2023.08.03.97
Files > Volume 8 > Vol 8 No 3 2023
Study pathological changes to evaluate efficient treatment of industrial dyes in wastewater
1 Ministry of Science and Technology;[email protected]. Irak
2 Al-Isra'a University College. Irak
3 Ministry of Agriculture. Irak
4 Ministry of Science and Technology, Irak
* Correspondence: [email protected]
Available from: http://dx.doi.org/10.21931/RB/2023.08.03.97
ABSTRACT
There is an increase in concern about the effects of environmental dangers on health related to toxic pollutants resulting from industrial activities, which are released into the aqueous environments in the whole world. Industrial dyes represent one of the major groups of toxic compounds used in different industries, including textile and leather. Releasing dyes into the water could cause vicious effects on the aqueous environment. This experiment aims to evaluate the efficiency of chemical treatment by Nano zero-valent iron (NZVI) of textile dye to investigate the histopathological effects in the liver, kidneys, and intestines of albino mice using histopathological diagnosis.
Three groups of mice were exposed. The control group was given tap water, the second group was assigned untreated textile dye solution (acid red 315) at a concentration of (100)ppm, and the third group was given treated textile dye solution. Histopathological alterations were evaluated after 2 months from exposure. The results from treated dyes showed no histopathological changes. In contrast, untreated stains showed histopathological changes in all organs, represented by dilation of sinusoids and congestion of blood vessels and hydropic degeneration, necrosis and aggregation of inflammatory cells in the liver. In contrast, in the intestine, there is edema between mucosa and submucosa, degeneration and necrosis in the intestinal mucosa, atrophy in the muscular layer, and inflammatory cell aggregation in the submucosa and mucosa of the intestine. We conclude that the treatment of textile dye is sufficient and decreases its effect.
Keywords: Histopathological alteration, Efficient evaluation, Textile dye acid red 315
INTRODUCTION
The textile industry is reported to be a vital, joint and essential industry sector in the world and plays a distinguished role in the nation's economy 1 and is considered a source that emerged in the 18th century as a mass production and became an important industry. The dye industry has been considered an essential source of water pollution because dyes are highly colored and resist degeneration. Workers in the textile industry are exposed to many different types of toxic dyes, bleaching agents, acids, alkaline and heavy metals like cadmium, chromium and iron, and also possibly exposed to carcinogenic compounds such as solvents and fixatives during the printing process of dyes on textile2. Most of the time, water from the textile dye industry, which is less than 10%, is treated, and the remainder is released into the drains and shallow pools untreated, causing a severe pollution problem 3. The final disposal of the treated dyes remains a challenge, and the ecto-taxological evaluation is essential to minimize environmental pollution; 10-15 % of the stains are released into the environment throughout the dying process and are considered carcinogenic and mutagenic 4. Large numbers of animals, including cattle, drink contaminated water because of the lack of access to safe water, and accidental pool drinking results in calf mortality 5. The toxicity of Azo dyes based on benzidine and congeners can cause bladder cancer in humans. Toxic effects were reported in several exposed species, including humans6, monkeys, rodents and dogs7. The study aimed to evaluate the efficient treatment of industrial dyes in wastewater.
MATERIALS AND METHODS
.Animals
consist of ten mice (five female and five male). The experiment persisted for two months. Mature Swiss albino mice (age 50-55 days), weighting 25-30 g, were housed in plastic cages of (60*10*10) cm3 dimension, well ventilated, temperature 25± 3 C°, 12h light-dark cycle and feed slandered rodent diet provided by the department of pharmaceutical control/ branch of experimental animals and drinking water were given. Animals were divided into three groups, including the control group, the untreated dye solution group and the treated dye solution group. Each group
Preparation of stock solution and exposure details
Azo dye acid red 315 (Figure. 1) used in this study was obtained commercially from the local source and directly used for experimental purposes. We prepared a 100 ppm concentration of azo dye and then treated it with zero iron in the chemical laboratory of the environmental and water directorate/Ministry of Science and Technology.
