2023.08.03.31
Files > Volume 8 > Vol 8 No 3 2023
Evaluation of the phytochemical content and antioxidant properties of different extracts of Achillea odorata L.
Hind Amira1 , Hassiba Benabdallah*1
, Hassiba Benabdallah*1 , Walid Mamache1
, Walid Mamache1 , Fatima Benchikh1
, Fatima Benchikh1 , Roumaissa Ounis1
, Roumaissa Ounis1 , Bensouissi Chawki2
, Bensouissi Chawki2 and Smain Amira1
and Smain Amira1
1- Laboratory of Phytotherapy Applied to Chronic Diseases, Department of Biology and Animal Physiology, Faculty of Nature and Life Sciences, University of Setif 1, 19000, Algeria. [email protected] [email protected]; [email protected] ; [email protected] ; [email protected] ; [email protected]
2-Biotechnology Research Center (CRBt), UV 03 BP E73, Nouvelle Ville Ali Mendjli, Constantine, Algeria. [email protected] ()
*Corresponding author: [email protected]
Available from: http://dx.doi.org/10.21931/RB/2023.08.03.31ABSTRACT
The present study reports the chemical profile and antioxidant activity of Achillea odorata L. aerial parts of different extracts obtained using five solvents of varying polarities (decocted, hydro-ethanol, hydro-methanol, hydro-ethyl acetate and hydro-chloroform extracts). The chemical profile and the antioxidant potential were evaluated by measuring the total phenolic content, total flavonoid content, total tannin content, 2,2′-azinobis-3-ethylbenzothiazoline-6-sulphonic acid (ABTS) radical-scavenging, reducing the power of FeCl3 and phenanthroline assays. The highest phenolic content and antioxidant potential were generally shown by the methanol extract (AME). AME exhibited the highest values for total polyphenols content (424.94 ± 0.58 mg gallic acid equivalent /g of dry extract), ABTS (13.46 ± 1.153 µg/mL), reducing the power of FeCl3 (9.918 ± 0.019 µg/mL) and phenanthroline (6.22 ±0.04 µg/mL). These results suggest that A. odorata L. possesses potential antioxidant compounds that could be tested as drug candidates against oxidative-related pathological processes.
Keywords: Achillea odorata L., Antioxidant activity, Free radical scavenging, Polyphenols, Reducing power.
INTRODUCTION
Reactive oxygen species (ROS), byproducts of aerobic metabolism, are critical players in several crucial signaling pathways 1. Robust scientific data, however, demonstrates that high ROS concentrations are harmful and are to blame for DNA mutations, lipid oxidation, and protein peroxidation. According to research, oxidative stress may play a significant role in developing or progressing some diseases, including cancer, diabetes mellitus, inflammatory diseases, and neurodegenerative disorders 2. Humans have discovered natural medicines, primarily plants, for various ailments. As a result, the development of new drugs has long been based on natural compounds 3. Although the pharmaceutical sector has extensively used organic chemistry, natural products are more in demand these days to replace synthetic ones with unfavorable side effects. This has led scientists to revert to nature as a critical source for developing new, safer, and more efficient molecules 4. Because of their antioxidant activity, antioxidants such as polyphenols, vitamins, terpenoids, and nitrogen compounds can significantly impact the body when consumed as fruits, vegetables, aromatic and medicinal plants 5. These large quantities of organic substances, which plants produce, are crucial parts of our food. These substances are valued by nutritionists, consumers, and researchers because they are suitable for human health 6.
One of the Asteraceae family’s more recent evolutionary genera is Achillea odorata L. This plant is a significant medicinal plant since it has been used for various medical conditions, including as a spasmolytic, choleretic, wound therapy, and anti-inflammatory agent 7. More than 100 species of the Achillea genus, which is widespread worldwide, have been identified 8. Most of its distribution is in North America, various regions of Europe, the Middle East, the Mediterranean, Eastern and Western Asia, Australia and New Zealand 9,10. This genus has been used as an anti-inflammatory 11, anti-diabetic 12, anti-rum, stimulating tonic, and for ethnoveterinary treatments (cattle, poultry and dogs) 13,14.
Moreover, previous papers reported the antioxidant properties 14,15 of the Achillea species’ plant extracts and essential oils. Due to the various antioxidant potentials of chemicals with varying polarities, both extraction yield and antioxidant activity of plant-origin extracts primarily rely on the solvent 16. Comparative research is necessary to choose the best solvent for each substrate to maximize antioxidant activity. While using different polarity substances, one can learn more in-depth details about the characteristics of the extracts.
The present study was undertaken to realize for the first time a comparative analysis of the phytochemical content and antioxidant properties of different extracts from A. odorata L. Five solvents were chosen: methanol 50%, ethanol 50%, ethyl acetate 50%, chloroform 50%, and decocted water as extract solvents which permit comparison of the antioxidant properties among the polarity based solvent extracts of medicinal plants. We should note that the ABTS and the reducing power activities of A. odorata L. have never been documented in the literature.
MATERIALS AND METHODS
Plant material
A. odorata L. plant was collected in June 2020 from Jijel (Algeria). The identification and taxonomic classification of the plant were developed by Professor Smain Amira, Department of Animal Biology and Physiology, University Setif 1, Algeria. The voucher number 302 AO 16/06/20 Jij/SA was deposed at the Phytotherapy Applied to Chronic Diseases laboratory. After being thoroughly cleaned, the plant’s aerial part was dried naturally. The air-dried plant was crushed into an excellent powder using an electric grinder.
