulation show a decreased expression of many genes in the set KRAS.300_UP.V1_UP that contains genes upregulated in epithelial cells expressing oncogenic KRAS (Figure 3A). These genes may represent the ERK-dependent targets of the KRAS signalling pathway. Surprisingly, cells that escape senescence also upregulate many genes highly expressed in cells transformed by KRAS (Figure 3B) or genes that were downregulated by salirasib, a RAS inhibitor (Figure 3C). 31 Taken together, the results suggest that the gene expression signature that underlies Ras-dependent transformation does not require high levels of ERK activation. It is also intriguing that cells that escape from senescence express genes linked to the adipogenic conversion of mouse fibroblasts 3T3-L1 (Figure 3D). Ras signalling is known for stimulating adipogenesis in this system while the ERK kinases oppose this process, 32 thus indicating another cellular response controlled by RAS that is not dependent on the ERK MAP kinases. The genes that matched between cells that escape from senescence and adipogenesis likely represent genes required for genome reprogramming since they have been shown to be expressed transiently as a cluster during the first hours of conversion of fibroblasts into adipocytes. 33
Figure 3. Gene set enrichment analysis (GSEA) of genes highly expressed in cells that escaped from senescence in comparison with senescent cells. (A-F) As described for Figure 2.
A network of 118 genes associated to a higher risk of breast cancer (XPRSS-Int network) includes several genes involved in centrosome functions. 34 Cells that escaped from senescence have an enrichment of this gene set (Figure 3E) suggesting that they may also undergo centrosome dysfunction and chromosome instability. We thus analysed the karyotype of cells that escaped from senescence due to ERK inhibition and observed a composite karyotype (cp) with hyperdiploid cells (65-90 chromosomes) and multiple chromosomal aberrations. On the other hand, tumors recovered from mice injected with these cells show a more homogenous chromosome distribution. This suggests that chromosome instability is an inherent property of cells that escape from senescence and that in vivo tumor growth selected for clones with more stable karyotypes. Intriguingly, all cells recovered from mice lost one X chromosome (Table I). Loss of the inactive X chromosome has been frequently observed in cells from breast cancer. 35 Remarkably, many of the genes highly expressed in cells that escaped from senescence and which also overlapped with the adipogenesis program and the XPRSS-Int network matched to a gene set associated to metastatic melanoma (Figure 3F). Together, these results suggest that cells escaping from senescence are highly tumorigenic. In fact, in previous work, we demonstrated that these cells form aggressive tumors in mice. 11
Table I. Karyotype of cells that escape from senescence before or after growth in mice.
Identification of transcription factors regulating the escape from senescence
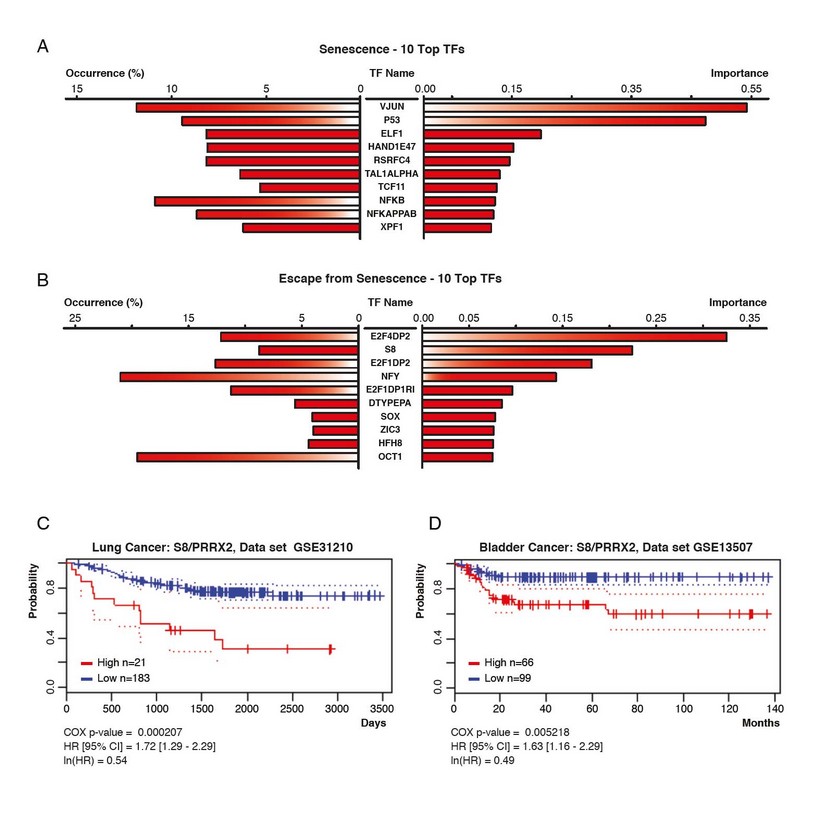
To identify candidate transcription factors that could explain the gene expression changes observed between senescent cells and cells that escaped from senescence, we first used the web-based platform DIRE. 18 The top transcription factors associated to genes upregulated in senescence was JUN (Figure 4A), which is consistent with previous work placing the AP1 (JUN/FOS) transcription factor downstream of the ERK kinases in multiple cellular responses including cell differentiation. 36, 37 The senescence regulators p53 38 and NF-kB 39 were also identified by the algorithm. As expected, transcription factors of the E2F family dominated the list associated to genes upregulated after senescence escape (Figure 4B), indicating that inhibition of the retinoblastoma checkpoint is a key property of growing cells. 12, 27 Interestingly, the transcription factor S8, also known as PRRX2, came second in this list. PRRX2 was recently linked to the TGFb pathway and conferred poor prognosis to breast cancer patients. 40 Using the PrognoScan webserver we found that PRRX2 also conferred bad prognosis to patients with lung and bladder cancer (Figure 4C and 4D).