Histopathological studies
Specimen of organs (liver, kidneys, lungs, brain, spleen and intestine) were collected from all animals after killing immediately and washed with normal saline and then fixed in 10% neutral buffered formalin for 48 hours at least for histopathological diagnosis
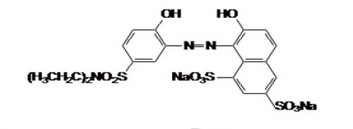
Figure 1. Chemical Structure of Acid Red 315 C20HI9N3Na2010S3,M.W 603.56 in the Study
RESULTS AND DISCUSSION
The result of this study showed histopathological alteration, as in Table (1). Histopathological responses have commonly been used as effective biomarkers for evaluating the effects of pollutants on health.
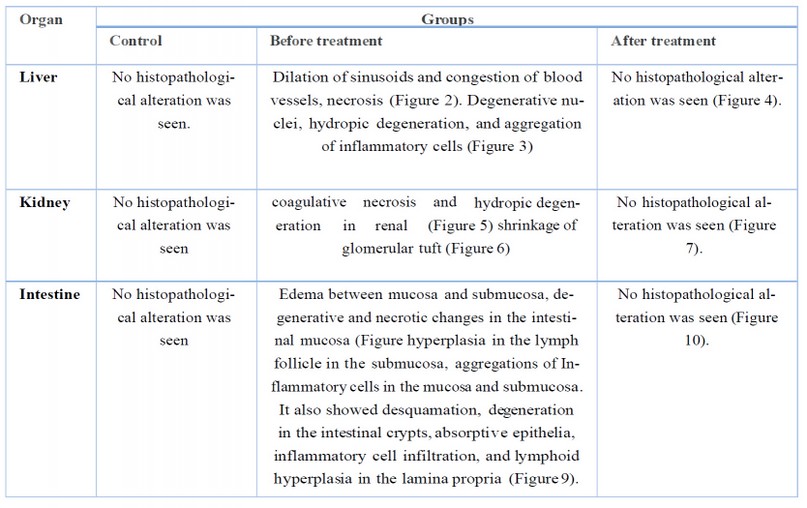
Table 1.The results of histopathological alteration
Histopathology of liver
The liver is associated with the detoxification and biotransformation process, and due to its function, position and blood supply, it is also one of the organs most affected by contaminants in the water 8. Gil and Cemek 9 reported that azo dyes are metabolized by azo reductase in the liver 10. The liver has a vital detoxification role of endogenous waste products and externally derived toxic chemicals or heavy metals11. Dilation of sinusoids and congestion of blood vessels, necrosis and aggregation of inflammatory cells and pycnotic nuclei, and hydropic degeneration changes agreed with our result after 2 months of exposure (Figures 2 and 3).
Also, Atamanal et al.12 show similar changes in rainbow trout exposed to copper sulfate. Abdel Moneim et al.13 observed cloudy swelling and mass and infiltration of inflammatory cells in the liver. Also, Hinton et al. 14 suggest that Hepatic necrosis and inflammation indicate toxic injury. Koca et al. 15 reported several histopathological alterations, such as swollen and ruptured parenchymal cells, loss of cord structure, vacuoles filled with cellular debris, focal necrosis and a significant increase in Kupffer cells in the liver due to metal accumulation. Results of the treatment group show no histopathological changes as in Figure 4.
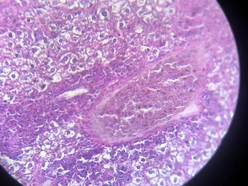
Figure 2. Showing congestion with periportal cuffing (before treatment)
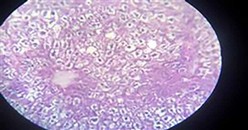
Figure 3. Showing liver with hydropic degeneration (before treatment)
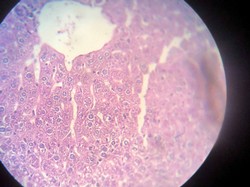
Figure 4. Showing normal liver tissue
Histopathology of kidney
The kidney showed coagulative necrosis (Figure 5) and hydropic degeneration in renal tubules, shrinkage of glomerular tuft (Figure 6) similar to 17, which showed some detrimental changes like atrophy of glomeruli, and degeneration of renal Tubules kidney. Other researchers mention that the histopathological changes in the kidney after exposure to different dye concentrations might indicate kidney dysfunction or failure 13. Whereas in the group of mice exposed to textile dye solution after treatment, no histopathological alteration was seen (Figure 7).