Chemicals
All chemicals were of analytical grade and purchased from Sigma (Paris, France). All chemicals were analytical-grade and were bought from Sigma and Aldrich.
Plant extraction
The current investigation used decoction and four different solvents to extract chemicals with various polarities. Green chemistry’s typical empirical techniques were used to obtain the decocted extract. To remove polar chemicals, methanol and ethanol were used. Semi-polar chemicals were extracted using ethyl acetate, and nonpolar compounds were extracted using chloroform.
Preparation of decocted extract (ADE)
50 g of the plant powder was boiled for 10 minutes in 500 mL of water and then cooled. The homogenate was obtained and filtered through muslin after the decoction period. The resulting filtrate underwent further filtration using filter paper before being put onto plates and baked in an oven at 38°C 17. The resulting extract (ADE) was then kept at 4°C in a covered bottle to prevent any molecular breakdown from light.
Preparation of hydro-ethanol (AEE), hydro-methanol (AME), hydro-ethyl acetate (AEAE) and hydro-chloroform (AChE) extracts (50%)
The extracts of A. odorata L. were prepared using different solvents with different polarities (methanol, ethanol, ethyl acetate, chloroform) according to Markham 18
However, 50 g of ground material of the plant was macerated in 50% solvent (ethanol, methanol, ethyl acetate, chloroform) at room temperature and protected from light for 72 hours, with maximum agitation. Then, the mixture is filtered and evaporated using a rotary evaporator (BÜCHI) at a temperature of 40-50°C. This mixture was then dried in an oven at 38°C. The extracts obtained (AEE, AME, AEAE, AChE, respectively) were stored at 4°C in a protected bottle.
Phenolics and pigments content determination
Calculation of the plant extraction yield
The yield of the plant extraction is the ratio between the weight of the extract and the weight of the plant to be treated. After extracting the active ingredients from the A. odorata L. plant, the yield is calculated using the following formula (1): 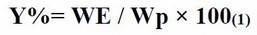
Where: Y= yield of the extract in percentage. WE = weight of the extract in grams. Wp = weight of the plant in grams.
Determination of total phenolic content
Total phenolic content (TPC) was assessed by Folin Ciocalteu reagent 19. A volume of 100 µL of each extract was mixed with 500 µL of Folin Ciocalteu reagent (diluted 10 times). After 4 min, 400 µL of 7.5% Na2CO3 solution was added. The completed mixture was agitated and allowed to react at room temperature in the dark for one hour. The reaction mixture’s absorbance was then measured at 760 nm. A gallic acid standard curve determined different extracts’ total amounts of polyphenols. The results were represented as mg of gallic acid equivalent (GAE) for each gram of dried plant extract.
Determination of total flavonoid content
Total flavonoid content (TFC) was determined using aluminum chloride assay 20. Briefly, 1 mL of AlCl3 (2%) was combined with 1 mL of each examined extract or standard (quercetin). After 10 min of incubation, the absorbance against a prepared blank was measured at 430 nm. Using a calibration curve for quercetin, the results were reported as mg of quercetin equivalent per gram of dry plant extract weight (mg QE/g DE).
Determination of total tannin content
Total tannin content (TTC) was estimated using Folin Ciocalteu reagent (FCR), taking tannic acid as standard 21. The reaction mixture was made by combining 0.5 mL of extract, 2.5 mL of 10% Folin-Ciocalteu reagent mixed with water and 2.5 mL of 7.5% Na2CO3. The same procedure was prepared blank without a sample. The samples were incubated in a thermostat at 45°C for 45 minutes. The absorbance was determined by using a spectrophotometer at λmax = 765 nm. The samples were produced in triplicate for each analysis, and the mean absorbance value was calculated. The calibration curve was created by repeating the procedure using the tannic acid standard solution. The concentration of the TTC was determined (mg/mL) from the calibration line based on the observed absorbance. The total tannin content is expressed in mg tannic acid equivalent (TAE) per gram of dry extract.
Evaluation of in vitro antioxidant activities
ABTS radical scavenging activity
The antiradical activity of the extracts using the ABTS radical was evaluated according to the method of Re et al. 22. Briefly, ABTS cation was generated by reacting 2 mM of ABTS with 2.45 mM of K2S2O8. The mixture was then kept at ambient temperature for 16 hours in complete darkness. Distilled water was used to dilute the ABTS+ solution to an absorbance of 0.700 at 734 nm before use. After mixing 40 µL of each sample at various concentrations with 160 µL of the ABTS+ solution for 10 minutes, the absorbance at 734 nm was measured. The percentage of inhibition was calculated using the following formula (2). Results were compared to the standards BHA and α-tocopherol.
Reducing the power of FeCl3
To estimate the possible reducing power capacity of A. odorata L., the protocol of Oyaizu 23 was used. A total of 40 µL of phosphate buffer (pH = 6.6; 0.2 M) was added to 10 µL of sample solution (extract or standard) in methanol, with various concentrations, followed by the addition of 50 µL of potassium ferricyanide (1%). At 50°C, the mixture was incubated for 20 min. The absorbance was then measured at 700 nm after adding 50 µL of trichloroacetic acid solution (10%), 40 µL of distilled water, and 10 µL of ferric chloride solution (0.1%). The obtained results were expressed as A0.50 values and compared with α-tocopherol.