Figure 4. Transcription factors associated to the transcriptome of senescent cells and cells that escape from senescence according to the algorithm DIRE. (A and B) The percentage of target genes found in the submitted list of transcripts is shown for each potential TF (occurrence). The importance indicates the product of a TF occurrence with is weight in the database. (A) Top transcription factors associated to senescent cells. (B) Top transcription factors associated to cells that escape from senescence. (C and D) Kaplan-Meier and survival analysis using Cox regression which indicates that high expression of PRRX2 is associated to lower survival in lung and bladder cancer.
Discussion
Our gene expression analyses of cells escaping from senescence uncover two apparently contradicting features. First, cells that escape from senescence have multiple genomic abnormalities judging from their highly aberrant karyotype and the expression of genes associated to chromosome instability. The molecular mechanism explaining why cells that escape from senescence exhibit genomic instability remains to be characterized but it could be the consequence of reduced ERK activity. Consistent with this explanation, MEK inhibitors induced genomic instability in glioblastoma cell lines. 41 Second, these cells express a common core of cancer genes previously reported in multiple transcriptomes of cancer cells. The data can be rationalized into a model of carcinogenesis where the escape from senescence provides an initial cellular heterogeneity upon which selective forces act to shape the final cellular composition of the tumor. However, and despite this heterogeneity, these cells converge towards a gene expression program that acts as a universal “cancer attractor” 42 (Figure 5). This attractor operates in different tissues and in cells growing in Petri dishes suggesting that it is an intrinsic genomic response to signals commonly generated in all those dissimilar conditions.
Figure 5. Model of carcinogenesis. Escape from senescence may create a population of unstable cells that can progress into a default proliferative state dubbed the cancer attractor. Therapies that can restore the normal microenvironmental cues might redirect cells that escaped senescence into normal differentiated cells, thus preventing carcinogenesis at its early stages.
More than 80 years ago, Waddington speculated that cancer is the response of cells that escape from the influence of a morphogenetic field. 43 The physical nature of this field remains enigmatic but the idea implies that in the absence of their normal environment cells by default will fall into the “cancer attractor” class and evolve into malignant tumors. On the other hand, differentiation states can be considered as attractors as well, suggesting that providing the right environmental cues, cells that escape from senescence might also redifferentiate and avoid tumorigenesis.
References
1. Bourdeau V, Ferbeyre G. Engaging a senescent response to cure leukemia. Nat Med. 2014;20(2):123-4.
2. Deschenes-Simard X, Lessard F, Gaumont-Leclerc MF, Bardeesy N, Ferbeyre G. Cellular senescence and protein degradation: Breaking down cancer. Cell cycle (Georgetown, Tex. 2014;13(12):1840-58.
3. Chen Z, Trotman LC, Shaffer D, Lin HK, Dotan ZA, Niki M, et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature. 2005;436(7051):725-30.
4. Toso A, Revandkar A, Di Mitri D, Guccini I, Proietti M, Sarti M, et al. Enhancing chemotherapy efficacy in Pten-deficient prostate tumors by activating the senescence-associated antitumor immunity. Cell reports. 2014;9(1):75-89.
