2019.04.03.3
Files > Volume 4 > Vol 4 No 3 2019
INVESTIGATION / RESEARCH
Composite of chitosan and Bentonite as coagulant agents in removing turbidity from Ismailia canal as water treatment plant.
A. Marey
Available from: http://dx.doi.org/10.21931/RB/2019.04.03.3
ABSTRACT
The use of chemical coagulants is not suitable due to health and economic considerations. In the last time, the use of natural additives that are biocompatible, are biodegradable, have low toxicity and are from renewable resources attracted the attention of many types of research due to their high ability to retain different pollutants from wastewaters. Ismaili Canal is one of the most important tributaries of the River Nile in Egypt. This study successfully proved the effectiveness of the combination of chitosan and bentonite as a coagulating agent in the (Ismailia canal) for the wastewater treatment process. Chitosan and Bentonite were known as coagulant agents, thus were used in this study. Laboratory tests were conducted to test the water quality based on turbidity values and basic drinking water parameters. Chitosan and Bentonite can be applied on (Ismailia canal) resulting in a turbidity removal 97.7% at 30 minutes. The optimal dosage of bentonite(clay) combined with chitosan(polymer) was 5 and 1mg/L at PH=7.4.
Keywords: Chitosan; Bentonite; wastewater; Ismailia canal.
INTRODUCTION
There are many research studies highlight the bio sorbent ability of chitosan and their composites for the pollutants from wastewaters such as heavy metal ions, organochloride pesticides, suspended solids, turbidity, organic oxidized substances, fatty and oil impurities or textile wastewater dyes. The natural water becomes wastewater after different usage, and finally, complete the hydrological cycle. The water becomes wastewater due to population growth, urbanization, industrialization; sewage from household, institutions, hospitals, industries, etc. wastewater can be destructive for the public because it contains a variety of organic and inorganic substances, biological substances, toxic inorganic compounds and the presence of toxic materials. The natural polymers are most efficient that provide several benefits such as prolific, except for physical and chemical changes from the treated water. Chitosan is the second largest biopolymer after cellulose. Chitosan (CS) is the N-DE acetylated derivative of chitin. It is characterized by high hydrophobicity and many hydroxyls and amino groups. It is environmentally friendly biodegradable and biocompatible material 1. CS has minimum toxicity, and it is highly available in nature. Because of its high hydrophilicity and many amino and hydroxyl groups, CS presents good compatibility with clay minerals. As known adsorbent, CS is widely used to remove heavy, transition metals and dyes from wastewaters because the amine group (-NH2) and hydroxyl group(-OH) on the polymer chain of CS can adsorb both cationic and anionic molecules 2.

