2021.06.04.2
Files > Volume 6 > Vol 6 No 4 2021
CARTA AL EDITOR / LETTER TO EDITOR
Chinese experience in the management and control of COVID-19 epidemic
Ricardo Silva Rodríguez and Lisset Hermida Cruz
Available from: http://dx.doi.org/10.21931/RB/2021.06.04.2
INTRODUCTION
In December 2019, a new epidemic of coronavirus disease appeared (COVID–19), caused by SARS-CoV-2 (formerly 2019-nCoV). The first reported disease cases occurred in Wuhan, Hubei province, resulting in the third zoonotic event related to lethal human coronavirus. Initially, the incubation period is 1-14 days (mean 5-6 days) in most cases but can be as long as 24 days1,2. The most commonly seen characteristics of COVID-19 are fever, cough, tiredness, and abnormal chest computed tomography3,4. So far, bat is thought to be the origin of SARS-CoV-2, based on sequence homology of 96% between SARS-CoV-2 and Bat-CoV-RaTG135,6,7, but more impartial scientific investigations on the origin-tracing of the virus are required to elucidate the issue. Human-to-human transmission of SARS-CoV-2 occurs mainly via respiratory droplets1, direct contact1, asymptomatic transmission8,9 , and intrafamilial transmission3,4.
At present, there are over 206 million cases of COVID-19 worldwide, with a 4.35 million death toll10. As of 12 August 2021, China had confirmed 94,260 cases with 4636 deaths (mortality rate 5%), and 87,740 recovered cases (93%)11. Other countries, even though they had much more time to prepare for the arrival of the virus, delayed their response and that meant lost control12. While the world is struggling to control COVID-19, China has been a good example of how to control the epidemic, and has shared information with other countries on the management and prevention of the disease. How was that possible?
Non-pharmaceutical and pharmaceutical intervention
COVID-19 began in China just before The Chinese New Year, the country's most important traditional holiday, which is associated with the world's most significant population flow. The New Year's holiday usually lasts about a week, and during that time, the population flow increases by more than 300 million. This population movement increased the transmission of COVID-19; as a result, the situation worsened significantly. The speed of China's response was the crucial factor in stopping transmission of the virus and saving lives. To this aim, China released the genomic sequence of the virus in early January 2020 and adopted a strategy that included a nationwide directive from the central government with governmental oversight, combining non-pharmaceutical and pharmaceutical intervention12,13,14. A series of rigorous measures were applied: a) early detection with an active screening at all levels; b) early diagnostic testing through the creation of new diagnostic tools (nucleic acid RT-PCR assay, gene sequencing, and IgM-IgG serology) aiming at performing as many tests as possible; c) early isolation of positive individuals, suspects, and close contacts, d) early treatment of all patients, those with mild symptoms and those with severe symptoms. Significantly, according to the Chinese experience, all patients should receive treatment15; e) protection of personnel: effectively protecting the most vulnerable groups and social distancing; f) local lockdowns along with systematic measures to control mobility and reallocate resources effectively (e.g., medical resources to the epicenter of the infection in Wuhan). The government implemented policies to support different provinces and patients, classified into different groups according to the severity of the infection. In turn, other measures such as setting up of hospitals, diagnostic algorithms, and specific treatments based on the combination of Chinese and Western medicine were implemented to cope with the virus12,13. Following the Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia (Trial Version 7) released by the National Health Commission and State Administration of Traditional Chinese Medicine (TCM) on 3 March 2020, different treatments were applied according to patient's conditions and careful evaluation of their adverse reactions and contraindications. Among these treatments were antiviral therapy [alpha-interferon, lopinavir/ritonavir, ribavirin (also suggested to be used jointly with interferon or lopinavir/ritonavir), chloroquine phosphate, and arbidol]. Antibiotic drug treatment was also used. In severe and critical-ill patients, effective oxygen therapy, non-invasive or invasive mechanical ventilation, rescue therapy, circulatory support, convalescent plasma treatment, glucocorticoids and immunotherapy were used. As for TCM, over 20 different products were applied according to physical conditions and clinical manifestations of patients16. Finally, the development of effective vaccines, capable of protecting against COVID-19 and its complications, was another tool incorporated into China's strategy to fight SARS-CoV2.
