2021.06.03.12
Files > Volume 6 > Vol 6 No 3 2021
INVESTIGATION / RESEARCH
Comparative study between lumpy skin disease virus and sheep pox virus vaccines against recent field isolate of lumpy skin disease virus
Nermeen G Shafik1, Heba A Khafagy1, Amal AM1, Ayatollah I Bassiuony2, Farid Fouad Zaki1, Christine A Mikhael1 and Mohamed Samy Abousenna1
Available from: http://dx.doi.org/10.21931/RB/2021.01.03.12
ABSTRACT
Keywords: Lumpy Skin Disease Virus, Sheep Pox Virus, Challenge test, Serological test
INTRODUCTION
The disease's first appear ance was in Africa in 1929, reported from Zambia given the name 'Ngamiland Cattle Disease'. The disease continued to spread and resulted in a panzootic; the first appearance of the disease in Egypt was during two outbreaks in Suez and Ismalia governorates in 1989. In early 2006, a severe LSD outbreak struck foreign (imported from Ethiopia) and local cattle in different Egyptian governorates, causing enormous economic losses; the disease also reappeared in 2012 and 2013 6.
Controlling lumpy skin disease depends mainly on vaccination and vector control. Capripoxviruses are cross-reactive within the genus. Consequently, it is possible to protect cattle against LSD using strains of capripoxvirus derived from sheep or goats in the vaccine 7. Live attenuated strains of capripoxvirus vaccines have been explicitly used for the control of LSD 8. According to these, there are three types of vaccine used against lumpy skin disease, attenuated LSDV (Lumpy Skin Disease) vaccines which provide good protection in cattle, attenuated SPPV (Sheep pox virus) vaccines which have been used in cattle against LSDV in those regions where LSD and SPP are both present as it believed in providing partial protection against lumpy skin disease, and Attenuated Gorgan GTPV vaccine containing GTPV Gorgan strain, this vaccine has been used in those countries where GTP and LSD overlap 9, 10. However, it is recommended to carry out evaluation trials using the most susceptible breeds before introducing a vaccine strain for vaccination into the field.
In Egypt, the control of LSD was applied by vaccination of cattle with SPPV using a Romanian strain for long time which proved to partially tackle the LSD outbreaks with partial cross protection with some notices on its duration of immunity as recommendation for revaccination after 8 months 11, and recently live attenuated LSDV vaccines containing Neethling strain were introduced to be use in Egypt.
The humoral immune response of vaccinated cattle against LSD either by SPPV and LSD vaccines can be evaluated using virus neutralization test (VNT) and ELISA 1, 4, 12, in this study we compare the efficacy of live attenuated lumpy skin disease vaccine and live attenuated sheep pox vaccine against lumpy skin disease virus field isolate, using serological and challenge tests.
MATERIAL AND METHODS
Vaccines
a- Live attenuated Lumpy skin disease virus vaccine (Neethling strain).
Three commercial batches.
b- Live attenuated Sheep pox virus vaccine (Romanian strain).
Three commercial batches.
Three local batches.
These vaccine batches were delivered to Central Laboratory for Evaluation of Veterinary Biologics (CLEVB), Abbassia-Cairo, and evaluated last year for sterility, safety, and potency.
Virus
Lumpy skin disease virus (LSDV):
- Virulent circulating field strain, was supplied by Animal Health Research Institute, the virus was isolated in 2018 13 It was used for the challenge test for vaccinated calves and non-vaccinated control calves.
- Tissue culture adapted strain, the virus was propagated on Madin-Darby bovine kidney (MDBK) cells with a titer 105.5 TCID50 as shown in Figure (1 & 2), it was used for the serum neutralization test.
Cell culture:
The Reference Strain Bank Department supplied Madin-Darby bovine kidney (MDBK) cells in the Central Laboratory for Evaluation of Veterinary Biologics (CLEVB). It was used for the serum neutralization test.