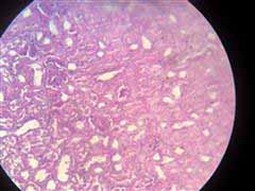
Figure 5. Showing necrosis in renal tubules and shrinkage of glomerular tuft (before treatment)
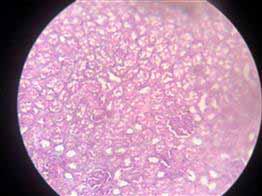
Figure 6. Showing coagulative necrosis and hydropic degeneration in the tubules of the kidney (before treatment)
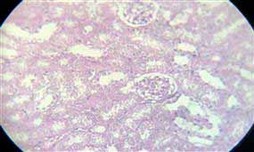
Figure 7. Normal kidney tissue
Histopathology of intestine
The intestine is involved in the uptake of pollutants from the surrounding medium. The histological change in the intestine for mice exposed to 100 textile dyes after two months is edema between mucosa and submucosa (Figure 8), which may be attributable to degradation products, including toxic metabolites characterized as aromatic amines 18.
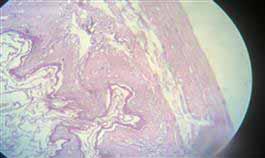
Figure 8. Showing edema between mucosa and submucosa, degenerative and necrotic
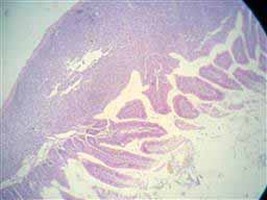
Figure 9. Showing hyperplasia in the lymph follicles of sub mucosa (before treatment).
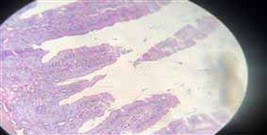
Figure10. Showing intestine with no histopathological changes (after treatment)
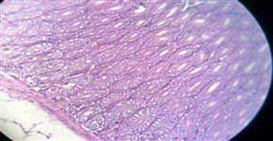
Figure 11. Showing normal intestine
CONCLUSIONS
The study concludes that the chemical treatment of textile dye with nano zero-valent iron (NZVI) effectively reduces its toxicity and histopathological effects in albino mice. The untreated dye caused a variety of histopathological changes in the liver, kidneys, and intestine, including dilation of sinusoids and congestion of blood vessels, hydropic degeneration, necrosis, aggregation of inflammatory cells, edema, degeneration and necrosis of the intestinal mucosa, atrophy of the muscularis layer, and inflammatory cells aggregation in the submucosa and mucosa of the intestine. However, the treated dye showed no histopathological changes in any organs.
These findings suggest that NZVI treatment could be a viable method for reducing the environmental and health risks associated with textile dye pollution. However, further research is needed to evaluate this treatment's long-term safety and efficacy.
In addition, the study highlights the importance of treating textile dye wastewater before it is released into the environment. Textile dye pollution is a global problem that can significantly impact aquatic ecosystems and human health. By treating textile dye wastewater, we can help to protect the environment and safeguard public health.
.
REFERENCES
- Bos RP, Van Der Krieken W, Smeijsters L, Koopman JP, De Jonge HR, Theuws JL, Henderson PT. Internal exposure of rats to benzidine derived from orally administered benzidine-based dyes after intestinal azo reduction. Toxicology. 1986 Aug 1;40(2):207-13.
- Sharma KP, SHARMA K, Bhardwaj SM, Chaturvedi RK. Environment impact assessment of textile printing industries in Sanganer, Jaipur: A case study. Journal of the Indian Botanical Society. 1999;78(1-2):71-85
- International Agency for Research on Cancer (IARC). Monographs on the evaluation of carcinogenic risk to humans. Lyon.1997; 69: 545-51.