Vaccine development
As early as January 2020, Chinese researchers began to develop vaccines against SARS-CoV2. After one year and a half, five Chinese vaccines are already approved for Emergency use outside China. Of them three are inactivated formulations: 1) BBIBP-CorV (Sinopharm, Beijing), approved in 59 countries; 2) Coronavac (Sinovac), approved in 39 countries; 3) Inactivated Vero cells (Sinopharm, Wuhan), approved in 7 countries; 4) Replicative viral vector vaccine, Ad5-nCoV (Cansino), approved in 8 countries, and 5) Subunit vaccine, RBD-Dimer (Anhui Zhifei Longcom), approved in 2 countries. BBIBP-CorV (Sinopharm Beijing) and Coronavac (Sinovac) have been included in the Emergency Use List of WHO17.
Due to the high effectiveness of the non-pharmaceutical intervention in China, the Chinese vaccine developers had to move abroad to test the efficacy of their vaccines after finishing local phase I and II clinical trials18,19,20,21. This significantly delayed the release of efficacy results compared to the Western vaccines, which have been developed in countries with a high incidence of COVID-19. In the past three months, important audited data have come out validating the Chinese vaccines. In this respect, the efficacy of Sinopharm vaccines was published based on the interim report, including data collected from 40,411 participants in the United Arab Emirates and Bahrain last year22. The efficacy rate vs. symptomatic disease was 78.1% and 72.8% for the Beijing and Wuhan vaccines. In turn, the effectiveness study of Coronavac vaccine in Chile, conducted from 2 February to 1 May 2021, with a cohort of approximately 10.2 million persons, was published23. As a result, the effectiveness was 65.9% for the prevention of COVID-19, 87.5% for the prevention of hospitalization, 90.3% for the prevention of ICU admission, and 86.3% for the prevention of COVID-19–related death. In turn, according to the WHO, Sinovac's vaccine efficacy stands at 51% against symptomatic disease and 100% against severe disease, while Sinopharm's efficacy seems to fare slightly better at 79% against mild and hospitalized disease24.
Although the level of immunogenicity induced by the Chinese inactivated vaccines is lower than that induced by other Western vaccines such as mRNA vaccines25, 26, in line with the difference of the effectiveness rates reported by the Chilean health authorities on 3 August 2021, among the vaccines currently under use in Chile27, this does not mean that inactivated Chinese vaccines are useless. First, the effectiveness rate is still over 50%, and second, the Chinese vaccines can be stored between 2-8º degrees Celsius, making them more feasible for a better cold chain distribution to developing countries, which may not have the required facilities to store large amounts of vaccine at very low temperatures, as is the case of the mRNA vaccines.
Since the beginning of July, COVID-19 cases in Chile started to go down. Based on the last Chilean report about Coronavac's effectiveness, the Chinese vaccine undoubtedly is contributing to this important result. Chile has now fully vaccinated more than 60% of its population, from which 70% are with Coronavac28.
To date, China has exported vaccines to more than 60 countries, with the total amount exceeding 770 million doses29. China has been the first country to work with other countries in the development and production of vaccines against COVID-19. Among these countries, Indonesia, Brazil, the United Arab Emirates (UAE) and Egypt are the first to establish production capacity for the Chinese vaccines29.
Up to 14 August, 2021, China had delivered more than 1.83 billion doses to its population, including 60 million administered to those between the ages of 12 and 17, according to the National Health Commission, being fully vaccinated about 55% of the total population30. Adding together doses administered in China and overseas, these may be the most widely used COVID-19 vaccines globally.
Of course, the development of updated schedules and potentially more effective vaccines is also of upmost priority for China and overseas. Early last month, the National Medical Products Administration had approved 22 COVID-19 vaccines for clinical trials using five different technologies. In summary, four vaccines have received conditional approval and three have been approved for emergency use31.