Animals and vaccination
Twenty four susceptible mixed breed apparently clinically healthy calves 6-12 months old free from specific antibodies against LSD virus, were supplied by CLEVB, these calves were divided into eight groups, each group contain three calves, six groups of them were used for vaccination and the other two groups kept as positive control and negative control groups. The six vaccinated groups were divided as follow, three groups of them were vaccinated with field dose of the three batches of live attenuated lumpy skin disease vaccine (one group for each vaccine batch), and the other three groups were vaccinated with field dose of the three batches of live attenuated sheep pox virus vaccine (one group for each vaccine batch), the route of vaccination followed as instruction of manufacture. After 21 days post vaccination serum samples were collected from vaccinated and unvaccinated calves for serological tests (SNT and commercial ELISA).
Challenge test for vaccinated calves
After 21 days from vaccination, the vaccinated groups and positive control (non vaccinated) group were challenged with lumpy skin disease virulent virus according to OIE 1, the animals were shaved at flank region then six log10 dilutions were prepared for inoculation, each dilution inoculated intradermally at four sites for each shaved part (0.1 ml per each inoculum). The challenged animals were kept under observation for seven days after challenge, after that the titer of the challenge virus was calculated for vaccinated and control animals through recording number of lesions per each dilution at inoculated sites (the lesion developed as inflammation and necrotizing fibrinoid cutaneous nodules), the difference in titer between control and vaccinated animals was calculated for each vaccine batch. The negative control was kept without vaccination or challenge.
Serum Neutralization Test (SNT)
It measures the humoral immune response against lumpy skin disease Virus for sera of vaccinated calves, it was applied according to the method described in OIE 1, the neutralizeng titer was calculated according to Reed and Muench 14 . The samples neutralizing antibody titer ≥ 1.10 is considered positive 15.
ELISA IDVIT KIT
ID screen® Capripox Double Antigen Multi-species, REF CPVDA-5A, LOT E83. was used to detect specific antibodies against capripoxvirus. The test was carried out according to the manufacturer's instructions insert. The results of each sample were calculated as aggregate to positive control ratio S/P[AGO20] percentage (S/P %) in (Formula 1). The samples less than 30% are considered negative, while samples higher than or equal to 30% are considered positive.
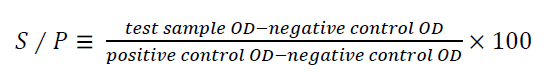
Formula 1. Calculation of S/P percentage.
RESULTS
The mean titers of the live attenuated lumpy skin disease virus vaccine batches and the live attenuated sheep pox virus vaccine batches on tissue culture were recorded. The titer was expressed as log10 tissue culture infective dose50 (TCID50 )/dose, indicated the titers for the three live attenuated lumpy skin disease virus vaccine batches and the three live attenuated sheep pox virus vaccine batches, as shown in table No.1.
Table 1. The titer of virus vaccine batches on tissue culture.
The humoral immune response against lumpy skin disease virus for the sera collected from vaccinated calves after 21 days post vaccination was determined by SNT and ELISA, the lumpy skin disease virus was adapted on MDBK cells for using in serum neutralization test, Figure 1 (A) showing normal MDBK cells and Figure 1 (B) showing cytopathic effect of lumpy skin disease virus on MDBK cells which represent by clustering, cell rounding and degeneration, the results of SNT for serum samples of calves vaccinated by live attenuated lumpy skin disease virus vaccines and live attenuated sheep pox virus vaccine indicated mean neutralizing antibody titer indicated as shown in table No. (2), while the results of ELISA test were calculated as S/P percentage, the mean percentage for the sera of vaccine batches of live attenuated lumpy skin disease virus vaccines and for live attenuated sheep pox virus vaccine batches recorded in table No.(3).