- Tiwari H. Reproductive performance of Swiss albino mice treated with leachate from CETP Pali, administered during various reproductive cycle stages. Research Journal of Chemical and Environmental Sciences. 2013;1(3).
- Sharma K. Environment impact assessment of textile industry wastewaters in Sanganer environment. Doctoral dissertation, Ph.D. Thesis, Rajasthan University, Jaipur.2000
- Lowry LK, Tolos WP, Boeniger MF, Nony CR, Bowman MC. Chemical monitoring of urine from workers potentially exposed to benzidine-derived azo dyes. Toxicology Letters. 1980 Nov 1;7(1):29-36.
- Rinde E, Troll W. Metabolic reduction of benzidine azo dyes to benzidine in the rhesus monkey. Journal of the National Cancer Institute. 1975 Jul 1;55(1):181-2.
- Camargo MM, Martinez CB. Histopathology of gills, kidney and liver of a Neotropical fish caged in an urban stream. Neotropical ichthyology. 2007;5:327-36.
- Cemek M, Büyükokuroğlu ME, Sertkaya F, Alpdağtaş S, Hazini A, Önül A, Göneş S. Effects of food color additives on antioxidant functions and bioelement contents of liver, kidney and brain tissues in rats. J Food Nutr Res. 2014 Sep 24;2(10):686-91.
- Gil C. Toxicological effects of food additives azo dyes MS thesis Swedish University of Agricultural Sciences, Uppsala, Sweden.2014.
- Authman MM, Abbas HH. Accumulation and Distribution of Copper and Zinc in Both Water and Some Vital Tissues of Two Fish Species(Tilapia zillii and Mugil cephalus) of Lake Qarun, Fayoum Province, Egypt. Pakistan Journal of Biological Sciences. 2007 Jul 1;10(13):2106-22.
- Atamanalp M, Sisman T, Geyikoglu F, Topal A. The histopathological effects of copper sulfate on rainbow trout liver (Oncorhynchus mykiss). J. Fish. Aquat. Sci. 2008;3(5):291-7
- Abdel-Moneim AM, Abou Shabana NM, Khadre SE, Abdel-Kader HH. Physiological and histopathological effects in catfish (Clarias lazera) exposed to dyestuff and chemical wastewater.2008.
- Hinton DE, Baumann PC, Gardner GR, Hawkins WE, Hendricks JD, Murchelano RA, Okihiro MS. Histopathologic biomarkers. InBiomarkers 2018 Jan 18 (pp. 155-210). CRC Press.
- Koca S, Koca YB, Yildiz Ş, Gürcü B. Genotoxic and histopathological effects of water pollution on two fish species, Barbus capito pectoralis and Chondrostoma nasus in the Büyük Menderes River, Turkey. Biological Trace Element Research. 2008 Jun;122(3):276-91.
- Himri I, Bellahcen S, Souna FA, Belmekki F, Aziz M, Bnouham M, Zoheir J, Berkia ZO, Mekhfi H, Saalaoui EA. A 90-day oral toxicity study of tartrazine, a synthetic food dye, in Wistar rats. Group. 2011;300(00).
- Himri I, Bellahcen S, Souna FA, Belmekki F, Aziz M, Bnouham M, Zoheir J, Berkia ZO, Mekhfi H, Saalaoui EA. A 90-day oral toxicity study of tartrazine, a synthetic food dye, in Wistar rats. Group. 2011;300(00).
- Fatima M, Saeed M, Aslam M, Lindström RW, Farooq R. Application of novel bacterial consortium for biodegradation of aromatic amine 2-ABS using response surface methodology. Journal of Microbiological Methods. 2020 Jul 1;174:105941.
Received: 25 June 2023/ Accepted: 26 August 2023 / Published:15 September 2023
Citation: Mustafa S R, Hadeer A.A., Ali V, Hakeem Ali S A.Study pathological changes to evaluate efficient treatment of industrial dyes in wastewater. Revis Bionatura 2023;8 (3) 97 http://dx.doi.org/10.21931/RB/2023.08.03.97