Concerning the administration of booster doses, one recent report described the potent boosting effect of one additional dose of Coronavac at 6-8 months upon administration of the second dose. According to recent news, in China and other countries using inactivated vaccines, this could be an appealing alternative to increase immunity for those working in risk areas, as well as for elderly32. On the other hand, in China, an alternative route of administration for the Cansino's vaccine was developed: The aerosolized Ad5-nCoV. As a result, two doses of aerosolized Ad5-nCoV elicited neutralizing antibody responses similar to one dose of intramuscular injection. In addition, an aerosolized booster vaccination 28 days after the first intramuscular injection also induced strong IgG and neutralizing antibody responses33.
New Delta variant of SARS-COV2. How to face it?
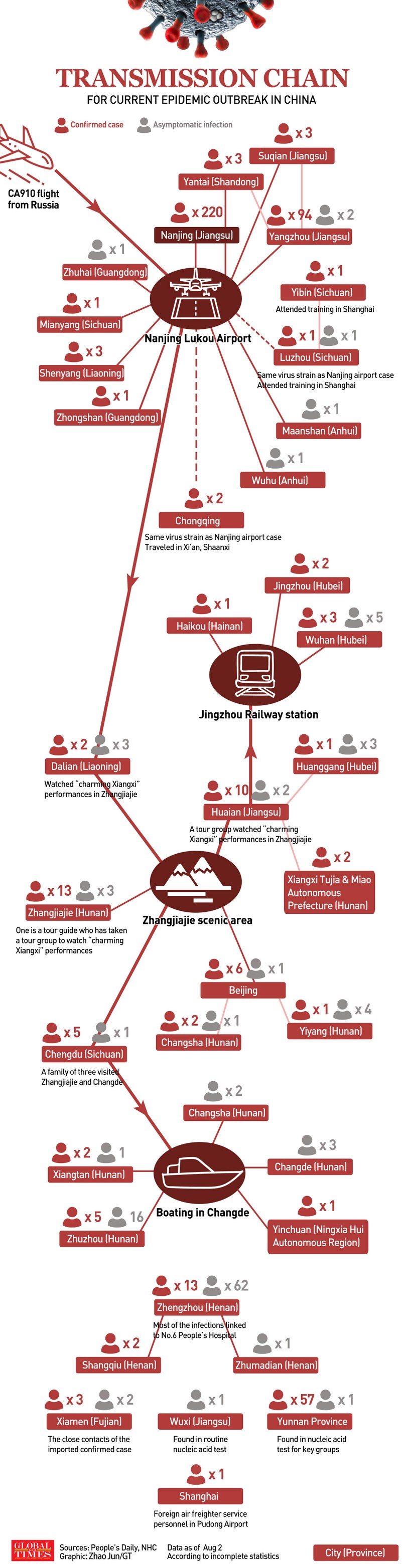
Since first appearing in India in late 2020, the Delta variant of SARS-CoV-2 has become the predominant strain in much of the world. Its fast-spreading nature (people infected with it produce far more virus than do those infected with the original version of SARS-CoV-2) and the combination of this high number of viruses (1000-fold) and a short incubation period (4 days) makes this Delta variant highly transmissible34. Based on the published data about the high viral load associated to infection35, the Delta variant is a real challenge for the current Chinese vaccines. The lack of yet effectiveness data of Chinese vaccines available against this important variant and its spreading around the world, make its control in China a real defiance. The latest COVID-19 surge with the Delta variant started in Nanjing on 20 July upon Nanjing Lukou International Airport's cabin cleaners tested positive for COVID-19 after cleaning an international flight. The recent virus cluster in Nanjing led to the outbreak spreading to other cities and other parts of China, but current epidemic control measures are still effective to prevent the spread of the Delta variant of the COVID-19 virus and the risk of seeing widespread outbreaks across the country is low, according to experts from the National Health Commission's Disease Prevention and Control Bureau30,36. Big data and information technology are important tools to avoid a rebound37. China has used big data to track people who have been in risk areas and are close contacts of new cases. These new, innovative and more specific anti-virus methods have led the Chinese authorities to control the emergence of the Delta variant quickly and effectively (Fig. 1)38. Therefore, to accelerate the pace of vaccination today becomes a must to build the herd immunity in the population and achieve a balance between economic development and efforts to combat the Delta variant of COVID-19. The Chinese strategy is addressed to combine its highly effective non-pharmaceutical intervention with the high vaccination coverage with the current vaccines. According to the Chinese top epidemiologist Zhong Nanshan, from a preliminary analysis of more than 100 patients in Guangdong province, it was shown that Chinese vaccines are effective in preventing the development of pneumonia and severe infection produced by Delta