Phenanthroline assay
To evaluate the phenanthroline antioxidant assay, we used the protocol described by Szydlowska-Czerniaka et al. 24. A total of 50 µL of FeCl3 (0.2%) was mixed with 10 µL of various dilutions of sample solutions (extract or standard) in methanol, followed by the addition of 30 µL of O-phenanthroline (0.5%) and adjusted with 110 µL of methanol. The absorbance at 510 nm was measured following 20 min of incubation at 30°C. The obtained results were expressed as A0.50 values and compared with the used antioxidant positive control (BHA).
Statistical analysis
The results are represented as the mean ± SD (standard deviation). The different IC50 and A0.5 values are calculated using the Graph pad Prism 7 software according to the log (concentration) vs. normalization (absorbance) method. The analysis of variance (one-way ANOVA) was performed to compare the different values, followed by the Tukey test (multiple comparisons). The difference is considered statistically significant at the risk of 5% (P<0.05).
RESULTS
Phytochemical study of the extracts
Extraction yield
Extraction yields obtained for the various extracts are shown in Table 1. AME extract demonstrated the most significant yield of 16.6%, then AQE (16.1%) and AEE (14.9%). While AEEA and AChE showed a lower extraction yield. When combined, the yield using polar solvents (AME, ADE and AEE) was 45.6%. In comparison, the yield from nonpolar solvents (AEAE and AChE) was only 12.2%, suggesting that A. odorata L. extracts had more polar components.
Total polyphenols, flavonoids and tannins content
The present study quantified the contents of total phenolic, flavonoids, and tannins in extracts of A. odorata L. The obtained results are summarized in Figure 1. The methanol extract recorded the highest total phenolic content with a value of 424.94 ± 0.58 mg GAE/g DE, followed by the ADE and AEE with a value of 372.88 ± 1.91 and 349 ± 0.03 mg GAE/g DE, respectively. At the same time, the ethyl acetate and chloroform extracts gave the lowest value (113.78 ± 0.33 and 97.01 ± 0.14 mg GAE/g DE), respectively.
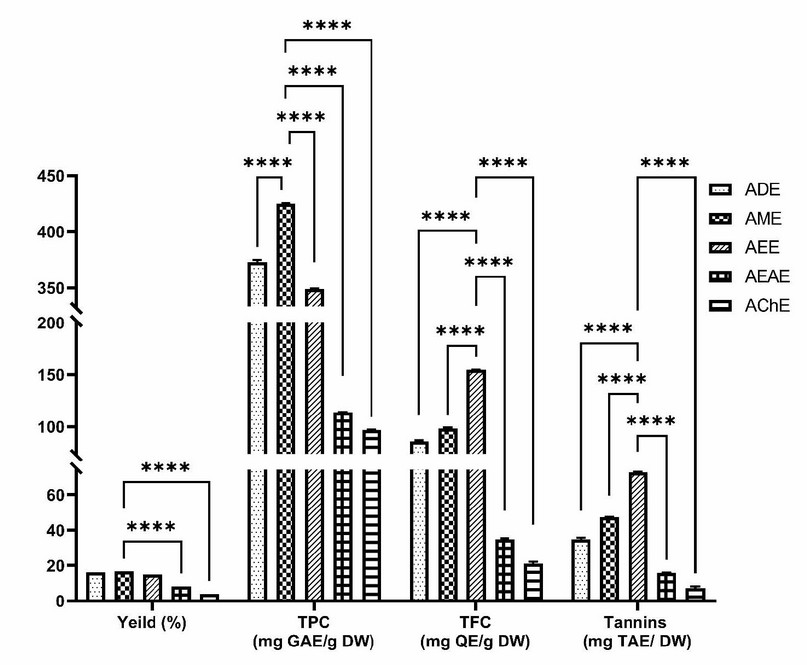
Figure 1. Yield extraction, total phenolics, flavonoids and tannins contents of A. odorata’s extract. ADE: decocted extract, AME: hydro-methanolic extract, AEE: hydro-ethanolic extract, AEAE: hydro-ethyl acetate extract, AChE: hydro-chloroformic extract, GAE: gallic acid equivalent, QE: quercetin equivalent, TAE: tannic acid equivalent, DW: dry weight. results are expressed as means ± sd (n=3). ****; p≤0.0001 compared to AME or AEE.
Furthermore, the AEE showed the highest levels of flavonoids and tannins (154.71 ± 0.35 mg QE/ g DE and 72.62 ± 0.30 mg TAE/ g DE, respectively) followed by the AME (98.33 ± 0.85 mg QE/ g DE and 47.15 ± 0.32 mg TAE/ g DE, respectively), then ADE with value close to the latter extract (86.01 ± 1.02 mg QE/ g DE and 34.87 ± 0.93 mg TAE/ g DE, respectively). Moreover, AEAE and AChE showed a small amount of flavonoids and tannins, as shown in Table 1.
Antioxidant activity
ABTS radical cation decolorization assay
According to the present work, the ability of A. odorata L. extracts to scavenge the radical ABTS is shown in Figure 2. Compared to standards, all extracts exhibited high antioxidant activity and in the following order: AME (IC50 = 13.46 µg/mL) > AEE (IC50 = 15.34 µg/mL) > ADE (IC50 = 16.38 µg/mL) > AEAE (IC50 = 18.84 µg/mL) > AChE (IC50 = 20.25 µg/mL). The AME and AEE showed a significant activity similar to standards (BHA and α-tocopherol).