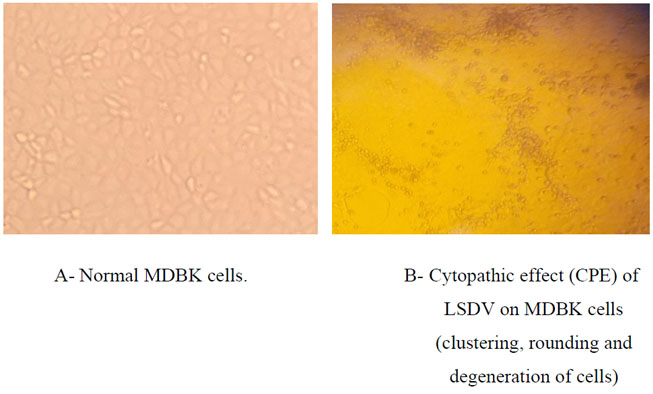
Figure 1 (A and B):Normal MDBK cells and adapted Lumpy skin disease effect on MDBK cell line
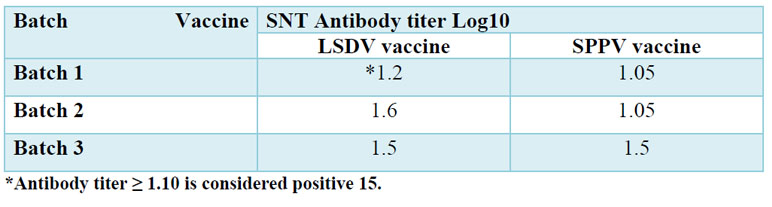
Table 2. Evaluation of humoral immune response in the sera of vaccinated calves by using SNT.
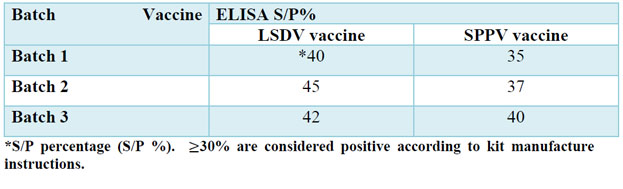
Table 3. Evaluation of humoral immune response in the sera of vaccinated calves using ELISA Kit against Capripox.
Vaccine potency in cattle; the challenge of vaccinated groups and positive control group by virulent lumpy skin disease virus was done, and after seven days, the difference in titer between vaccinated and positive control group was calculated for each group (reading the lesion in inoculated sites), the mean titer for the groups of calves vaccinated with live attenuated lumpy skin disease vaccine and for groups vaccinated with local and imported live attenuated sheep pox vaccines as shown in table No. 4.
Table 4: Challenge of vaccinated calves by LSDV.
DISCUSSION
In Egypt and Middle East live attenuated sheep pox vaccine is used to protect against LSD, although some researches recorded the effectiveness of this vaccine against LSD 16, there are some other recorded that it may offer incomplete protection against LSD 17, 18, 19, 20, recently live attenuated LSD vaccine was introduced to be applied in Egypt, but we still need many researches to investigate and compare the efficacy and adverse reactions between live attenuated LSD virus vaccine and other Capripox virus vaccines.
In this study we compare the efficacy of live attenuated LSDV vaccine and live attenuated sheep pox virus vaccine against LSD in calves as the main host for the disease, using three batches for each vaccine type, showing the vaccines batches titer, evaluate humoral immune response of vaccinated calves (using SNT and ELISA) and challenge vaccinated calves by virulent strain of LSDV.
The titers of live attenuated LSDV vaccine batches per dose were satisfactory where the recommended field dose for LSDV in the vaccine is log10 3.5 TCID50 and minimum protective dose is log10 2.0 TCID50. The titers of live attenuated SPPV vaccine batches were also satisfactory where the recommended dose of sheep pox virus in the vaccine for cattle is log10 3 TCID50 1.
The SNT was carried out to detect the neutralizing antibody titer against LSDV for sera of vaccinated calves after 21 days post vaccination, the results for live attenuated LSDV vaccine batches indicated that all the three batches reached to positive neutralizing antibody titer ≥1.10 15, and the results for the live attenuated SPPV vaccine batches indicated that only the batch (3) reached positive neutralizing antibody titer ≥1.10 while the other two batches (1 and 2) didn’t record the positive titer. These results reflect that live attenuated LSDV vaccine achieved best results in titer and number of batches reaching the positive titer, but we must put in consideration if the antibody is low it doesn’t reveal that the animal isn’t definitely protected as commented by OIE1 which clarify the conflict in results of SNT and challenge test as this disease mainly depend on cellular immunity and the VNT or SNT only tests the positivity. Neutralization test is used for evaluation of humoral immune response for LSDV as recommended by OIE, as it was applied by other similar studies for evaluation of serum samples of cattle vaccinated by live attenuated SPPV vaccine against LSDV 16, also applied by Christine 21 who used it in evaluation calves vaccinated by Attenuated Sheep Pox and Inactivated Lumpy Skin Disease Vaccines against Lumpy Skin Disease, and used in evaluation of the cross-protection between Sheep pox and bovine Lumpy skin vaccines in vaccinated sheep 22.