CONCLUSIONS
Bentonite has a strong adsorption capacity because of its large surface area and surface energy. In this study, we focused on the Bentonite pre-treatment. Bentonite used in this study were both intercalated and without treatment. By intercalating process, the interlayer of Bentonite might be wider, and the chitosan could enter into the layers of Bentonite structure. The use of bentonites can improve the size and the density of the formed flocs which can increase its flocculation rate and at the same time can promote the adsorption of organics 3 for a given level of ”contaminants removal.” The use of bentonites can help to reduce the required dosage of chitosan and thus, the treatment costs. The objective of this study first evaluated the efficiency of different chitosan treatments for the removal of dissolved and colloidal materials .the possible efficiency improvement on using chitosan derivatives or by combined addition of the native chitosan with anionic bentonite microparticles. Ismailia Canal was constructed in 1862 by two agreements between the Egyptian government and the Suez Canal Company for creating waterway between the Nile and the Suez Canal. Ismailia Canal is one of the most branches of the Nile River in Egypt. It is the main source of drinking and irrigation water in many cities. The canal has its inlet from the Nile at Cairo and runs directly to the east to the town of Ismailia passing the governorates of Cairo- Kalioubeya-Sharkeya and Ismailia. Ismailia Canal is one of the most important tributaries of the River Nile in Egypt .along the canal there are several sources of pollution industries in the region of Cairo as well as agricultural run-offs in the Eastern part. Measuring of a specific physic-chemical parameter in the contaminated aquatic ecosystem is important in determining its effects on the living organisms inhabiting that environment 4. The purpose of the present work was to remove the turbidity in Ismailia Canal wastewater by the composite of Chitosan-Bentonite Coagulant.
MATERIALS AND METHODS
Materials
The wastewater used in this study was obtained from the Ismailia Canal (Mostourd branch): Turbidity (TSS) is (100.7) NTU, PH= (8.32), Temperature= (24.04oC). Samples of canal water were taken in a one-gallon container and transported to the laboratory.1 M NaOH, 1% acetic acid. Tools used in this study include beaker glass, stir bar, PH.
Coagulants
Chitosan (Deacetylated chitin: poly-[1-4]-B-glucosamine). (C6H11NO4) N with a minimum 85% deactyl prepared from crab shells was obtained from ACROS ORGANICS Company. It was in the form of a pale brown powder soluble in dilute acetic acid, hydrochloric acids. With molecular weight 100.000 -300.000. The bentonite powder is prepared with chemical reaction by dissolving the 50gm bentonite to 100 ml oh HCl at 10M. The magnetic stirrer was employed to mix the solution at 300 rpm and temperature 70oC.
Preparation of Chitosan-Bentonite Composite.
The most common way of preparing the chitosan/bentonite composite materials is by dissolving chitosan (CS) in an acidic (acetic, hydrochloric, formic) solution.The bentonite is first swelled in distilled water and then added to the chitosan solution followed by stirring — the PH of the resulting solution adjusted with sodium hydroxide (or) hydrochloric acid. The chitosan-bentonite beads are filtered and washed with water to remove excess sodium hydroxide (or) hydrochloric acid, then dried in the oven until the weight becomes constant, then grinding the particles 5.
In some cases, before or after the mixing of bentonite with the chitosan solution, they are cross-linking agents are used to enhancing the mechanical strength and stability of chitosan in acidic media 5.
The most favorable concentration weight ratio of the natural polymer to natural clay of bentonite is in between 1:5 to 1:20 6.to achieve a good removal; the optimum dosage needs to be studied. The term “primary coagulant” refers to the main coagulant which formed a metal salt (bentonite) added alone or being added together with the secondary coagulant as “coagulant aid”. The term “coagulant aid” refers to a polymer substance 6.anionic particles of bentonite are electrostatically attracted by the protonated amino groups of chitosan 7. This reaction facilitates the neutralization of the anionic charges which can bind together and settle rapidly by the effect of gravity 8.the natural coagulant currently is very famous and being used in most water treatment plants. It is proven that low doses of the natural polymer of chitosan together with bentonite as coagulant aid can perform better in the removal of particulate matter and color from water 8.the chitosan coated bentonite composite was prepared by dissolving 1g of deacetylated chitosan into 100 ml of 5% hydrochloric acid — the solution in for 6 hours. Then added 5g of bentonite slowly and the mixture was stirred for 3 hours and then added 1ml of sodium hydroxide aqueous solution dropwise until PH=7 to precipitate the dissolved chitosan into bentonite. Then the mixture was vacuum filtered and dried in an oven overnight at 600C.
In this study we prepare (4 beakers) were filled with 500ml water from (Ismailia Canal).then the composite was added with different dosages and mixed at 100 rpm for 5minutes. Then reduce the speed to 50 rpm for 15 minutes. Then the mixer is turned off, and flocs are allowed to settle for 30 minutes 9.
XRD analysis
The XRD is a fast resolution technique used to identify the crystal phase of a crystalline material and can provide information about the morphology.
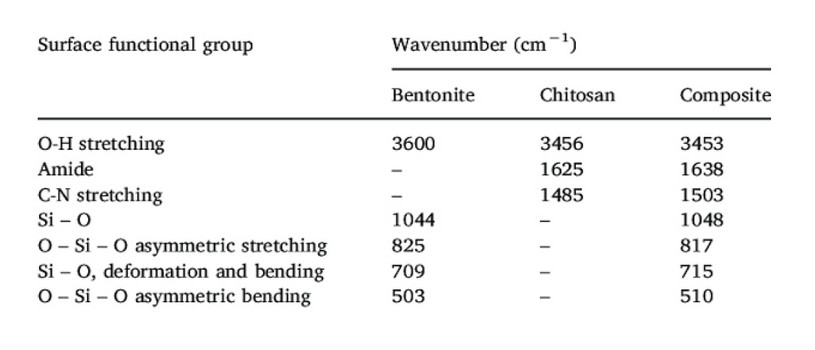
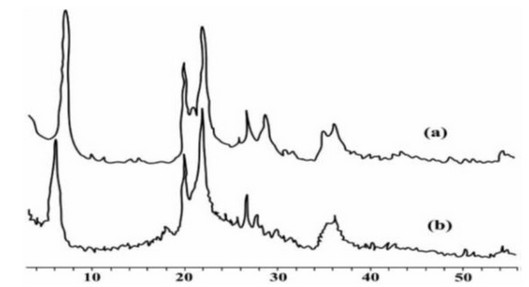
In most studies 10, 11, 12, 13, the prepared chitosan/bentonite composites showed different diffraction patterns from the two pure components (chitosan and bentonite). It was found that the diffraction peak of chitosan/bentonite slightly shifted to a lower angle compared to the one of raw bentonite. Also, a decrease in the intensity of this peak was observed [Figure 1]. This decrease can be attributed to a slight deformation of silicate layers and a decrease in the crystallinity caused by the interaction of bentonite with chitosan