variant. The epidemiologist urged more people to get vaccinated, meaning 80% or more of the population vaccinated in the country to build-up the immune barrier39,40. The priority of vaccination in China is to prevent people from getting ill, but not from getting infected. That's why a proper combination of both interventions constitutes the unique way, in the short-term, to bring the Delta variant spread under control, whereas alternative vaccines and schedules of immunization are coming. The progress of the ongoing rebound of Delta variant in China will tell the last word, although no country is safe until all countries are safe, a statement very clear for China.
Figure 1. Transmission chain for current epidemic outbreak in China. Global Times. 3 August, 2021.
CONCLUSIONS
China offers an appealing solution to other countries and teaches the rest of the world that even the most difficult situations can be overcome. The high level of collaboration between government officials and health experts, has proven to be effective in containing and controlling COVID-19, which is greatly admired by the entire world. Today, normal life has returned in all regions in an orderly manner, including in Wuhan, the epicenter of the epidemic in China. Countries should take experience of China's response to COVID-19 and implement their own prevention and control strategies immediately, as none is exempt from becoming the new epicenter of the virus. In addition, countries must do more to improve their health systems and personnel so as to be more prepared for future outbreaks and reduce their consequences. We recognize the huge contribution of China in the fight against the pandemic and call the international community to join to this fight and strengthen cooperation and solidarity41.
REFERENCES
1. Huang, C. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395, 497–506 (2020).
2. Guan W-J, Ni Z-Y, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020;382: 1708-20.
3. Sun P, Qie S, Liu Z, Ren J, Li K, Xi J. Clinical characteristics of hospitalized patients with SARS-CoV-2 infection: A single arm meta-analysis. [e-pub before print]. J Med Virol https://doi.org/10.1002/jmv.25735.
4. China Centers for Disease Control: China-WHO new coronavirus pneumonia (COVID-19) joint inspection report. Available at: http://www.nhc.gov.cn/jkj/s3578/202002/87fd92510d094e4b9bad597608f5cc2c.shtml. Accessed 29 February, 2020.
5. Zhou P, Yang X-L, Wang X-G, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020; 579:270-3.
6. Wu A, Peng Y, Huang B, et al. Genome composition and divergence of the novel coronavirus (2019-nCoV) originating in China. Cell Host Microbe 2020;27: 325-8.
7. Li X, Zai J, Zhao Q, et al. Evolutionary history, potential intermediate animal host, and cross-species analyses of SARS-CoV-2 [e-pub beforeprint]. J Med Virol https://doi.org/10.1002/jmv.25731.
8. Rothe C, Schunk M, Sothmann P, et al. transmission of 2019-nCoV infection from an asymptomatic contact in Germany. N Engl J Med 2020;382: 970-1.
9. Wang F-S, Zhang C. What to do next to control the 2019-nCoV epidemic? Lancet 2020;395: 391-3.
10. www.worldometers.info/coronavirus/.
11. Latest on the COVID-19 pandemic - Chinadaily.com.cn.
12. www.thelancet.com/infection Vol 20 November 2020.