ELISA kit against Capripox is also used in the evaluation of humoral immune response for the sera of vaccinated calves ( this kit is not specific for LSDV only but it against genus capripox, on the other hand the SNT is specific against LSDV only), the results for live attenuated LSDV vaccines batches indicated positive results ≥30% (as mentioned in ELISA kit) for the three batches, also live attenuated SPPV vaccine indicated results ≥ 30% for the three batches, but we noticed that the percentages for live attenuated LSDV vaccines batches is higher than percentages of live attenuated SPPV vaccine batches. Although the neutralization test is the standard test for evaluation of humoral immune response for LSDV as recommended by OIE, there are other studies recommending to use commercial ELISA, commercial Capripox Double Antigen ELISA (ID.Vet) was used in evaluation of serological tests for detection of antibodies against LSDV and find strong correlation between VNT/MDBK and ELISA 23, also ELISA method and neutralization test were used for detection antibodies against LSDV24.
Challenge test is used for testing potency of the vaccines, and it was carried out in vaccinated calves (as a main host for LSDV) by calculating the difference in titer (between control positive and vaccinated challenged calves in each group) of the inoculum (virulent LSDV) which developed specific lesion, the results for live attenuated LSDV vaccines batches indicated a protective titer for the three batches where the minimum protective result is >log10 2.5 as recommended by OIE 1, and for the live attenuated SPPV vaccine batches indicated only first and third batches are protective with border titer limit but second batch didn’t reach protective titer, and that indicated that live attenuated LSDV vaccine is more protective than live attenuated SPPV vaccine against LSDV. These results agreed with Hamdi 22 who used virulent LSDV in challenge of two groups of calves, one group vaccinated with Romanian SPPV vaccine and the other with Neethling LSDV vaccine and reported that Romanian SPPV vaccine provide partial protection against LSDV while Neethling LSDV vaccine provide fully protection against LSDV, also recombinant LSDV vaccine (LSD-Rift Valley Fever.mf vaccine) was used in two groups of calves, one group challenged by virulent LSDV and other group by virulent Rift Valley Fever Virus and recorded that tested vaccine is safe and protective for both diseases25, and Neethling vaccine is reported significantly more effective than x10 RM65 Sheep Pox vaccine strain in preventing LSD morbidity26.
CONCLUSION
It was believed that the wide cross protection within Capripoxvirus genus allows using of any Capripoxvirus isolate as an effective vaccine against LSDV, otherwise and avoiding this confirmation bias, it is clear from the relevant results that the potential using of Neethling LSDV vaccine against LSDV is superior for combating and prevention the Lumpy skin disease. Also it was be recommended to evaluate both homologous or heterologous virus strain vaccines against LSD in cattle, as it the main host of the lumpy skin disease.
Conflict of interest
The authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
Institutional Animal Care and Use Committee at Central Laboratory for Evaluation of Veterinary Biologics acknowledge the research manuscript, and it has been reviewed under our research authority and is deemed compliance with bioethical standards in good faith.
Author Contributions
NGS designed experiments, AAM and MSA the experiments were performed HAK, AAM AIB, CAM, and MSA. Data analysis was accomplished by NGS, FFZ, HAK, AAM, and MSA. AAM,AIB and MSA wrote the manuscript.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
Data Availability
All data generated or analyzed during this study are included in this published article
REFERENCES
1- OIE. Organization for Animal Health. Lumpy Skin Disease. En: OIE. World Organization for Animal Health. Manual of diagnostic tests and vaccines for terrestrial animals (mammals, birds, bees). Paris:OIE; 2018. p. 1158-1171. Available from: https://www.oie.int/.
2- FAO (Food and Agriculture Organization of the United Nation) (2017): FAO Animal Production and Health (20), Lumpy Skin Disease, a field manual for veterinarians.