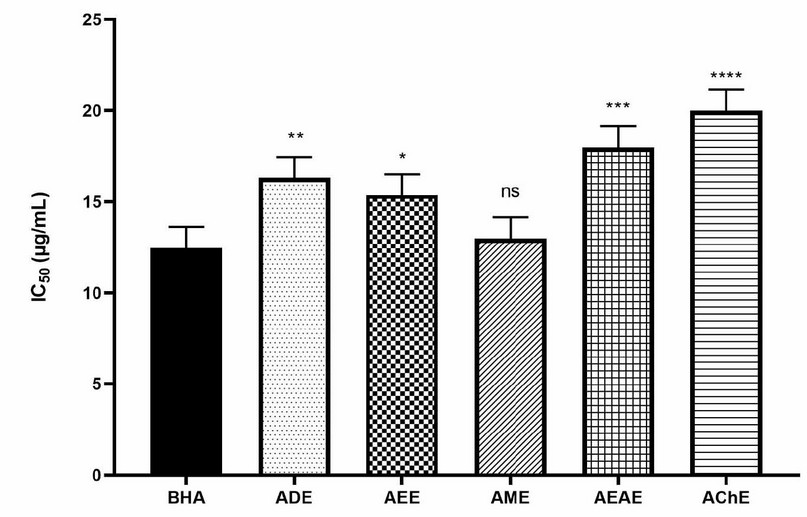
Figure 2. Free radical scavenging (ABTS) activity of A. odorata L. extracts. ADE: decocted extract, AME: hydro-methanolic extract, AEE: hydro-ethanolic extract, AEAE: hydro-ethyl acetate extract, AChE: hydro-chloroformic extract. Data were presented as IC50 means±SD (n=3) (ns: not significant, *; P≤0.05, **; P≤0.01, ***; P≤0.001, ****; P≤0.0001) vs BHA as standard.
Reducing power (reducing power of FeCl3 and phenanthroline)
The reducing power of FeCl3 and phenanthroline activity of the extracts was determined and compared to standard (BHT or α-tocopherol). The results were in the following order of activity for reducing power of FeCl3: AME (IC50 = 9.91 µg/mL) > ADE (IC50 = 19.50 µg/mL > AEE (IC50 = 20.02 µg/mL) > AEAE (IC50 = 24.38 µg/mL) > AChE (IC50 = 39.61 µg/mL) (Figure 3), and in the following order for phenanthroline assay: AME (IC50 = 6.22 µg/mL) > ADE (IC50 = 8.50 µg/mL > AEE (IC50 = 12.16 µg/mL) > AEAE (IC50 = 24.05 µg/mL) > AChE (IC50 = 24.75 µg/mL) (Figure 4). The highest reducing power was shown by AME, which is the best and relatively closer compared with standards.
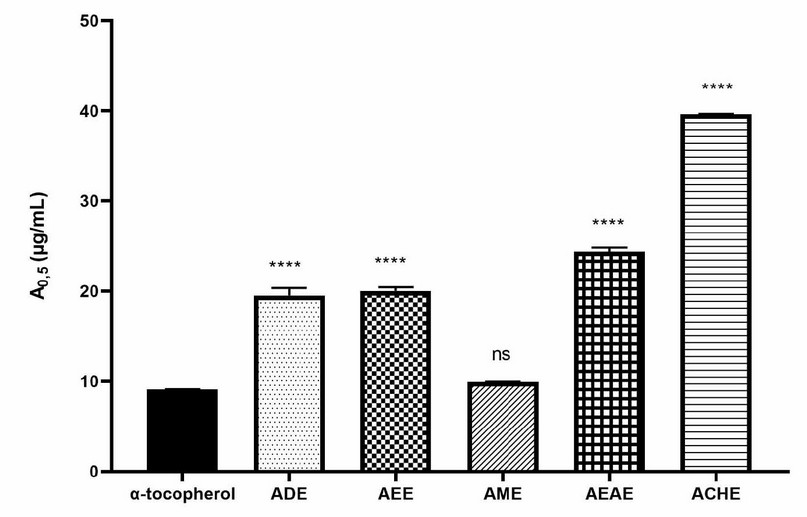
Figure 3. Reducing power of FeCl3 of A. odorata L. extracts. ADE: decocted extract, AME: hydro-methanolic extract, AEE: hydro-ethanolic extract, AEAE: hydro-ethyl acetate extract, AChE: hydro-chloroformic extract. Data were presented as IC50 means±SD (n=3) (ns: not significant, ****; P≤0.0001) vs α-tocopherol as standard.