5. Guerra C, Collado M, Navas C, Schuhmacher AJ, Hernandez-Porras I, Canamero M, et al. Pancreatitis-induced inflammation contributes to pancreatic cancer by inhibiting oncogene-induced senescence. Cancer Cell. 2011;19(6):728-39.
6. Caldwell ME, DeNicola GM, Martins CP, Jacobetz MA, Maitra A, Hruban RH, et al. Cellular features of senescence during the evolution of human and murine ductal pancreatic cancer. Oncogene. 2012;31(12):1599-608.
7. Maldonado JL, Timmerman L, Fridlyand J, Bastian BC. Mechanisms of cell-cycle arrest in Spitz nevi with constitutive activation of the MAP-kinase pathway. Am J Pathol. 2004;164(5):1783-7.
8. Michaloglou C, Vredeveld LC, Mooi WJ, Peeper DS. BRAF(E600) in benign and malignant human tumours. Oncogene. 2008;27(7):877-95.
9. Michaloglou C, Vredeveld LC, Soengas MS, Denoyelle C, Kuilman T, van der Horst CM, et al. BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature. 2005;436(7051):720-4.
10. Castro P, Giri D, Lamb D, Ittmann M. Cellular senescence in the pathogenesis of benign prostatic hyperplasia. Prostate. 2003;55(1):30-8.
11. Deschenes-Simard X, Gaumont-Leclerc MF, Bourdeau V, Lessard F, Moiseeva O, Forest V, et al. Tumor suppressor activity of the ERK/MAPK pathway by promoting selective protein degradation. Genes Dev. 2013;27(8):900-15.
12. Vernier M, Bourdeau V, Gaumont-Leclerc MF, Moiseeva O, Begin V, Saad F, et al. Regulation of E2Fs and senescence by PML nuclear bodies. Genes Dev. 2011;25(1):41-50.
13. Romanov SR, Kozakiewicz BK, Holst CR, Stampfer MR, Haupt LM, Tlsty TD. Normal human mammary epithelial cells spontaneously escape senescence and acquire genomic changes. Nature. 2001;409(6820):633-7.
14. Deschenes-Simard X, Kottakis F, Meloche S, Ferbeyre G. ERKs in cancer: friends or foes? Cancer Res. 2014;74(2):412-9.
15. Blazejczyk M, Miron M, Nadon R. FlexArray: A statistical data analysis software for gene expression microarrays. Genome Quebec, Montreal, Canada. 2007;http://genomequebec.mcgill.ca/FlexArray.
16. Huang da W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4(1):44-57.
17. Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A. 2005;102(43):15545-50.
18. Gotea V, Ovcharenko I. DiRE: identifying distant regulatory elements of co-expressed genes. Nucleic Acids Res. 2008;36(Web Server issue):W133-9.
19. Tchkonia T, Zhu Y, van Deursen J, Campisi J, Kirkland JL. Cellular senescence and the senescent secretory phenotype: therapeutic opportunities. J Clin Invest. 2013;123(3):966-72.
20. De Heller-Milev M, Huber M, Panizzon R, Hohl D. Expression of small proline rich proteins in neoplastic and inflammatory skin diseases. The British journal of dermatology. 2000;143(4):733-40.
21. Deng Z, Matsuda K, Tanikawa C, Lin J, Furukawa Y, Hamamoto R, et al. Late Cornified Envelope Group I, a novel target of p53, regulates PRMT5 activity. Neoplasia (New York, NY. 2014;16(8):656-64.
22. Fridman AL, Tainsky MA. Critical pathways in cellular senescence and immortalization revealed by gene expression profiling. Oncogene. 2008;27(46):5975-87.
23. Gaumont-Leclerc MF, Mukhopadhyay UK, Goumard S, Ferbeyre G. PEA-15 is inhibited by adenovirus E1A and plays a role in ERK nuclear export and Ras-induced senescence. J Biol Chem. 2004.
24. Bartkova J, Rezaei N, Liontos M, Karakaidos P, Kletsas D, Issaeva N, et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature. 2006;444(7119):633-7.
25. Acosta JC, O'Loghlen A, Banito A, Guijarro MV, Augert A, Raguz S, et al. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell. 2008;133(6):1006-18.
26. Collado M, Gil J, Efeyan A, Guerra C, Schuhmacher AJ, Barradas M, et al. Tumour biology: senescence in premalignant tumours. Nature. 2005;436(7051):642.