3- Norian RN, Afzal NA, Varshovi HR , and Azadmehr A. Comparative efficacy of two heterologous capripox vaccines to control lumpy skin disease in cattle, Bulgarian Journal of Veterinary Medicine, 2019, 22, No 2, 171–179.
4- Yasser FE, El-Tholoth M, Saher S, EL- Said A.A, Mohamed AS, and Younis EE. Investigation of lumpy Skin Disease virus infection in young calves from cows vaccinated with sheep poxvirus vaccine..(2015); 5thinternational conference of virology 112.
5- Tuppurainen ES, Stoltsz WH, Troskie M, Wallace DB, Oura CA, Mellor PS, Coetzer JA, Venter EH. A potential role for ixodid (hard) tick vectors in the transmission of lumpy skin disease virus in cattle. Transbound Emerg Dis. 2011 Apr;58(2):93-104. doi: 10.1111/j.1865-1682.2010.01184.x. Epub 2010 Nov 30. PMID: 21114790.
6- FAO Regional Office for the near east (FAO-RNE) .Emergence of lumpy skin disease in the eastern Mediterranean Basincountries . VOL. 29. (2013) Rome. Available at: http://www.fao.org/3/a-i6155e.pdf.
7- Coakley W. & Captickp B. Protection of cattle against lumpy skin disease. Factors affecting small scale production of tissue culture propagated virus vaccine. Res. Vet. Sci.,(1961); 2, 369–371.
8- Brenner J, Haimovitz M, Oron E, Stram Y, Fridgut O, Bumbarov V, Kuznetzova L, Oved Z, Waserman A, Garazzi S, Perl S, Lahav D, Edery N. & Yadin H. Lumpy skin sease (LSD) in a large dairy herd in Israel. Isr. J. Vet. Med.(2006), 61, 73–77.
9- Tuppurainen ES, Pearson CR, Bachanek-Bankowska K, Knowles NJ, Amareen S, Frost L, Henstock MR, Lamien CE, Diallo A, Mertens PP. Characterization of sheep pox virus vaccine for cattle against lumpy skin disease virus. Antiviral Res. 2014 Sep;109(100):1-6. doi: 10.1016/j.antiviral.2014.06.009. Epub 2014 Jun 25. PMID: 24973760; PMCID: PMC4149609.
10- Abutarbush, S.M. Lumpy Skin Disease (Knopvelsiekte, Pseudo-Urticaria, Neethling Virus Disease, Exanthema Nodularis Bovis). In: Bayry J (eds.) Emerging and Re-emerging Infectious Diseases of Livestock. Springer International Publishing, Gewerbestrasse 11, 6330 Cham, Switzerland, pp: 309-326. (2017).
11- Ayatollah IB, Christine AM and Amal A. Comparison between field and laboratory immune response of cattle vaccinated with sheep pox vaccine against lumpy skin disease. Egypt vet. Med. Assoc. Journal (2015):Vol 75, no (4):649-662.
12- Christine AM, Ibrahim MM, Manal A, Soad MS and Michael A. Comparative study on the efficacy of some capripox vaccines in protection against Lumpy Skin Disease. SCVMJ,(2014); XIX (2):177-189.
13- Hodhod A, Elgendy E, Abd El-Moniem MI and Ibrahim MS. ISOLATION AND MOLECULAR CHARACTERIZATION OF LUMPY SKIN DISEASE VIRUS IN EGYPT DURING 2017- 2018, ejpmr, (2020),7(1), 96-103.
14- Reed LJ and Muench H . A simple method of estimating fifty per cent endpoints. Am. J. Hyg.(1938). 27: 493-497.
15- Milovan Milovanovic, Klaas Dietze, Vesna Milicevic, Sonja Radojicic, Miroslav Valcic, Tom Moritz, and Bernd Hoffmann. Humoral immune response to repeated lumpy skin disease virus vaccination and performance of serological tests. BMC veterinary research 2019 (15:80). https://doi.org/10.1186/s12917-019-1831-y.