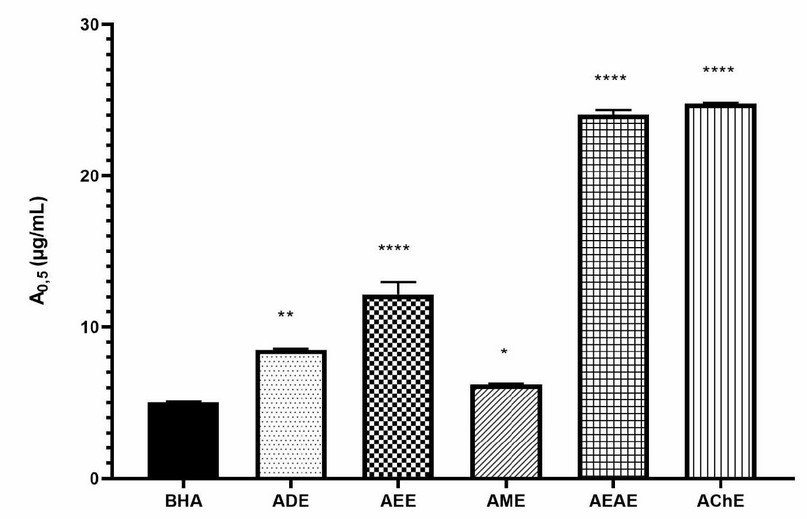
Figure 4. Phenanthroline activity of A. odorata L. extracts. ADE: decocted extract, AME: hydro-methanolic extract, AEE: hydro-ethanolic extract, AEAE: hydro-ethyl acetate extract, AChE: hydro-chloroformic extract. Data were presented as IC50 means±SD (n=3) (ns: not significant, *; P≤0.05, **; P≤0.01, ****; P≤0.0001) vs BHA as standard.
DISCUSSION
Achillea genus is used traditionally for treating many disorders and is viewed as a popular plant in many countries in the world and may be considered a functional food; this feature has been attributed to its high bioactive content and diversity of terpenes, flavonoids, coumarins, phenolic acids7. The distinctive structures and functional groups of plant phytochemicals are known to affect their polarity and solubility in the different extraction solvents used. Thus, the yields would be dependent on such characteristics. Methanol is frequently reported to generate the highest yields when used as an extracting solvent 25. Moreover, the extraction yield for most extract plants was directly proportional to the polarity of solvents. For instance, methanol and chloroform gave the highest and lowest extraction yields, respectively. This finding agrees with Do et al. 26, who demonstrated the superiority of polar solvents with hydroxyl groups such as methanol and water in extracting solid mass from plant samples. In this study, the overall crude extraction yield, which ranged from 4.0% to 16.6% (Table 1), was lower than those reported by Boutennoun et al. 11 (18.2%) and higher than the yield extraction found by Benali et al. 14 (0.37-1.04%) for the essential oil of the same plant. This could be explained by the different concentrations and types of solvents and extraction methods used in those studies.
A. odorata L. extracts were examined for their total polyphenols, flavonoids and tannins; the results revealed that they have high amounts of polyphenols. The highest polyphenol level has been identified in AME. These findings were lower than the previous study realized by Boutennoun et al. 11. These results are in agreement with those of Spingo et al. 33, who suggested that polar solvents are the best-extracting media for polyphenols and flavonoids, which may be due to an increase in polarity of flavonoids upon conjugation through glycosides with hydroxyl groups that enhances their solubility in polar solvents. The chemical makeup of the extraction media used significantly impacts the extraction yield of phenolics and their antioxidant efficiency 28. These factors include plant part/type, genetic makeup of the species, agroclimatic conditions, harvest timing, and post-harvest processing. The type of extraction solvent chosen significantly impacts the accuracy of botanical chemicals separated from plant material 29. For the extraction of phenolic substances in a prior study, polar and nonpolar solvents like methanol, ethanol, acetone, propanol, ethyl acetate, and water were frequently employed 30. The solubility of these phenolic compounds in the solvent used for the extraction procedure affects the recovery of phenolics from plant sources.
Polyphenolic compounds are the primary constituents present in the plant and mainly possess a potent antioxidant activity 34. Hence, the antioxidant activities of the plant sample were evaluated via three complementary techniques: ABTS radical cation decolorization, reducing power and phenanthroline assays to give better conclusive results of the antioxidant properties of the extracts.
The IC50 of ABTS scavenging assay and A0.5 of phenanthroline and reducing power assays of each extract were compared to the IC50 and A0.5 of α-tocopherol or BHA as standards. The parameters IC50 and A0.5 are used to express the antioxidant activity of a molecule. The lower value of IC50 or A0.5 means that the molecule has the highest antioxidant activity. The antioxidants can be classified according to the following scale: The extract that has an IC50 or A0.5 of less than 50 μg/mL is a potent antioxidant, the one that has an IC50 or A0.5 between 50- 100 μg/mL is a powerful antioxidant, the sample that has an IC50 or A0.5 between 101-150 μg/mL is a medium antioxidant, while the extract with an IC50 or A0.5 greater than 150 μg/mL is a weak antioxidant 35. The figures show that all extracts tested in the present study are potent antioxidant agents since their IC50 and A0.5 are less than 50 μg/mL.
The oxidation of ABTS by potassium persulfate results in the stable radical known as the ABTS reagent 36. At 734 nm, the ABTS blue/green chromophore decolorizes due to being scavenged by antioxidant molecules. The drop in ABTS absorbance in the presence of the tested extract shows the proportion of ABTS inhibition. The capacity of antioxidant substances to generate stable radicals is frequently linked to their scavenging abilities 37. BHA and α -tocopherol are potent antioxidants, especially with significant antiradical activity, according to several authors 38,39. The AME and AEE showed considerable activity similar to standards (BHA and α-tocopherol). Typically, extracts produced using solvents with high polarity exhibit more excellent radical scavenging activity. The polar phase of the extract contributes to the inhibition of ABTS radical through simple electron transfer 40. These high radical scavenging potential results could be attributed to the oxygenated monoterpenes dominated in Achillea species 14. The oxygenated monoterpenes possess a solid radical scavenging potential 41.