27. Chicas A, Wang X, Zhang C, McCurrach M, Zhao Z, Mert O, et al. Dissecting the unique role of the retinoblastoma tumor suppressor during cellular senescence. Cancer Cell. 2010;17(4):376-87.
28. Young AR, Narita M, Ferreira M, Kirschner K, Sadaie M, Darot JF, et al. Autophagy mediates the mitotic senescence transition. Genes Dev. 2009;23(7):798-803.
29. Passos JF, Saretzki G, Ahmed S, Nelson G, Richter T, Peters H, et al. Mitochondrial dysfunction accounts for the stochastic heterogeneity in telomere-dependent senescence. PLoS Biol. 2007;5(5):e110.
30. Moiseeva O, Bourdeau V, Roux A, Deschenes-Simard X, Ferbeyre G. Mitochondrial dysfunction contributes to oncogene-induced senescence. Mol Cell Biol. 2009;29(16):4495-507.
31. Blum R, Elkon R, Yaari S, Zundelevich A, Jacob-Hirsch J, Rechavi G, et al. Gene expression signature of human cancer cell lines treated with the ras inhibitor salirasib (S-farnesylthiosalicylic acid). Cancer Res. 2007;67(7):3320-8.
32. Font de Mora J, Porras A, Ahn N, Santos E. Mitogen-activated protein kinase activation is not necessary for, but antagonizes, 3T3-L1 adipocytic differentiation. Mol Cell Biol. 1997;17(10):6068-75.
33. Burton GR, Nagarajan R, Peterson CA, McGehee RE, Jr. Microarray analysis of differentiation-specific gene expression during 3T3-L1 adipogenesis. Gene. 2004;329:167-85.
34.Pujana MA, Han JD, Starita LM, Stevens KN, Tewari M, Ahn JS, et al. Network modeling links breast cancer susceptibility and centrosome dysfunction. Nat Genet. 2007;39(11):1338-49.
35.Sirchia SM, Ramoscelli L, Grati FR, Barbera F, Coradini D, Rossella F, et al. Loss of the inactive X chromosome and replication of the active X in BRCA1-defective and wild-type breast cancer cells. Cancer Res. 2005;65(6):2139-46.
36. Lopez-Bergami P, Huang C, Goydos JS, Yip D, Bar-Eli M, Herlyn M, et al. Rewired ERK-JNK signaling pathways in melanoma. Cancer Cell. 2007;11(5):447-60.
37. Leppa S, Saffrich R, Ansorge W, Bohmann D. Differential regulation of c-Jun by ERK and JNK during PC12 cell differentiation. EMBO J. 1998;17(15):4404-13.
38. Levine AJ, Oren M. The first 30 years of p53: growing ever more complex. Nat Rev Cancer. 2009;9(10):749-58.
39. Moiseeva O, Deschenes-Simard X, St-Germain E, Igelmann S, Huot G, Cadar AE, et al. Metformin inhibits the senescence-associated secretory phenotype by interfering with IKK/NF-kappaB activation. Aging Cell. 2013;12(3):489-98.
40. Juang YL, Jeng YM, Chen CL, Lien HC. PRRX2 as a novel TGF-beta-induced factor enhances invasion and migration in mammary epithelial cell and correlates with poor prognosis in breast cancer. Mol Carcinog. 2016.
41. Stepanenko AA, Andreieva SV, Korets KV, Mykytenko DO, Baklaushev VP, Chekhonin VP, et al. mTOR inhibitor temsirolimus and MEK1/2 inhibitor U0126 promote chromosomal instability and cell type-dependent phenotype changes of glioblastoma cells. Gene. 2016;579(1):58-68.
42. Huang S, Ingber DE. A non-genetic basis for cancer progression and metastasis: self-organizing attractors in cell regulatory networks. Breast Dis. 2006;26:27-54.
43. Waddington CH. Cancer and the theory of organizers. Nature. 1935;135:606-8.
Recibido: Marzo de 2016.
Aprobado: Junio de 2016.
1- Faculty of Medicine, McGill University, Montréal, Québec, H3G 1Y6, Canada
2-Department of Stomatology, Université de Montréal, C.P. 6128, Succ. Centre-Ville, Montréal, Québec, H3C 3J7, Canada.
3- Department of Biochemistry and Molecular Medicine, Université de Montréal, C.P. 6128, Succ. Centre-Ville, Montréal, Québec, H3C 3J7, Canada
Corresponding author: Department of Biochemistry and Molecular Medicine, Université de Montréal, C.P. 6128, Succ. Centre-Ville, Montréal, Québec, H3C 3J7, Canada
*Correspondence: [email protected]