16- Shaimaa El-gbily, Emad M. Al-Ebshahy, Heba A. AbdelHady, AboulSoud, E.A. and Samy A. Khalil. Evaluation of Humoral Immune Response Against Lumpy Skin Disease Virus in Cattle Vaccinated with Sheep Pox Virus Vaccine. AJVS. (2019).Vol. 61 (1): 128-132.
17- Ali AA, Esmat M, Attia H, Selim A and Abdelhamid YM. Clinical and pathological studies on lumpy skin disease in Egypt. Vet. Rec.(1990) 127, 549–550.
18- Omyma ME. Recent isolation and identification of lumpy skin disease virus from cattle in Egypt. Egypt J.Comp. Path and Clin. Path. (2008).21,(1): 139-147.
19- Brenner J, Bellaiche M, Gross E, Elad D, Oved Z, Haimovitz M, Wasserman A, Friedgut O, Stram Y, Bumbarov V, Yadin H. Appearance of skin lesions in cattle populations vaccinated against lumpy skin disease: statutory challenge. Vaccine. 2009 Mar;27(10):1500-1503. DOI: 10.1016/j.vaccine.2009.01.020.
20- Somasundaram MK. An outbreak of lumpy skin disease in a holstein dairy herd in Oman: a clinical report. Asian J. Anim. Vet. Adv.2011; 6, 851–859.
21- Christine AM, Ibrahim MM and Saad MA.. Efficacy of Alternative Vaccination with Attenuated Sheep Pox and Inactivated Lumpy Skin Disease Vaccines against Lumpy Skin Disease. SCVMJ,(2016) XXI (2), 125-142.
22- Hamdi J, Zahra B, Mohammed J, Zineb B, Khalid O T, Ouafaa FF and Mehdi E. Experimental evaluation of the cross-protection between Sheep pox and bovine Lumpy skin vaccines. J. Scientific Reports (2020) 10, Art. No.8888, P.1-9. https://doi.org/10.1038/s41598-020-65856-7.
23- Krešić N, Šimić I, Bedeković T and Acinger-Rogić Ž Lojkić I. Evaluation of Serological Tests for Detection of Antibodies against Lumpy Skin Disease Virus. J Clin Microbiol. 2020 Aug 24;58(9):e00348-20. doi: 10.1128/JCM.00348-20. PMID: 32434783; PMCID: PMC7448653.
24- Samojlović M, Polaček V, Gurjanov V, Lupulović D, Lazić G, Petrović T, and Lazić S. Detection of antibodies against Lumpy skin disease virus by Virus neutralization test and ELISA methods. Acta Veterinaria (2019) 69, 1, 47-60, Available From: Sciendo https://doi.org/10.2478/acve-2019-0003.
25- Wallace DB, Mather A, Kara PD, Naicker L, Mokoena NB, Pretorius A, Nefefe T, Thema N, Babiuk S. Protection of Cattle Elicited Using a Bivalent Lumpy Skin Disease Virus-Vectored Recombinant Rift Valley Fever Vaccine. Front Vet Sci. 2020 May 19;7:256. doi: 10.3389/fvets.2020.00256. PMID: 32509806; PMCID: PMC7248559.
26- Ben-Gera J, Klement E, Khinich E, Stram Y, Shpigel NY. Comparison of the efficacy of Neethling lumpy skin disease virus and x10RM65 sheep-pox live attenuated vaccines for the prevention of lumpy skin disease - The results of a randomized controlled field study. Vaccine. 2015 Sep 11;33(38):4837-42. doi: 10.1016/j.vaccine.2015.07.071. Epub 2015 Aug 1. PMID: 26238726
Received: 1 May 2021
Accepted: 9 July 2021
Nermeen G Shafik1, Heba A Khafagy1, Amal AM1, Ayatollah I Bassiuony2, Farid Fouad Zaki1, Christine A Mikhael2 and Mohamed Samy Abousenna1
1 Central Laboratory for Evaluation of Veterinary Biologics (CLEVB), Agricultural Research Center, Cairo, Egypt.
2 Veterinary Serum and Vaccine Research Institute, Pox department, Agricultural Research Center, Cairo, Egypt.
Corresponding author: Mohamed samy Abousenna and Aml AM
https://orcid.org/0000-0003-2202-9544