A reducing power test is frequently used to evaluate an antioxidant’s capacity to give an electron. Numerous studies have demonstrated a clear link between some plant extracts’ reducing power and antioxidant properties. In the reducing power of FeCl3, the presence of antioxidant reductants in the extracts causes a reduction of the Fe3+/ferricyanide complex to its ferrous form. It is monitored by measuring the formation of Perl’s Prussian blue of ferrous form at a wavelength of 700 nm 35. In this study, AME showed the highest reducing power, which is the best and relatively closer than standards. It can be observed from these results that the change in solvent polarity alters its ability to dissolve antioxidant compounds, and it is evident from the above finding that the polar extracts have more activity than the nonpolar ones. However, this is in agreement with the report of Hertog et al. 43 that methanol is a widely used and effective solvent for the extraction of antioxidants and phenolic compounds. Comparing our data with published literature is tricky because studies on the antioxidant activity of A. odorata L. are minor and almost unobtainable.
The composition of phenolic compounds in the samples with many hydroxyl groups may cause a difference in antioxidant activity. These groups were in charge of the significant antioxidant action. In addition, these activities were influenced by the extraction method and circumstances (temperature, polarity, solvent concentration, and time) 44.
CONCLUSION
A. odorata L. collected from the Jijel region (Algeria) showed a considerable quantity of total phenolic compounds and flavonoids. A significant capacity of A. odorata L. extract to scavenge the ABTS free radical was observed. Moreover, a strong reducing power was exhibited. The most potent antioxidant activity was obtained for AME. Our work also reveals that different solvents have different extraction efficiencies, which suggests that when addressing the antioxidant potential of any sample, the solvent effect should be considered. It is generally known that the antioxidant capacity of plants is primarily due to phenolic chemicals found in the plant kingdom. A. odorata L. extracts are good sources of antioxidants; it is, therefore, possible to valorize this plant in the pharmaceutical and food industries.
ACKNOWLEDGMENTS
The Algerian Ministry of Higher Education and Scientific Research (MESRS) has supported this research.
CONFLICT OF INTEREST The authors declare that they have no conflicts of interest. | AUTHOR CONTRIBUTION Hind Amira: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Software; Writing-original draft; Writing-review & editing. Hassiba Benabdallah: Supervision; Validation; Visualization; Writing-review & editing. Walid Mamache: Co-Supervision; Validation; Visualization; Formal analysis; Writing-review & editing. Fatima Benchikh: Conceptualization; Visualization; Writing-original draft; Writing-review & editing. Chawki Bensouici: Data curation; Methodology; Resources; Validation. Romaissa Ounis: Data curation; Formal analysis; Investigation. Smain Amira: Project administration; Validation; Visualization; Writing-review & editing
REFERENCES
1. Qasim, M.; Abideen, Z.; Adnan, M.Y.; Gulzar, S.; Gul, B.; Rasheed, M.; Khan, M.A. Antioxidant properties, phenolic composition, bioactive compounds and nutritive value of medicinal halophytes commonly used as herbal teas. S. Afr. J. Bot., 2017, 110, pp. 240-250. DOI: https://doi.org/10.1016/j.sajb.2016.10.005
2. Ron, M. ROS Are Good, Trends in Plant Science, 2017, 22 (1), pp. 11-19. DOI: https://doi.org/10.1016/j.tplants.2016.08.002
3. Butler, M.S. The role of natural product chemistry in drug discovery. J. Nat. Prod., 2004, 67, pp. 2141-2153. DOI: https://orcid.org/0000-0003-4296-9365
4. Mohammedi, H.; Idjeri-Mecherara, S.; Menaceur, F.; Hassani, A. The effect of solvents and extraction procedure on the recovery of phenolic compounds and the antioxidant capacity of Algerian Bassia muricata L. extracts. Chem. J. Mold., 2019, 14(2), pp. 79-89.
5. Cai, Y.Z.; Luo, Q.; Sun, M. Antioxidant activity and phenolic compounds of 112 Chinese medicinal plants associated with anticancer. Life Sci., 2004, 74, pp. 2157-2184. DOI: https://doi.org/10.1016/j.lfs.2003.09.047
6. Quinones, M.; Miguel, M.; Aleixandre, A. Beneficial effects of polyphenols on cardiovascular disease. Pharmacol. Res., 2013, 68, pp. 25-131. DOI: https://doi.org/10.1016/j.phrs.2012.10.018
7. Rahimmalek, M.; Sayed Tabatabaei, E.; Etemadi, N.; Goli, SAH; Arzani, A.; Zeinali, H. Essential oil variation among and within six Achillea species transferred from different ecological regions in Iran to the field conditions. Ind Crops Prod, 2009, 29, pp. 348-355. DOI: https://doi.org/10.1016/j.indcrop.2008.07.001
8. Goli, S.; Rahimmalek, M.; Sayed Tabatabaei, B. Characteristics and fatty acid profile of yarrow (Achillea tenuifolia) seed oil. Int J Agric Biol, 2008, 10, pp. 355-357.
9. Bremer, K.; Christopher, J. Generic monograph of the Asteraceae-Anthemideae. Bull. br. Mus. nat. Hist. Bot. Botany series, 1993, 23(2), pp. 71-177.