Table 1. Functional groups of chitosan, bentonite and composite coagulants.

Figure 1: XRD patterns of (a) Bentonite; (b) chitosan/bentonite beads. 14
SEM analysis
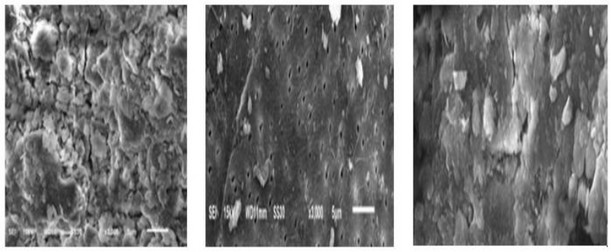
The Scanning Electron Microscope (SEM) is a common technique for characterization of the surface morphology and physical properties of the adsorbent. It is used to determine the particle shape, size distribution, and porosity. Fig (1) displays SEM pictures of raw bentonite and chitosan-bentonite composite before the adsorption process. The roughness and irregular surface morphology of the raw bentonite illustrated on the shape (a) shows a more porous material. Upon modification to form chitosan-bentonite, the sharp edges observed on the raw bentonite material are smoothened, and the distinguished gray areas have darkened. This could be an indication of a successful coating of the chitosan onto the bentonite material. And in Fig (2) shows SEM images of unmodified bentonite (a) usually show bulk agglomerated particles tightly bound together by intermolecular forces. While SEM images of chitosan usually show that its surface is tight and porous (b).but in (C) appears that bentonite is covered by chitosan which shows dense accumulation, where the uneven surface is covered by many peaks and activities, because chitosan molecules interact with bentonite on the surface.
(a) (b)

Figure 2: A Characterization of Bentonite (a) –Chitosan Composite (b). (SEM analysis). 15

Figure 3. Example of SEM analysis of (a) bentonite; (b) chitosan and (c) complex bentonite/chitosan 16.
RESULTS AND DISCUSSION
Turbidity is an important parameter to be analyzed because it is a parameter that can be clearly seen. Increasingly clear water processing results (percent close to 100% means the better the results processing.
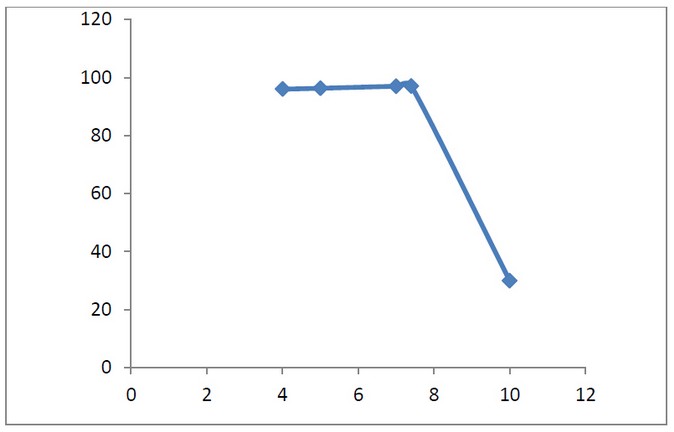
Effect of PH
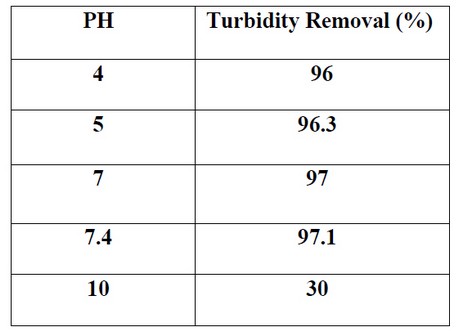
The turbidity removals for Chitosan-Bentonite coagulation at different PH, in the experiment we adjusting the PH from 4 to 10, using the dosage of 500mg/l with mixing time 15 minutes. The difference in PH from 4 to 8 is not very much.the coagulants provide the best result of turbidity removal when the condition is acidic or slightly basic, which shows with 97.7%effeciency. The turbidity removal is decreased or distorted when the PH is over 8.this is because the positive charges on the chitosan-bentonite surface decreases as PH increases, so will contribute to the decreasing charge of both chitosan and bentonite to attract the negatively charged suspended particles.