13. The Asian Economic Panel and the Massachusetts Institute of Technology 2020. https://doi.org/10.1162/asep_a_00800.
14. Lancet. 2020 Feb 22;395(10224):565-574. doi: 10.1016/S0140-6736(20)30251-8.
15. https://enapp.chinadaily.com.cn/a/202003/19/AP5e73422ba3103a24b110b9cb.html.
16. Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia (Trial Version 7) released by the National Health Commission and State Administration of Traditional Chinese Medicine (TCM) on 3 March, 2020.
17. Status_COVID_VAX_15July2021.pdf (who.int).
18. https://doi.org/10.1016/ S1473-3099(20)30831-8. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBIBP-CorV: a randomized, double-blind, placebo-controlled, phase 1/2 trial.
19. https://doi.org/10.1016/ S1473-3099(20)30843-4. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: a randomized, double-blind, placebo-controlled, phase 1/2 clinical trial.
20. https://doi.org/10.1016/ S0140-6736(20)31605-6. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: a randomized, double-blind, placebo controlled, phase 2 trial.
21. https://doi.org/10.1016/ S1473-3099(21)00127-4. Safety and immunogenicity of a recombinant tandem-repeat dimeric RBD-based protein subunit vaccine (ZF2001) against COVID-19 in adults: two randomized, double-blind, placebo-controlled, phase 1 and 2 trials.
22. JAMA. 2021;326(1):35-45. doi:10.1001/jama.2021.8565. Effect of 2 Inactivated SARS-CoV-2 Vaccines on Symptomatic COVID-19 Infection in Adults A Randomized Clinical Trial.
23. DOI: 10.1056/NEJMoa2107715. Effectiveness of an Inactivated SARS-CoV-2 Vaccine in Chile.
24. Microsoft PowerPoint - 2- SAGE critical evidence of Sinopharm.pptx (who.int).
25. https://doi.org/10.1038/s41591-021-01377-8. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection.
26. https://doi.org/10.1016/ S2666-5247(21)00177-4. Comparative immunogenicity of mRNA and inactivated vaccines against COVID-19.
27. Efectividad del Programa de Vacunación contra SARS-CoV-2, Ministerio de Salud de Chile. PowerPoint Presentation (minsal.cl).
28. In China We Trust: Field-testing CoronaVac in Chile | Wilson Center.
29. China COVID-19 Vaccine Tracker - Bridge Consulting (bridgebeijing.com).
30. enapp.chinadaily.com.cn/a/202108/14/AP61172c7ba310f03332fa797f.html.
31. State Drug Administration (nmpa.gov.cn).
32. doi: https://doi.org/10.1101/2021.07.23.21261026.
33. https://doi.org/10.1016/ S1473-3099(21)00396-0. Safety, tolerability, and immunogenicity of an aerosolized adenovirus type-5 vector-based COVID-19 vaccine (Ad5-nCoV) in adults: preliminary report of an open-label and randomized phase 1 clinical trial.
34. www.scientificamerican.com/article/how-the-Delta-variant-spreads-so-quickly1/.
35. Li, B. et al. Preprint at medRxiv https://doi.org/10.1101/2021.07.07.21260122 (2021).
36. www.chinadaily.com.cn/a/202107/31/WS610512bea310efa1bd665ba7.html.
37. www.weforum.org/agenda/2020/03/coronavirus-covid-19-hangzhou-zhejiang-government-response/.
38. www.globaltimes.cn/page/202108/1230490.shtml.
39. www.chinadaily.com.cn/a/202106/27/WS60d864dfa310efa1bd65e2c4.html.
40. Expert gives update on Delta variant, vaccines - Chinadaily.com.cn.
41. http://www.chinadaily.com.cn/a/202108/13/WS6115a0eca310efa1bd6687b7.html.
Received: 25 August 2021
Accepted: 10 October 2021
Dr. Ricardo Silva Rodríguez1 https://orcid.org/0000-0002-4784-0264
Dr. Lisset Hermida Cruz1 https://orcid.org/0000-0001-8470-183X
Representative Office BioCubaFarma, Beijing, China1
Corresponding author. [email protected]
There are no reviews yet.