10. Mozaffarian, V. 2002. A Dictionary of Iranian Plant Names; Farhang Moaser Press: Tehran, Iran, 1996, 37 p.
11. Boutennoun, H.; Boussouf, L.; Rawashdeh, A.; Al-Qaoud, K.; Abdelhafez, S.; Kebieche, M.; Madani, K. In vitro, cytotoxic and antioxidant activities of phenolic components of Algerian Achillea odorata leaves. Arab. J. Chem., 2017, 10, pp. 403-409. DOI: https://doi.org/10.1016/j.arabjc.2014.05.013
12. Idm’hand, E.; Msanda, F.; Cherifi.; K. Ethnopharmacological review of medicinal plants used to manage diabetes in Morocco. Clin. Phytoscience, 2020, 6, 18 p. DOI: https://doi.org/10.1186/s40816-020-00166-z
13. Khabbach, A.; Libiad, M.; Ennabili, A.; Bousta, D. Medicinal and cosmetic use of plants from the province of Taza, Northern Morocco. Bol. Latinoam. Caribe Plantas Med, 2012, 11(1), pp. 46-60.
14. Benali, T.; Habbadi, K.H.; Khabbach, A.; Marmouzi, I.; Zengin, G.; Bouyahya, A.; Chamk, I.. GC–MS analysis, antioxidant and antimicrobial activities of Achillea Odorata Subsp. Pectinata and Ruta Montana essential oils and their potential use as food preservatives. Foods, 2020, 9, pp. 668. DOI: https://doi:10.3390/foods9050668
15. Mehlous, S.; Amira, S.; Benchikh, F.; Benabdallah, H.; Mamache, W.; Bensouici, C.; Loucif K. Alkaline DMSO superoxide and radical scavenging, cupric reducing antioxidant capacity (CUPRAC) and polyphenol contents of aqueous and methanol extract from Achilllea santolinoides L. aerial parts. Turk J Food Agric. Sci, 2022, 10(10), pp. 2045-2050. DOI: https://doi.org/10.24925/turjaf.v10i10.2045-2050.5249
16. Moure, A.; Franco, D.; Sineiro, J.; Dominguez, H.; Nunez, M.; Lema, J. Evaluation of extracts from Gevuina avellana hulls as antioxidants. J. Agric. Food Chem., 2000 48, pp. 3890-3897. DOI: https://doi.org/10.1021/jf000048w
17. Perera, N.; Soysa, P.; Abeytunga, T.; Ramesha, R. Antioxidant and cytotoxic properties of three traditional decoctions used for the treatment of cancer in Sri Lanka. Pharmacogn. Mag, 2008, 4, pp. 172-181.
18. Markham, K.R. Techniques of flavonoid identification (A. Press Ed. Academic Press ed.). London, UK: Academic press, London Chap. 1 and 2, 1982, pp. 1-113
19. Li, HB.; Cheng, K.W.; Wong, C.C.; Fan, KW; Chen, F.; Jiang, Y. Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem, 2007, 102(3), pp. 771-776. DOI: https://doi.org/10.1016/j.foodchem.2006.06.022
20. Bahorun, T.; Gressier, B. ; Trotin, F. Oxygen species scavenging activity of phenolic extracts from hawthorn fresh plant organs and pharmaceutical preparations. Arzneimittel-forschung, 1996, 46(11), pp. 1086-1089.
21. Rahate, K.P, Padma, R, Parvathy, NG; Renjith, V. Quantitative estimation of tannins, phenols and antioxidant activity of methanolic extract of Imperata cylindrical. Int. J. Pharm. Sci, 2013, 4, pp. 73-7.
22. Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med., 1999, 26(9-10), pp. 1231-1237. DOI: https://doi.org/10.1016/S0891-5849(98)00315-3
23. Oyaizu, M. Studies on products of browning reaction: Antioxidative activity of products of browning reaction. Jpn. J. Nutr. Diet., 1986, 44(6), pp. 307-315. DOI: https:// doi.org/10.5264/eiyogakuzashi.44.307
24. Szydłowska-Czerniak, A.; Dianoczki, C.; Recseg, K.; Karlovits, G.; Szłyk, E. Determination of antioxidant capacities of vegetable oils by ferric-ion spectrophotometric methods. Talanta, 2008, 76(4), pp. 899-905. DOI: https://doi.org/10.1016/j.talanta.2008.04.055
25. Abrahim, N.; Kanthimathi, M.; Abdul-Aziz, A. Piper betle shows antioxidant activities, inhibits MCF-7 cell proliferation and increases activities of catalase and superoxide dismutase. BMC Complement Altern. Med, 2012, 12, pp. 220. DOI: https://doi.org/10.1186/1472-6882-12-220
26. Do, Q.; Angkawijaya, A.; Tran-Nguyen, P.; Huynh, L.; Soetaredjo, F.; Ismadji, S.; Ju, Y. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. J Food Drug Anal, 2014, 22(3), pp. 296-302. DOI: https://doi.org/10.1016/j.jfda.2013.11.001
27. Singh, M.; Kumar, A. Influence of the solvents on the extraction of major phenolic compounds (punicalagin, ellagic acid and gallic acid) and their antioxidant activities in pomegranate aril. J. Food Sci. Technol, 2014, 51, pp. 2070-2077. DOI: https://doi.org/10.1007/s13197-014-1267-0
28. Dai, J.; Russell, J. Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties. Molecules, 2010, 15, pp. 7313-7352. DOI: https://doi:10.3390/molecules15107313