Table 2. Effect of PH with Turbidity Removal percentage (%).

Figure 4: Shows Effect of PH with Turbidity Removal percentage (%).
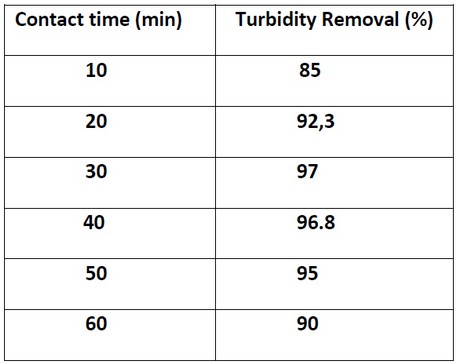
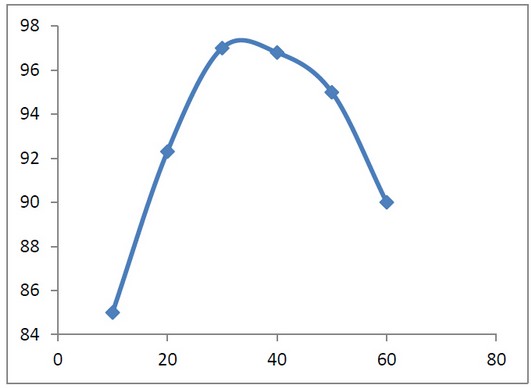
Effect of contact Time (min)
The effect of mixing time with a concentration of 500mg/L with PH=7.4 with coagulant concentration (chitosan-bentonite) is 1:5. It was conducted with 120 rpm from 10 to 20 minutes and slowly from 40 to 60 and remained under settling time of 30 minutes to allow the settlement. When analyzing the data represented that 97.1% of turbidity reduction achieved in 30 minutes (optimum condition). By analyzing the reason, at lower agitation time, for example, 10 minutes, the collisions between the coagulant and suspended particles are low and will lead to lower flocculation rate 17. Instead, if the mixing time is too long, the flocculation chains tend to stop, the small size flocs are not dense to settle down in raw water, indirectly cause the sample to be turbid again. 17

Table 3. Effect of contact time with Turbidity Removal percentage (%).

Figure 5: Shows Effect of contact time with the Turbidity Removal percentage (%).
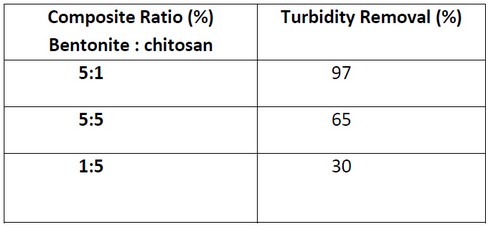
The Effect of Bentonite: Chitosan Ratio on turbidity removal.
To study the Effect of ratio bentonite to chitosan 5:1, 5:5, and 1:5. At optimum PH=7.4 and optimum time 30 minutes. From various mixture ratios of Chitosan-Bentonite, the result shows that 5:1 is the optimum ratio with the highest turbidity removal efficiency of 97%. Effective coagulation was achieved with much lower doses of chitosan for complete neutralization of bentonites charge 18. This means, Bentonite has less positive charge than the chitosan; one particle of chitosan is enough to attract and bind five particles of Bentonite to perform the best coagulant agent. Overdosing of chitosan causes slightly decreased in removal efficiencies, due to reversal surface charge .when the chitosan dosage exceeds the saturation of polymer bridging and the remain of chitosan destroy the polymer bridge between particles and the remaining increase in residual 19.