29. Kalidindi, N.; Thimmaiah, V.; Jagadeesh, N. Antifungal and antioxidant activities of organic and aqueous extracts of Annona squamosa Linn. leaves. J Food Drug Anal, 2015, 23, pp. 795-802. DOI: https://doi.org/10.1016/j.jfda.2015.04.012
30. Alothman, M.; Bhat, R.; Karim, A. Antioxidant capacity and phenolic content of selected tropical fruits from Malaysia, extracted with different solvents. Food Chem, 2009, 115, pp.785-788. DOI: https://doi: 10.1016/j.foodchem.2008.12.005
31. Agati, G.; Azzarello, E.; Pollastri, S.; Tattini M. Flavonoids as antioxidants in plants: location and functional significance. Plant Sci, 2012, 196, pp. 67-76. DOI: https://doi.org/10.1016/j.plantsci.2012.07.014
32. Aneta, W.; Jan, O.; Magdalena, M.; Joanna, W. Phenolic profile, antioxidant and antiproliferative activity of black and red currants (Ribes spp.) from organic and conventional cultivation. Int. J. Food Sci. Technol, 2013, 48(4), pp. 715-726. DOI: https://doi.org/10.1111/ijfs.12019
33. Spigno, G.; Tramelli, L.; de Faveri.; D. Effects of extraction time, temperature and solvent on concentration and antioxidant activity of grape marc phenolics. J. Food Eng., 2007, 81, pp. 200-208. DOI: https://doi.org/10.1016/j.jfoodeng.2006.10.021
34. Fidrianny, I.; Wirasutisna, K.R.; Windyaswari, A.S. Antioxidant capacities of various leaves extract from five colors varieties of sweet potatoes tubers using ABTS, DPPH assays and correlation with total flavonoid, phenolic, carotenoid content. J. Med. Plant Res., 2013, 7(3), pp. 1819-3455. DOI: https://scialert.net/abstract/?doi=rjmp.2013.130.140
35. Dudonne, S.; Vitrac, X.; Coutiere, P.; Woillez, M.; Merillon, J.M. Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem, 2009, 57, pp. 1768-1774. DOI: https://doi.org/10.1021/jf803011r
36. Wootton-Bearda, P.; Morana, A.; Ryan, L. Stability of the total antioxidant capacity and total polyphenol content of 23 commercially available vegetable juices before and after in vitro digestion measured by FRAP, DPPH, ABTS and Folin–Ciocalteu methods. Food Res. Int. (Epub ahead of print), 2010, 44, pp. 217-224. DOI: https://doi.org/10.1016/j.foodres.2010.10.033
37. Zhu, Q.; Hackma, R.M.; Ensunsa, L.; Holt, R.; Keen, L. Antioxidative activities of oolong tea. Journal of Agricultural and Food Chemistry, 2002, 50, pp. 6929-6934. DOI; https://doi.org/10.1021/jf0206163
38. Gulcin, I.; Mshvildadze, V.; Gepdiremen, A.; Elias, R. Antioxidant activity of saponins isolated from ivy: a-hederin, hederasaponin-C, hederacolchiside-E and heder-acolchiside-F. Planta Med, 2004, 70, pp. 561-563. DOI: https://doi: 10.1055/s-2004-827158
39. Prior, R.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem, 2005, 53 (10), pp. 4290-4302. DOI: https://doi.org/10.1021/jf0502698
40. Badawy, M.; Marei, G.; Rabea, E.; Taktak, N. Antimicrobial and antioxidant activities of hydrocarbon and oxygenated monoterpenes against some foodborne pathogens through in vitro and in silico studies. Pestic Biochem Physiol., 2019, 158, pp. 185-200. DOI: https://doi.org/10.1016/j.pestbp.2019.05.008
41. Benzie, I.; Strain, J. The ferric reducing ability of plasma (FRAP) as a measure of “Antioxidant Power”: The FRAP assay. Anal. Biochem, 1996, 239, pp. 70-76. DOI: https://doi.org/10.1006/abio.1996.0292
42. Yen, G.; Wu, S.; Duh, P. Extraction and identification of antioxidant components from the leaves of mulberry (Morus alba L.). J. Agric. Food Chem 1996, 44, 1687 p. DOI: https://doi.org/10.1021/jf9503725
43. Hertog, M.; Hollman, P.; Van de Putte, B. Content of potentially anti-carcinogenic flavonoids of tea infusions, wines, and fruit juices. J. Agric. Food Chem, 1993, 41, pp. 1242-1246. DOI: https://doi.org/10.1016/B978-012543590-1/50019-6
44. Arabshahi-D, S.; Vishalakshi, D.; Urooj, A. Evaluation of antioxidant activity of some plant extracts and their heat, pH and storage stability. Food Chem, 2007, 100(3), pp. 1100-1105. DOI: https://doi.org/10.1016/j.foodchem.2005.11.014
Received: 28 May 2023/ Accepted: 15 July 2023 / Published:15 September 2023
Citation: Amira H, Benabdallah H, Mamache W, Benchikh F, Ounis R, Chawki B, Amira S. Evaluation of the phytochemical content and antioxidant properties of different extracts of Achillea odorata L. Revis Bionatura 2023;8 (3) 31. http://dx.doi.org/10.21931/RB/2023.08.03.31