Table 4: Effect of Bentonite:Chitosan Ratio on turbidity removal

Fig 6: Effect of Bentonite: Chitosan Ratio on turbidity removal
CONCLUSIONS
This study successfully proved the effectiveness of the combination of chitosan and bentonite as a coagulating agent in Ismailia canal as a water treatment plant. The optimal conditions were determined based on turbidity removal. In this research, the optimum conditions of the synthesis of composite chitosan-bentonite were at a weight ratio of bentonite/ chitosan 5:1 and time 30 minutes and an optimal PH of 8. Coagulation with chitosan-Bentonite successfully removed the turbidity with the efficiency of 97.7%. On these conditions, more chitosan can interact with the matrix of bentonite to form a chitosan-bentonite composite. The chitosan located on the outer surface of the bentonite and don’t enter in the layer structure of bentonite.
REFERENCES
1. Chang MY, Juang RS. Adsorption of tannic acid, humic acid, and dyes from water using the composite of chitosan and activated clay. J Colloid Interface Sci. 2004; 278: 18–25.
2. Chatterjee T., Chatterjee S., Seung Han Woo. Enhanced coagulation of bentonite particles in water by a modified chitosan biopolymer, Department of Chemical Engineering, Hanbat National University, Korea, 2009.
3. Guo J, Chen S, Liu L. Adsorption of dye from wastewater using chitosan-CTAB modified bentonites. J Colloid Interface Sci. 2012; 382: 61–66.
4. Hariani PL, Fatma F, Riyanti F. Adsorption of Phenol Pollutants from Aqueous Solution Using Ca-Bentonite/Chitosan Composite. J Mns dan Lingkung. 2015; 22: 233.
5. Hassan M. A., Tan P. L., Zainura Z. N. Coagulation and flocculation treatment of wastewater in textile industry using chitosan,Faculty of Chemical and Natural Resources Engineering, UTM, 2008.
6. Huang R, Liu Q, Zhang L, et al. Utilization of cross-linked chitosan/ bentonite composite in the removal of methyl orange from aqueous solution. Water Sci Technol. 2015; 71: 174–182.
7. Huang R, Zheng D, Yang B, et al. Preparation and characterization of CTAB-HACC bentonite and its ability to adsorb phenol from aqueous solution. Water Sci Technol. 2011; 64: 286–292.
8. Mbakop S, Nthombeni N, Onyango M.S. Evaluation of Chitosan-Bentonite Composite Performance towards Remediation of Sulphate Containing Effluent. Annual Conference on Sustainable Research and Innovation. 2016; 4-6.
9. Murcott S. E, Donald R. Herleman F. Method of drinking water treatment with natural cationic polymers, Massachusetts,Institute of Technology, Cambridge, 1996.
10. Noor Z. Z., Hassan M. A, Lim S. H., Zaini U. Removal of boron from industrial wastewater by chemical precipitation using chitosan, Faculty of Chemical and Natural Resources Engineering, UTM, 2008.
11. Pan, J. R., Chih P., Huang, S. C., Chen, Ying C. Evaluation of modified chitosan biopolymer for coagulation of colloidal particals. Colloids and Surfaces A: Physicochemical and Engineering Aspects 1999; 147: 359-364.
12. Pereira FAR, Sousa KS, Cavalcanti GRS, et al. Chitosan-montmorillonite biocomposite as an adsorbent for copper (II) cations from aqueous solutions. Int J Biol Macromol [Internet]. 2013; 61: 471–478.
13. R. Nicu, R. Miranda, E. Bobu, A. Blanco, Improved efficiency of chitosans with bentonites forthe retention and drainage of pulp suspensions. Submittted to Bioresources (2013).
14. Sakaew S, Umpuch C. Removal of Azo Dyes from Aqueous Solution by using Chitosan-coated- Montmorillonite clay. 2011; 46: 172-178.
15. Shellshear, M. S. Urban stormwater treatment using chitosan. B. Eng. In civil Engineering Thesis, Faculty of Engineering and Surveying, University of Southern Queensland, 2008.
16. Syafalni, Abustan, Farhana. Raw water treatment using Bentonite-Chitosan as coagulant, Water Science & Technolgy: Water Supply, 2012; 480-487.
17. Wan Ngah WS, Ariff NFM, Hanafiah MAKM. Preparation, characterization, and environmental application of crosslinked chitosancoated bentonite for tartrazine adsorption from aqueous solutions. Water Air Soil Pollut. 2010; 206: 225–236.
18. Wan Ngah W S et al 2011 Carbohydr.Polym.83 1446-1456.
19. Wrona EJ, Cash KJ. 1996. The ecosystem approach to environmental assessment: moving from theory practice Agu.Ecosys.5, 89-97.
Received: 20 April 2019
Accepted: 20 June 2019
A. Marey
Department of Basic Science, The Valley Higher Institute for Engineering and Technology, Cairo, Egypt.
Corresponding author: [email protected]
